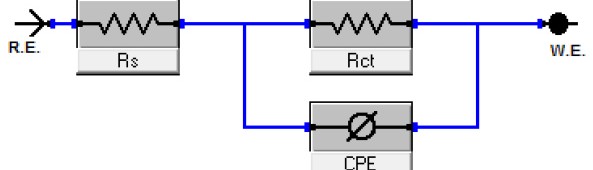
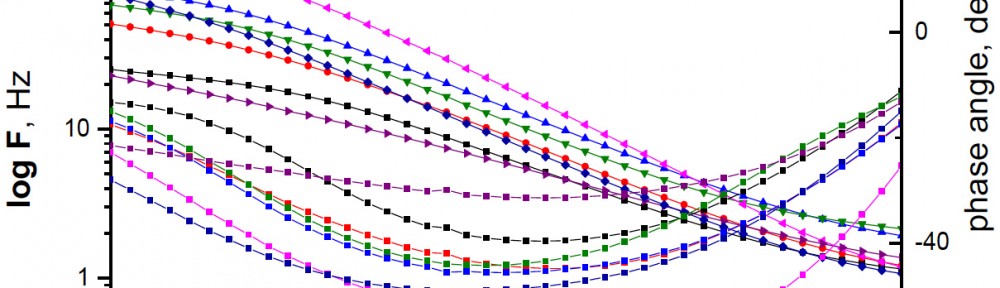
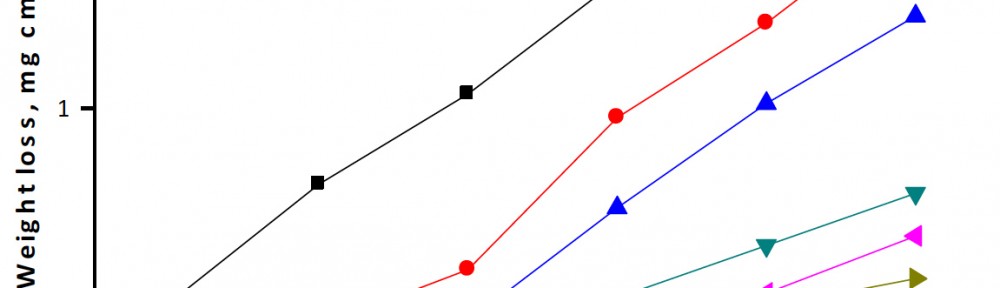
The efficiency of plant extract as corrosion extract for copper in 1M HNO3 medium was carried out using weight loss, potentiodynamic polarization, electrochemical impedance spectroscopy (EIS) and electrochemical frequency modulation (EFM) techniques. The results showed variation in inhibition performance of the extract with varying concentration, immersion time and temperature. Langmuir isotherm was tested to describe the adsorption behavior of the extract on the copper surface. Potentiodynamic polarization study clearly revealed that this extract acts as a mixed type inhibitor i.e. the addition of the extract enhances both cathodic and anodic reactions. The results of the electrochemical impedance study showed a decrease in double layer capacitance and an increase in the charge transfer resistance. The results showed that rosmarinus extract could play significant role as corrosion inhibitor for copper in 1M HMO3.
Author Archives: Azza A. Attia (Assistant Professor)
Allium sativum (Garlic) as green corrosion inhibitor for low carbon steel in HCl solutions

The use of allium sativum (garlic) as a green inhibitor for the corrosion of low carbon steel in 1M HCl has been studied using potentiodynamic polarization, electrochemical impedance spectroscopy (EIS), electrochemical frequency modulation (EFM) techniques and weight loss measurements. Allium sativum has been proved to be good inhibitor. This reduction in the corrosion rate was due to the formation of an external layer formed by S-containing film present in the extract which was adsorbed physically on the metal surface. Allium sativum acted as a mixed type of inhibitor. The inhibition efficiency increases with increasing the inhibitor concentration, but decreases with raising the temperature. The adsorption of allium sativum on the metal surface follows Temkin’s adsorption isotherm. From EFM the causality factors are very close to theoretical values which indicate that the measured data are of good quality. Nyquist plots show a single capacitive loop in uninhibited and inhibited solutions.