Incredible claims of electroplating in materials synthesis lies in tailoring its property by proper modulation of the bath composition and operating parameters, such as current density (c.d.), pH and temperature. Electroplating of metals/ alloys is one of the most complex process because of the unusually large number of critical elementary phenomena involved during deposition. Due to lack of quantitative guiding principles to develop a coating of desired property, it is very difficult and time consuming to optimize the bath composition. Even though Hull Cell method is an established method to optimize a bath, in terms of its constituents and operating parameters its application is limited to know only the effect of c.d. on deposit patterns; and is incapable for predicting the desired properties of the coating, like hardness, reflectivity, thickness etc. In this direction, this paper describes Taguchi’s statistical method for optimization of deposition conditions of Ni-P alloy, using Minitab 16, Statistical software, by reducing the number of experiments to a practical level. In the present study, bath variables, i.e., [glycerol], c.d. and pH of the bath are taken as chosen parameters and micro-hardness and thickness of the coatings as parameters for characteristic performance. Experimental conditions were optimized to maximize the coating properties. Taguchi’s method demonstrated that the basic Ni-P bath, having [glycerol] = 20 mL L-1, c.d.= 4.0 A dm-2 and pH = 8.0 as ideal for developing coatings of highest micro-hardness and thickness. Experimental data revealed that both [glycerol] and c.d. have close dependency on thickness and micro-hardness of coating, compared to pH of the solution. The experimental steps followed for applying Taguchi’s method, for tailoring the deposit characters are discussed with Tables and Figures.
1. Introduction
Electroplating is an extremely important technology, and is concerned with covering inexpensive and widely available base materials with plated layers of different metals/alloys with superior properties to extend their use to applications which otherwise would be prohibitively expensive [1]. Generally electrodeposited alloys usually have a better appearance than the parent metals, being smoother, brighter, and finer grained. It is further asserted that relative to the single metals involved, alloy deposits can have different properties in certain composition ranges. They can be denser, harder, more corrosion resistant, more protective of the underlying base metal, tougher and stronger, more wear resistant, superior with respect to magnetic properties, more suitable for subsequent electroplate overlays and conversion chemical treatment, and superior in antifriction applications etc. [2]. Further, alloy deposition is almost as old and art and/or science as is the electrodeposition of individual metals, and is a subject of same scientific principles as individual metal plating. The subject of alloy electroplating is being dealt with an ever increasing number of scientific publications. The reason for this is due to the vastness of the number of possible alloy combinations and the concomitant possible practical applications. From the literature, it is well recognized that, more often than not, alloy deposition provides properties not attainable by employing electrodeposition of single metals.
In principle, electroplating of a metal/alloy is not a simple dip and dunk process. It is probably one of the most complex process known because of the unusually large number of critical elementary phenomena, or process steps involved [1]. Generally a practicable method for electrodepositing any alloy involves three steps. The first step is concerned with the development of a suitable plating bath. This requires a practical knowledge on the electrochemistry of elements, the solubility of their salts, and the chemistry of their complexes. Since all necessary information on process of alloy deposition are usually not available, the researcher must draw on the analogy with known successful procedures for deposition as a guide, and to make a large number of electrolytic experiments on solutions. Such a research may be partly empirical, and therefore a tendency exists to ignore it. Hence, due to lack of some quantitative guiding principles, the electroplaters are required to rely on some semi-empirical procedures upon which generalizations and theories can be built [2].
In this regard, Hull Cell (named after its inventor Richard Hull) has been brought into practice in electroplating industry as early as 1939 [3]. The Hull Cell is a miniature plating unit designed to produce cathode deposits on a panel that correlates the characteristics of the plating bath being evaluated. Interpretation of the ‘as plated’ cathode panel give rapid information about brightness levels, irregular plate deposits, uniformity of deposits, coverage, throwing power, impurities and plating bath chemistry. Depending upon the bath chemistry as analyzed, condition of the panel relative to brightness, hardness, uniformity, burning, etc., modification by controlled additions can be made to the Hull Cell plating solution and procedures can be repeated. Changes caused by addition to the Hull Cell will duplicate results to be expected by the same proportionate additions to the main plating bath. Correlations of properties, such as hardness and thickness of the coatings in the given time, temperature and current density (c.d.) will also tell the optimum plating range to obtain plating of desired properties. Within the parameters of recommended operating characteristics of a particular plating solution, the Hull Cell will duplicate what is actually occurring in the main plating unit. Correlation of the ‘as plated’ panel and the ‘Hull Cell Scale’ allows rapid, non-destructive testing of plating solutions for research, preventative maintenance, troubleshooting, and quality control.
Even though Hull cell can give information about the operating window, i.e., current density range, it is insufficient to control or decide other process parameters. Apart from that, Hull Cell optimization is found to be laborious in minimizing the effect of causes of variation in a response influenced by multi-variables [3-4]. Hence, to optimize process parameters the experimental design methods like Taguchi’s method is widely accepted because of its competency to overcome the demerits of conventional methods such as, complexity, large number of experiments with increasing the number of process parameters and failure to reach the desired objectives [5]. Taguchi’s statistical method for optimizing the process parameters of multivariable system was developed by Genichi Taguchi in 1966. It was designed to maximize the efficiency of industrially manufactured products [6]. This method uses a special set of arrays called orthogonal arrays. These standard arrays stipulate the way of conducting the minimal number of experiments which could give the full information of all the factors that affect the performance parameter. The crux of the orthogonal arrays method lies in choosing the level combinations of the input design variables for each experiment. This method is a simple and effective technique to optimize manufacturing processes in order to obtain the optimal performance. Nowadays, engineering, pharmaceutical, and biotechnology industries are frequently applying this statistical tool [7, 8]. Basically Taguchi’s method uses a special design of orthogonal arrays that allows studying the whole parameter space with a limited number of experiments [8]. This method has many advantages, like: i) it is an economically feasible method for studying the variability of the response variable, ii) shows the best way to find out the optimum process conditions during laboratory experiments, iii) it is an important tool for improving the productivity of the R&D activity and iv) it can be applied to any process [9, 10].
Since electrodeposition is very complex process, its reproducibility depends on the level of impurity, producer of chemicals and power source. Further, the nature of deposits are controlled by many parameters, like bath composition and operating parameters (like c.d., pH, agitation, temperature etc.), Taguchi’s method is a suitable method to statistically analyze thin film coatings, and to understand the impact of operating variables upon the properties of coatings. Accordingly, Taguchi’s approach has already been used for optimization of bath conditions for development of various thin film coatings, like Cu-In-Se2, TiN, Ni etc [6-9]. Thus by knowing the limitations of Hull cell (as the tool to know only the effect of c.d. on the nature of the deposit) and the widespread application of Taguchi’s statistical method for optimizing the conditions for industrial process, it has been tried to optimize the bath conditions to electrodeposit Ni-P alloy of highest hardness and thickness. It may be noted that in the present study, the chosen parameters are bath variables and optimal design is the development of coatings of specific properties. Hence the basic bath was optimized primarily through standard Hull cell method, and then the process parameters were optimized using Taguchi’s statistical method to develop the coatings of both maximum hardness and thickness, and experimental results are discussed as below.
2. Experimental
2.1 Hull cell – As invaluable analytic tool in electroplating
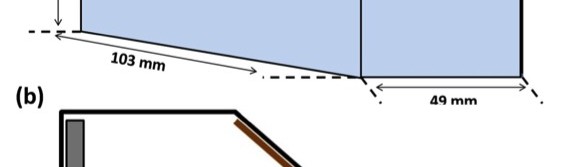
Because a Hull cell produces a deposit that is a true reproduction of the electroplate obtained at various c.d.’s within the operating range of a particular system, it allows experienced operators to determine multiple process parameters, such as brightness, concentration of primary bath components and additives, presence of metallic and organic impurities, effects of temperature and pH etc [3, 11]. Hence the proposed Ni-P alloy bath has been primarily optimized by conventional Hull cell method. The schematic diagram of Hull cell (267 mL) used in the present study is shown in Figure 1. It is may be seen that Hull cell is a miniature electrodeposition tank, in which the cathode is angled with respect to the anode as shown in Figure 1 (b). As a result, when a voltage is applied across the anode and cathode, the resulting c.d. will vary along the length of the cathode, being highest at the point it is closest to the anode. In this way, one can, within a single test run, assess the effect of varying c.d. Accordingly, after such a run, the cathode was removed and inspected. At the highest c.d.’s, the deposit was found to be burned. At the lowest c.d.’s, no deposition was observed. Based on the visual observation of plated panels the operating window of the c.d. required for the proposed bath was determined, using the Hull cell scale.
Thus Hull cell test is very simple to use, and does not require advanced technical training for interpretation of results. It was used to assess any factor that produces a change in the property of the electrodeposits over a wide range of c.d.’s, and is considered as invaluable analytical tool for determining the influence of any parameter on the appearance of the deposit [11].
2.2 Basic bath optimization
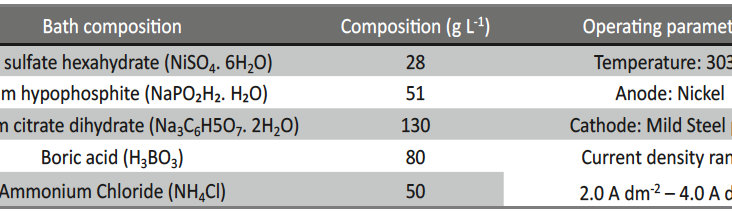
The basic bath composition for electrodeposition of Ni-P alloy was formulated by using conventional Hull Cell method. The bath constituents and general operating conditions are given in Table 1. Nickel sulphate and sodium hypophosphite were used as the primary sources for nickel and phosphorous. Tri-sodium citrate was used as the complexing agent, boric acid as buffer, ammonium chloride as conducting salt and glycerol as additive to increase the brightness of the coating. From the experimental observations, it was found that concentration of glycerol, pH of the bath and the c.d. are the prominent variables which can influence the process and product of electrodeposition at 303 K.
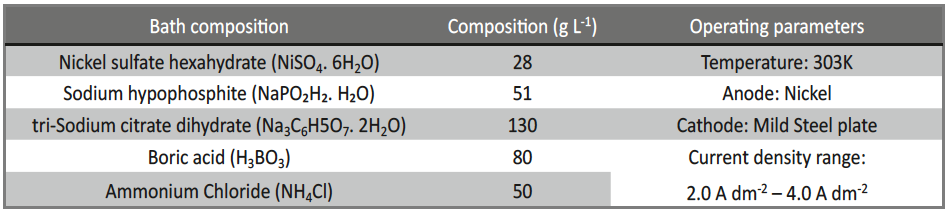
Tab. 1: Composition and operating parameters of basic bath for electrodeposition of bright Ni-P alloy
In order to optimize the process parameters of Ni-P electrolytic bath, to produce a thick, hard adherent and corrosion resistant coating, Taguchi’s experimental design method has been utilised. It may be noted that in the above conventional method of optimization of bath using Hull cell, at a given time only one variable is varied, keeping all other variables constant. Under this situation, the possibility to fix the exact value of all the variables to get the optimal yield is rather far from the reality. Moreover, Hull cell results do not provide the exact c.d. (primary and secondary) distribution, it will show only a range of c.d. Further, the optimization of all process parameters using conventional method needs more number of trials and is highly time consuming. All those problems can easily be handled by the use of Taguchi’s approach, and the errors arising from any of this effect is negligible in the present case [6].
In the present study, Taguchi experimental design method was used to optimize the three important variables of deposition process, namely concentration of glycerol (mL L-1), pH of the plating bath and c.d. used for deposition to impart better properties to the coating, in terms of Vickers micro-hardness and thickness of the electrodeposited coating. The hardness of the deposits was measured by Vickers method, using Micro-hardness Tester (CLEMEX, CMT. HD, Canada). The thickness of the deposits were calculated from Faradays law, while verifying it using Digital Thickness Tester (Coatmeasure M&C, ISO–17025).
2.3 Steps involved in the Design process
Taguchi has envisaged a new method of conducting the design of experiments which are based on well-defined guidelines. This method uses a special set of arrays called orthogonal arrays. These standard arrays stipulates the way of conducting the minimal number of experiments which could give the full information of all the factors that affect the performance parameter. The crux of the orthogonal arrays method lies in choosing the level combinations of the input design variables for each experiment.
- Selection of characteristic performance. / Selecting a characteristic performance that is affected by different parameters in the manufacturing process is the initial step in the Taguchi’s method. In current study, the properties of the electrodeposited samples, i.e., micro-hardness and thickness are influenced by the process parameters, and are selected as the characteristic performance.
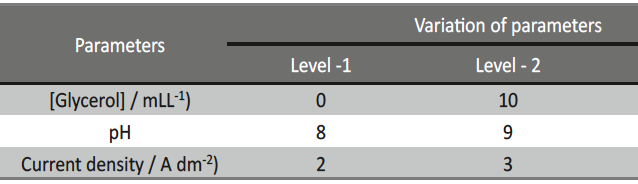
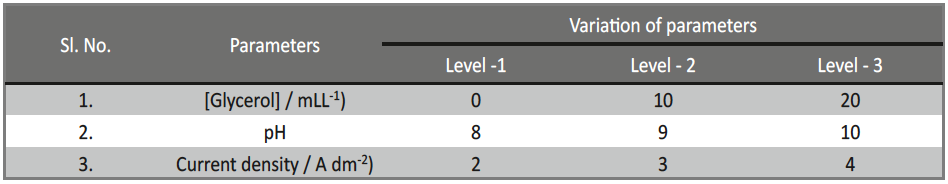
- Selection of deposition parameters and their levels / Based upon the preliminary experimental results, giving information on significantly affecting deposition parameters on the selected characteristic performance of the electrodeposits, their three different levels are to be selected. Then values of the levels were changed to study the effects of individual deposition parameters, as well as their interactions, upon the responses, with the least number of experimental runs. The levels for each deposition parameter were chosen such as to cover the combinations where optimal conditions could potentially exist.
- Selection of orthogonal array Taguchi design of experiments / Taguchi’s orthogonal array L9 design of experiments (27) was used here to identify the critical deposition parameters. L9 orthogonal array allows for conducting experiments at three levels for three deposition parameters. Adoption of the experimental design speeds up the process of optimization, and additionally saves time and resources.
- Conducting of experiments / As abbreviation suggests, in L9 design of experiment, it needs to complete 9 experiments to analyse the response behavior of the chosen parameters.
- Analyses of data and to determine optimal parameter levels. / With the Taguchi’s method, the analysis consisted of analysis of means (ANOM) and analysis of variance (ANOVA) for S/N ratios. The effect of each deposition parameter at a given level upon the quality of coatings can be best estimated using ANOM. The basic goal of ANOVA is to estimate the variance in the coating quality, owing to the deposition parameters in terms of S/N ratios. A main effect of plot for S/N ratios will help to determine the optimal value for each deposition parameter.
- Confirmation by performing the experiment. / The final step in the Taguchi Method involves the validation of experiment and results from optimal deposition parameters to acquire the targeted value of the evaluating property of the coating.
2.4 Design of experiment
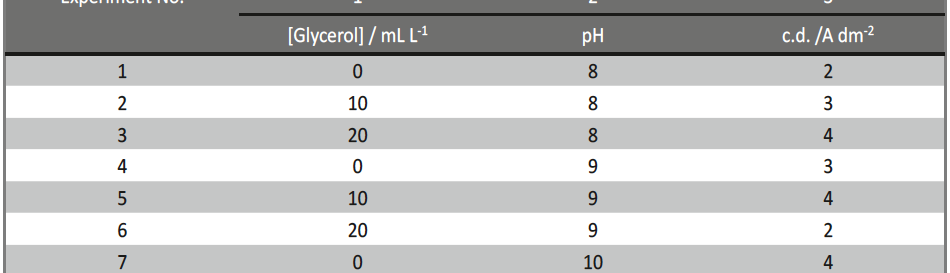
Based on the Taguchi’s method, an orthogonal array (OA) was employed to reduce the number of experiments for determining the optimal coating process parameters, like pH, concentration of additive and c.d. to improve micro-hardness and thickness. The selected parameters for optimization and their levels used in this study were shown in Table 2. L9 OA which has 9 rows corresponding to the number of tests with 3 columns at three levels was chosen. The experimental layout chosen for optimizing the process parameters are given in Table 3.

Tab. 3: Experimental layout (L9) employed for optimizing the process parameters for electrodeposition of Ni-P alloy on mild steel at 303K
Based on the experimental layout, 9 experiments were performed for optimizing the process parameters for developing hard and thick Ni-P alloy coatings, and the obtained results are discussed in the following section.
3. Results and Discussion
3.1 Analysis of S/N ratio
Based on the experimental results, effect of each deposition parameter at a given level upon the quality of coatings estimated using ANOM and ANOVA, in terms of S/N ratio will obtain from the selected (L9) orthogonal array. Taguchi defined the sensitivity (signal-to-noise ratio, S/N) as the logarithmic function for the response analysis of chosen parameters to generate optimal design [8]. The S/N ratio method was adopted for the deduction of optimum operation level by varying the factors and it cannot be accounted from the average responses. Since the aim of experiments was to enhance the hardness and thickness of the coating, S/N has been chosen as larger- the better and S/N ratio was calculated using equation (1).
Where, n represents the number of samples obtained from a given experiment and y represents the output from the experiment.
Accordingly, S/N ratio of a factor pH, at Level 1 was calculated by averaging the S/N ratio of experiments. Vickers micro-hardness and thickness of the coatings were determined experimentally and their corresponding S/N ratios obtained by analysing Taguchi design are reported in Table 4.
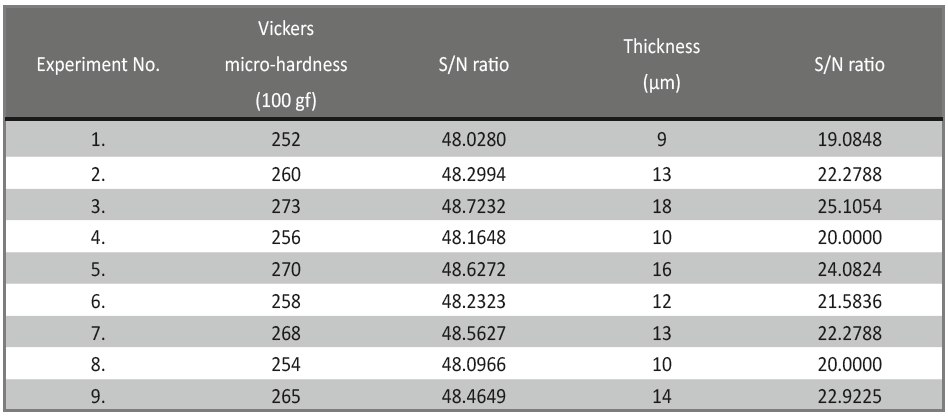
Tab. 4: The S/N ratios of different experiments, in terms of variation of micro-hardness and thickness of the deposit
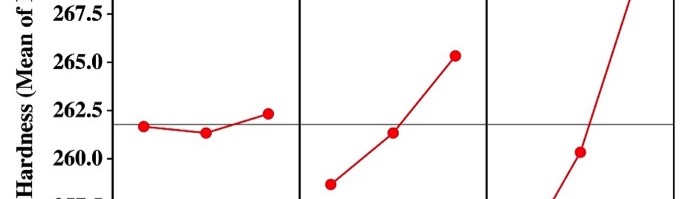
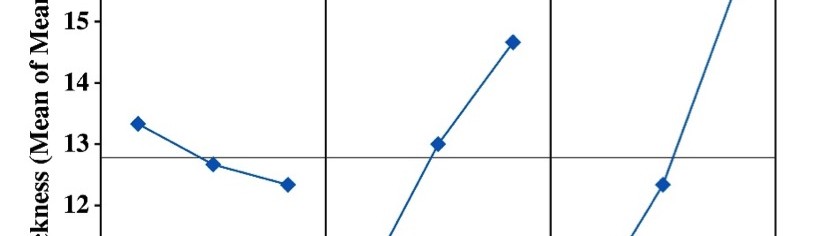
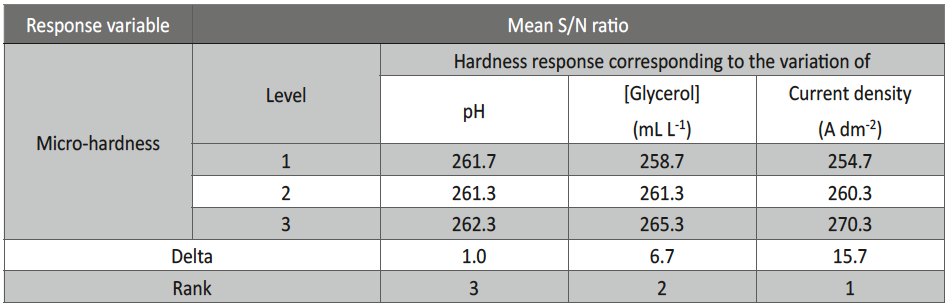
From Table 4, it may be observed that the Vickers micro-hardness ranges from 252-273, and the thickness ranges from about 9-18 μm. The response table of mean S/N ratio for hardness and thickness of Ni-P alloy coating were given in Table 5 and 6, and corresponding Figures are shown in Figure 2 and Figure 3. The delta values shown in the Tables indicate the difference between the highest and the lowest S/N ratio. Then, ranks were assigned on the basis of the delta value; rank 1 is assigned to highest delta value, rank 2 is assigned to next highest value and so on. Micro-hardness of the coatings was found to be more influenced by the applied c.d. and concentration of the additive as per the response values of mean S/N ratio, shown in Table 5. The contribution of pH of the plating bath on the micro-hardness of Ni-P alloy coatings can be neglected due to its significantly low delta value (Table 5), compared to other two parameters.

Fig. 2: Graphical representation showing the variation of micro-hardness at different conditions of bath variables

Fig. 3: Graphical representation showing the variation of thickness of the coating at different conditions of bath variables
From the data in Table 6, it may be noted that the mean S/N ratios of Ni-P alloy coating for thickness has close dependency with c.d., next to [glycerol], and then to pH. In the case of thickness also pH was found to have the least influence as compared with the c.d. and additive concentration.
Based on the statistical data derived from the nine set of experiments (Table 2), it was found that, under condition of mean S/N ratio, the basic Ni-P bath is capable of producing the coatings of highest micro-hardness at [glycerol] = 20 mL L-1, pH = 10 and c.d. = 4.0 A dm-2; and coating of highest thickness at [glycerol] = 20 mL L-1 pH = 8 and c.d. = 4.0 A dm-2.
3.2 Confirmatory experiments
The statistically found optimal data, obtained by Taguchi’s model, were put to test by conducting confirmatory tests under the optimal conditions, keeping the basic bath composition as the same. Accordingly, it was found an increase in the micro-hardness and thickness of Ni-P alloy coatings developed under optimal conditions and the obtained results are given in the Table 7. The variation in micro-hardness of the coating was found to be dependent on the P content in the coating. The coating under optimal conditions for micro-hardness having the maximum P content (9.03 wt.%). It was found to be least influenced by the variation in pH and most subjective to c.d. changes. Hence the optimization resulted in maximising the micro-hardness and thereby the P content in the coatings too. The results from the confirmation tests, using the best set of predictions as obtained from the Taguchi analysis reveals that, this method is suitable for optimizing the processing conditions of electrodeposition.

Tab. 7: Results of confirmatory experiments performed for micro-hardness and thickness under optimal conditions
3.3 Optimal bath and operating parameters for electrodeposition Ni-P alloy coatings
From the optimization results it is clear that the variation in pH of the plating bath plays very less influence in the properties of the deposited alloy coatings as compared with the other parameters. Table 8 shows the optimal bath and process parameters for electrodeposition of bright, hard and thick Ni-P alloy coating.

Tab. 8: Composition and operating parameters of optimal bath for electrodeposition of bright Ni-P alloy coatings
4. Conclusions
Based on the experimental results of investigation following conclusions are drawn:
- Taguchi’s experimental design method was used successfully to optimize a new Ni-P bath, for developing the coatings of maximum micro-hardness and thickness.
- The maximum micro-hardness (275 HV) and thickness (20 μm) of Ni-P alloy coatings, obtained by confirmatory tests have validated the proposed bath (Table 4), in conformation with Taguchi’s approach.
- The influence of pH is found to be negligible, compared to that of [glycerol] and c.d., on both micro-hardness and thickness of the electrodeposited Ni-P alloy.
- Taguchi’s method is found to be effective for achieving the coatings of desired property, by reducing the number of trails to a practical level by proper selection of chosen variables and characteristic performance.
ACKNOWLEDGMENTS
Mr. Liju Elias acknowledges National Institute of Technology Karnataka (NITK), Surathkal for financial support in the form of Institute Fellowship.
REFERENCES
- Paunovic, M., Schlesinger, M. (2006): Fundamentals of electrochemical deposition. 2nd edition: John Wiley & sons, ISBN 978-0-471-71221-3.
- Dini, J.W. (1993): Electrodeposition: the materials science of coatings and substrates. Noyes Publications, 120 Mill Rd, Park Ridge, New Jersey 07656, USA, ISBN 0-8155-1320-8.
- Kanani, Nasser (2004): Electroplating. Basic principles, processes and practice. 1st edition, Oxford: Elsevier Advanced Technology, ISBN 1856174514.
- Dabholkar, A., Sundaram, M.M. (2012): Study of micro-abrasive tool-making by pulse plating using Taguchi method. Mater. Manuf. Processes. 27, 1233–1238, ISSN 1042-6914.
- Rosa, J.L., Robin, A., Silva, M.B., Baldan, C.A., Peres, M.P. (2009): Electrodeposition of copper on titanium wires: Taguchi experimental design approach. J. Mater. Process. Technol. 209, 1181–1188, ISSN 0924-0136.
- Cox, D.R., Reid, N. (2000): The theory of the design of experiments. Chapman and Hall/CRC Press, ISBN 9781584881957 – CAT# C195X.
- Roy, R.K. (2010): A primer on the Taguchi method. 2nd edition: Society of Manufacturing Engineers, ISBN 13: 978- 0872638648.
- Farzaneh, A., Ehteshamzadeh, M., Mohammadi, M. (2011): Corrosion performance of the electroless Ni–P coatings prepared in different conditions and optimized by the Taguchi method. J. Appl. Electrochem. 41, 19–27, ISSN 1572-8838.
- Mughal, M.A., Newell, M.J., Engelken, R., Vangilder, J., Thapa, S., Wood, K., Carroll, B.R., Johnson, J.B. (2013): Statistical analysis of electroplated indium (III) sulfide (In2S3) films, a potential buffer material for PV,(Heterojunction Solar Cell) systems, using organic electrolytes. NSTI-Nanotech 2013, At Washington, D.C., Volume: 3, 523–527.
- Antony, J. (2014): Design of experiments for engineers and scientists. Elsevier Science Limited, ISBN 0080994172, 9780080994178.
- Parthasaradhy, N.V. (1989): Practical electroplating handbook. University of Michigan: Prentice Hall, ISBN 0133808661, 9780133808667.
PDF Version of the article |
Flash Version of the article |
|
| [qr-code size=”2″] | ||





Thank you for reading our article and giving good comments.