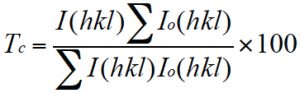The condensation product of Vanillin and Cysteine Hydrochloride (VC) was used as an additive for the electrodeposition of Zn-Ni alloy on mild steel substrate. The bath constituents and operating conditions were optimized by Hull cell experiments. The electrochemical behaviour and nucleation mechanism was studied using cyclicvoltammetry and chronoamperometric techniques. The electrochemical studies revealed that electrocrystallisation process of zincnickel alloy coating was governed by three-dimensional (3D) nucleation process, controlled by diffusion. The model of Schariffker and Hills was used to analyze the current transients and it revealed that, in bright zinc-nickel alloy coating, the electrocrystallization process is regulated by instantaneous nucleation mechanism. The electrochemical impedance spectroscopy and Tafel polarization studies were used to study corrosion nature of Zn-Ni electrodeposits. Corrosion studies showed an improved corrosion resistant nature of bright Zn-Ni alloy coatings on mild steel substrate. The scanning electron microscopy (SEM) and X-ray diffraction (XRD) studies depicted smooth, compact and fine-grained structure of Zn-Ni electrodeposit in presence of VC, in plating bath solution.
1 Introduction
Electrodeposition of metals and alloys has become widely used in many industries, with distinct advantages compared to most of the other alloy coating technologies [1-3]. This technique has proved to be very convenient due to its simplicity and low cost in comparison to other methods and yield porous free finished products [4]. The electrodeposition of Zn-Ni alloy is interesting because this alloy exhibits significant higher corrosion resistance and better surface morphology than pure zinc [5]. The aeronautical industry has shown increasing interest in Zn-Ni alloy coating as a substitute for toxic and high-cost cadmium coatings [5]. The deposited Zn-Ni alloy is always richer in zinc than nickel when electrodeposited from the solution containing Zn2+ and Ni2+ ions. The electrodeposition of Zn-Ni alloys is classified by Brenner [7] as an anomalous codeposition, where zinc a less noble metal is preferentially deposited [6]. Many studies have been carried out to understand the characteristics of the Zn-Ni alloy coatings and the nucleation mechanism of electrodeposition process. It is found that the characteristics of the deposits depend on electrodeposition parameters like applied voltage, current density, pH, bath-composition, additives, temperature etc. [8, 9]. Many theories and experimental procedures are available in literature related to nucleation mechanisms explained with the help of Schariffker and Hills models. This model considers the random nature of nucleation and the coupled growth of hemispherical nuclei under diffusion conditions.
In the present study the condensation product of Vanillin and Cysteine Hydrochloride (VC) has been used as an organic additive for electroplating bath solution to get bright Zn-Ni alloy electrodeposits.
2 Experimental
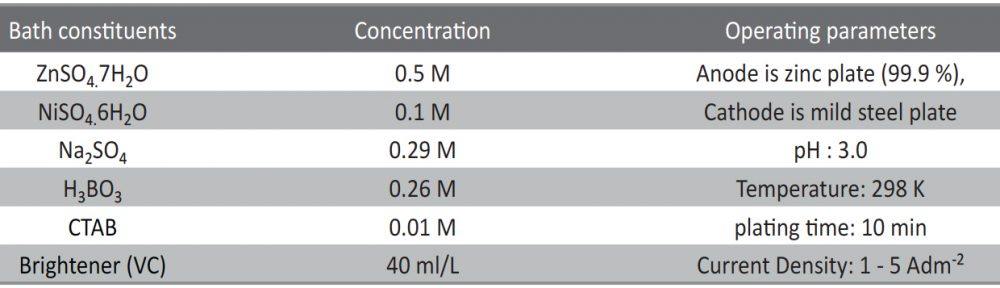
All chemicals used were of AR grade. The bath solutions were freshly prepared using distilled water. The standard Hull cell of 267 ml capacity was used to optimize the bath constituents and bath variables. The Hull cell experiments were carried out without agitation. The pH of the bath solution was adjusted with 10 % by vol. H2SO4 (Sulphuric acid) solution or NaHCO3 (sodium bicarbonate) solution. Zinc plate of 99.99 % purity was used as anode and activated each time by immersing in 10 % by vol. HCl for 4-5 sec followed by washing with water. The mild steel plates of standard Hull cell size were mechanically polished to get smooth surface. The scales on the steel plates were removed by dipping in 10 % by vol. HCl solution. These steel plates were washed with distilled water, dried and used for the experiments. After electrodeposition, the coated plate was removed from the solution and subjected to bright dip in 1 % by vol. HNO3 solution for 3 – 5 seconds followed by water wash and dried.
The condensation product was synthesised from equimolar amounts of Vanillin (1mM) with chemical formula C8H8O3 (AR grade, S. D. fine chemicals) and Cysteine hydrochloride monohydrate (1mM) with chemical formula C3H10ClNO3S (AR grade, S. D. fine chemicals) in acetic acid medium, under reflux condition for 2-3 hrs on hot plate at 343 K [10, 11]. The resulting reaction solution was diluted to 100 ml with distilled water and a known amount of this solution was added into the plating bath solution. The plating bath solution with additives was stirred for two hours before electrodeposition, to ensure proper mixing of two solutions. The completion of the reaction was confirmed by thin layer chromatography technique (TLC).
The cyclicvoltammetric and chronoamperometric studies were performed using CHI660D electrochemical workstation with a three-electrode system. A graphite electrode of geometrical area 0.02 cm2, a platinum wire and saturated calomel electrode were used as working, counter and reference electrodes respectively. Before conducting each experiment, working electrode was polished to a mirror finish with 0.05 μm alumina.
The corrosion behaviour of Zn-Ni electrodeposits were studied in 3.5 wt. % NaCl solution by galvanostatic polarization method and electrochemical impedance spectroscopic technique. The Zn-Ni electrodeposit was used as a working electrode (exposed area 1 cm2). A platinum foil was used as a counter electrode with saturated calomel as a reference electrode, in the potential range of – 0.9 V to – 1.4 V and at a scan rate of 0.01 Vs-1.
The surface morphology of Zn-Ni electrodeposits was examined using scanning electron microscope (JEOL-JSM-6400). The Philips X’pert PRO MPD X-ray Diffractometer was used to determine the orientation of Zn-Ni crystallites in the deposit.
The preferred orientation of the deposits was determined using Muresan’s method by calculating the texture coefficient (Tc) using the following equation:
Where I(hkl) is the peak intensity of Zn-Ni alloy electrodeposits and ΣI is the sum of the intensities of the independent peaks. The index ‘0’ refers to the intensities for the standard Zn-Ni sample (JCPDS 10-0209). The orientation with maximum texture coefficient value is the preferred orientation of the deposit [12].
The percentage reflection of the deposits was determined using ocean optics USB 4000 Spectrophotometer, referenced against a silver mirror. The reflectivity of silver mirror was set at 100 % and the measurements were carried out at different surface points of the deposited sample.
3 Results and Discussion
3.1 Electrodeposition process
Hull cell is a miniature plating tank having trapezoidal shape, 267 ml capacity and used for the optimisation of the parameters. Similar to our earlier studies [11, 13-14] the effects of bath constituents, pH, current density and temperature were optimized by Hull cell experiments. The optimized bath composition and optimized conditions are given in Table 1. CTAB was used as a wetting agent in the bath solution. The basic bath gave coarse dull deposit between the current density range of 0.5 – 3 A/dm2. In order to improve the nature of the deposit, a known volume of VC was added to the bath solution. The optimized bath solution gave bright deposits between the current density range of 1 – 5 A/dm2.
3.2 Cyclic voltammetric studies
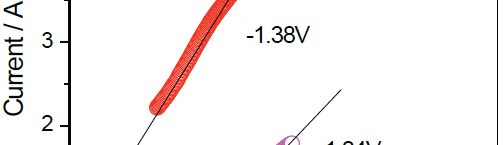
Figure 1 shows effect of VC on electrochemical behaviour of Zn-Ni electrodeposits, analysed by cyclic voltammetric studies. In absence of VC during cathodic scan, one cathodic peak is observed and the peak potential was at – 1.172 V (E1). This peak was associated with simultaneous reduction of Zn2+ to Zn and Ni2+ to Ni. On reversing the sweep direction, two current crossovers appear in the cathodic region. The potential at which the crossover occurs at more cathodic region is known as nucleation overpotential (Eη) [13]. And the second crossover at the zero-current region (- 1.059 V) was known as crossover potential (Eco). The two crossovers were characteristic of reduction overpotential.

Fig. 1: Typical cyclic voltammogram of Zn-Ni electrodeposits obtained on graphite electrode from optimized bath solution (0.5M ZnSO4 + 0.1M NiSO4 + 0.29M Na2SO4 + 0.26M H3BO3 + 0.01M CTAB, pH = 3), in presence (solid line) and absence (dotted line) of VC, in the potential range of – 0.1 V to – 1.6 V at scan rate of 0.05 Vs-1
In presence of VC, the cathodic peak potential (E2) shifted to more negative potential (– 1.386 V). This shows that VC may act at the interface by adsorbing on the cathodic surface, creating barrier near the electrode surface and hence inhibit the reduction of metal ions leading to a decrease in the grain size of the deposit [13]. Hence the cathodic potential associated with the reduction of zinc and nickel ions in presence of VC shifts to more cathodic potential when compared with its value in absence of VC.
In absence of VC, in optimized bath solution, four oxidation peaks (A1, A2, A3 and A4) are seen in the voltammogram. The multiple peaks detected, during electrochemical oxidation of alloys are due to dissolution of the metals in alloy via different intermediate phases [13]. The four anodic dissolution peaks for Zn-Ni alloy correspond to the dissolution of three phases in Zn-Ni deposits. They are η-phase, δ-phase (Ni3Zn22) and γ-phase (Ni5Zn21) [11]. The first anodic peak (A1) at a potential of – 0.885 V correspond to the oxidation of zinc from the η-phase (zinc rich phase). The second anodic peak (A2) at potential – 0.759 V and the third anodic peak (A3) at potential of – 0.547 V correspond to the dissolution of zinc from δ- and γ-phases respectively. The fourth anodic peak (A4) at a more noble potential (– 0.220 V), corresponds to the dissolution of nickel from δ- and γ- phases [11].
In presence of VC in optimized bath solution, two oxidation peaks (a1 and a2) were observed at – 0.880 V and – 0.214 V respectively. The first broad anodic peak (a1) corresponds to oxidation of zinc from the η- phase and second broad peak (a2) corresponds to the oxidation of nickel from η- and γ- phases. The oxidation peaks are slightly shifted to more positive potential. This indicates that in presence of VC, the oxidation of Zn-Ni alloy becomes more difficult and hence the coating appears fine grained and smooth. Similar results were observed in the literature [13].
3.3 Transient analysis
Chronoamperometric technique is a potential technique for explaining electro-crystallization process [15].

Fig. 2: Chronoamperometric curves for zinc-nickel alloy deposition process for zinc-nickel plating bath solution in (A) absence of VC and (B) presence of VC (working electrode: graphite electrode with area 0.02 cm2; counter electrode: Platinum wire and reference electrode: SCE; Zinc-Nickel bath: 0.5M ZnSO4 + 0.1M NiSO4 + 0.29M Na2SO4 + 0.26M H3BO3 + 0.01M CTAB and pH = 3)
Figure 2 shows the current time (I – t) transients recorded during the reduction of alloy, in the potential range from – 1.10 V to – 1.18 V in absence of VC and in the potential range from – 1.32 V to – 1.44 V in presence of VC in the plating bath solution. The behaviour of these current transients is typical of a nucleation process with three-dimensional (3D) growth of nuclei controlled by the diffusion of the electroactive species. All the curves showed initial sharp rise in short duration until the current reaches a maximum value (Imax). This sharp rise in current is probably due to the growth and stabilisation of initial nuclei. The decay in current density after reaching Imax of each transient converges almost to a limiting current, which correspond to a linear diffusion of the electroactive species to a planar electrode [16]. The change in limiting current at lower potentials may be due to adsorption of brightener on the electrode surface (Figure 2B). In order to characterize nucleation process, the current transients were normalized to (I/Imax)2 versus t/tmax and then compared to the well-known theoretical (I/Imax)2 versus t/tmax curves derived for instantaneous and progressive three-dimensional (3D) nucleation or growth models [15], whose equations are given below.
For instantaneous nucleation,
For Progressive nucleation,
Where I is current density at any instant of time, Imax is maximum current density with respect to maximum time (tmax).

Fig. 3: Non-dimensional (I / Imax)2 versus t / tmax plot for electrodeposition of zinc-nickel alloy coating obtained from zinc-nickel bath: 0.5M ZnSO4 + 0.1M NiSO4 + 0.29M Na2SO4 + 0.26M H3BO3 + 0.01M CTAB and at pH = 3 (A) in absence and (B) in presence of 40 mlL-1 VC
Non-dimensional plots obtained with the experimental and theoretical data for Zn-Ni electrodeposition in absence and presence of VC are shown in Figure 3 at different potentials. The following mechanism explains the anomalous behaviour of metal ions on the electrode surface
where M indicates nickel, iron and cobalt atoms. Although many attempts have been made to explain the anomalous codeposition of alloys, there is still no universally accepted theory [16]. The reduction rate of M mainly depends on the stability of M (OH)+ads or M (OH)+. The stability of the Nickel and Zinc metal monohydroxide ions or metal hydroxides can be sorted in the following order: Ni (OH)+ > Zn (OH)+. As explained above, the amount of nickel in the electrodeposit is lower than the amount of ions in plating bath solution [16]. Also the release of hydrogen gas in addition with the electrodeposition of Zn-Ni alloy on the electrode surface is seen from Equation (4) and Equation (7).
Figure 3A shows the rising part of transients located between the instantaneous nucleation and progressive nucleation curves at lower potentials. As soon as the potential turns to more negative values, curves are shifted in close proximity to instantaneous nucleation curve.
Figure 3B shows the rising part of transients, located on instantaneous nucleation curve under the studied potentials. These results confirm that the nucleation process occurs by instantaneous nucleation mechanism in presence of VC, in plating bath solution.
Another way to further verify the nucleation type of Zn-Ni electrodeposition, given by Schariffker and Hills nucleation model is based on the rising part of current -time transient curves, which is nothing but the analysis of early stage of deposition [15]. The rising current should have a good linear relationship with t3/2 for progressive nucleation and t1/2 for instantaneous nucleation respectively.

Fig. 4: Dependence between I versus t1/2 plot for initial transient portion of zinc-nickel electrodeposit in presence of VC from Fig. 2B
Figure 4 shows the plot of I versus t1/2 fitted linearly, in presence of VC, which indicates that the nucleation occurs by instantaneous nucleation mechanism under the studied experimental conditions.
3.4 Impedance and Polarisation studies
The corrosion resistance studies were carried out using Electrochemical Impedance Spectroscopy and Tafel extrapolation method.

Tab. 2: Electrochemical parameters for the fitted impedance diagram and for polarization curves, measured in 3.5 wt. % NaCl solution, obtained from dull and bright zinc-nickel alloy deposit, on mild steel electrode of 1cm2 area and pH = 3
Figure 5A shows the comparison of Nyquist plots of Zn-Ni electrodeposit in absence and presence of VC in optimized bath. The charge transfer resistance value of the deposit obtained in absence and presence of VC was found to be 16 Ωcm2 and 486.6 Ωcm2 respectively with the help of ZSimp-Win 3.21 software.
Figure 5B shows Tafel curves for dull and bright Zn-Ni alloy coatings on mild steel substrate. The data obtained from Tafel plots revealed that, there is decrease in Icorr. value, from 0.14 A/cm2 (dull deposit) to 0.06 A/cm2 (bright deposit).

Fig. 5: (A) Nyquist Impedance plot at amplitude = 0.005 V and in the frequency range of 0.1Hz to 1e+4 Hz (B) Tafel polarisation
curves of Zn-Ni coating in 3.5 wt. % NaCl solution. Zinc-nickel
alloy electrodeposit obtained in a absence and b presence of
VC, in plating bath solution [Inset in Fig. 5(A) shows electrical
equivalent circuit used to simulate the recorded EIS data]
These results revealed that increase in corrosion resistance is strongly influenced by the presence of additive by changing the alloy composition of the deposits.
3.5 SEM and Reflectance studies
Figure 6 shows the SEM image of dull and bright deposit obtained in absence and presence of VC, in Zn-Ni plating bath solution. Dull deposits obtained in absence of VC showed coarse grained structure, having randomly distributed Zn-Ni alloy crystallites whereas bright deposit showed fine grained, uniform, smooth and compact deposit. This indicates that VC promotes grain refinement by increasing the number of nucleation sites and retards the random growth of nuclei during electrodeposition process. The percentage of each element present in electrodeposit was analysed by EDX. The EDX analysis shows the percentage of Ni in absence and presence of VC, in plating bath solution as 7.8 % and 9.45 % respectively.

Fig. 6: SEM images of zinc-nickel electrodeposits at current density 4 Adm-2 in (A) absence and (B) presence of VC, in the optimized zinc-nickel bath solution
The ideal reflectance and the degree of total reflection is a function of wavelength of visible light. Figure 7 shows reflectance curves for zincnickel alloy electrodeposits obtained in absence and presence of VC in the plating bath solution. From the Figure 7, it is clearly visible that the presence of VC in plating bath increases the percentage reflection in the visible region. Only 4-7 % of variation in the total reflectance was observed at different points of electrodeposits. This result confirmed that VC may act as a good brightener for Zn-Ni electrodeposits.

Fig. 7: Reflectance spectra of zinc-nickel electrodeposit at 4 Adm-2 in (a) absence and (b) presence of VC, obtained from the optimized zinc-nickel bath solution
3.6 X-Ray Diffraction studies
XRD pattern of dull and bright Zn-Ni electrodeposits is shown in Figure 8.

Fig. 8: XRD pattern of Zn-Ni alloy electrodeposit observed in (a) absence and (b) presence of VC in optimized Zn-Ni bath solution
It shows formation of lines corresponding to η- phase of zinc and additional lines that can be indexed considering γ-Ni5Zn21 phase of zinc-nickel alloy. All peaks are corresponding to only zinc and zinc-nickel alloy, which means that there are no impurities in the deposit. Figure 8(b) shows line broadening in presence of VC, which indicates that the organic additive has a remarkable influence in refining the grain size of deposit.
To know the influence of VC on preferred orientation of the Zn-Ni alloy crystallites, texture coefficient (Tc) was calculated for each peak of diffraction pattern. In absence of VC, texture coefficient value was maximum for (330) planes. Hence (330) was the preferred orientation of Zn-Ni alloy crystallites in dull deposit. Presence of VC in bath leads to the formation of more predominant (100) and (330) planes (confirmed by PDF#06- 0653 JCPDS Database). The morphological and micro structural changes such as grain refinement, smoothening (Fig. 6B) and also the formation of more predominant preferred orientation along (100) and (330) planes of zinc-nickel alloy crystallites, contributed towards bright appearance of the deposit. These are the essential conditions required for bright appearance of Zn-Ni electrodeposit.
4 Conclusions
- Newly synthesised brightener gave bright Zn-Ni electrodeposits, in optimized bath solution.
- Voltammetric response gave information regarding characteristics of the components of electrodeposits and also phases of Zn-Ni alloy. It indicated that, in presence of VC the oxidation of Zn-Ni alloy becomes more difficult. It also reveals that VC partially adsorbs on the steel surface, reduces dissolution or reduction process of Zn and Ni metal ions and hence results in the formation of compact Zn-Ni electrodeposit on steel substrate.
- The chronoamperometric studies showed that, the nucleation mechanism involved in formation of bright Zn-Ni electrodeposit is instantaneous nucleation mechanism.
- Corrosion studies confirmed the enhancement of corrosion resistance and hence the protection ability of bright Zn-Ni electrodeposit.
- SEM and Reflectance studies proved the fact that, presence of VC in Zn-Ni alloy bath, promotes the formation of smooth and shiny coatings.
- XRD studies revealed the formation of predominant (100) and (330) planes as preferred orientation of Zn-Ni alloy crystallites, responsible for grain refinement in bright deposit.
The proposed studies suggest that, newly synthesised additive may be used as a brightener for Zn-Ni alloy coating on steel substrate.
Acknowledgment- The authors are grateful to BTLITM and Kuvempu University, Department of Science and Technology for providing the lab facilities to carry out this work.
References:
- Y. P. Lin and J. R. Selman, Electrodeposition of corrosion resistant Ni-Zn alloy.I.Cyclic voltammetric study, Journal of electrochemical society, (1993) v. 140, No. 5, p.1299 -1303.
- Ramesh S. Bhat, Udaya Bhat K., A. Chittaranjan Hegde, Corrosion behaviour of electrodeposited Zn-Ni, Zn-Co and Zn-Ni-Co alloys, Anal & Bioanalytical Electrochem, (2011) v. 3, no.3, p. 302-315.
- N. Eliaz, K. Venkatakrishna, A. Chittaranjan Hegde, Electroplating and characterisation of Zn-Ni, Zn-Co and Zn-Ni-Co alloys, Surface and Coatings technology, (2010) v. 205, p.1969-1978
- H. B. Muralidhara, J. Balasubramanyam, Y. Arthoba Naik, K. Yogesh Kumar, H. Hanumanthappa, M. S. Veena, Electrodeposition of nanocrystalline Zinc on steel substrate from acid sulphate Bath and its corrosion study, J. Chem. Pharm. Res., (2011) v. 3 , No. 6, p.433-449.
- Basavanna S., Arthoba Naik Y., Electrochemical studies of Zn-Ni alloy coatings from acid chloride bath, J. Appl. Electrochem, (2009) v. 39, p.1975 https://doi.org/10.1007/s10800-009-9907-1
- Basavanna S., Arthoba Naik, Y., Study of the effect of new brightener on Zn-Ni electrodeposition from acid sulphate bath, J. Appl. Electrochem., (2011) v. 41, p.535, DOI: 10.1007/s10800-011-0263-6
- Brenner A., Electrodeposition of alloys, Academic Press, Principles and practise, New York and London, (1963) v. 1, eBook ISBN: 9781483223117
- Hiane, M. And Ebothe, J., The European physical journal, (2001) B 22, p. 485-495
- Rahman, M. J., Sen, S. R., Moniruzzaman, M., Morphology and properties of electrodeposited Zn-Ni alloy coatings on mild steel, Journal of Mechanical Engineering, (2009) v. 40, No.1, p.9, http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.619.2785&rep=rep1&type=pdf
- Basavanna S., Arthoba Naik, Y., Electrochemical and reflectance studies of bright Zn-Ni alloy coatings, Indian Journal of Chemical Technology, (2012) v. 19, p.91 http://nopr.niscair.res.in/handle/123456789/13703
- Nayana K. O., Venkatesha, T. V., Effect of ethyl vanillin on Zn-Ni alloy electrodeposition and its properties, Bull. Material Sci., (2014) v. 37, p.1137, https://www.ias.ac.in/article/fulltext/boms/037/05/1137-1146
- Conceicao A. M. D., Jose W J S., Roberto Z. N., Corrosion resistance of Zn-Ni electrodeposits: Morphological characterisation and phase identification, Material Sciences and applications, (2013) v. 4, p.644 http://www.scirp.org/journal/PaperInformation.aspx?PaperID=38134
- Chen D. & Martell A.E., Dioxygen affinities of synthetic cobalt Schiff base complexes Inorg chem., (1987) v. 26, p.1026 DOI: 10.1021/ic00254a013
- Nayana K.O., Venkatesha, T.V., Synthesis and reactivity in Inorganic, Metal-organic and Nano-metal chemistry, (2010) v. 40, No. 3, p.170
- Schariffker B., Hills G., Theoretical and experimental studies of multiple nucleation, Electrochimica Acta., (1983) v. 28, p.879, https://scholar.google.com/citations?user=7mMLt6UAAAAJ&hl=en
- Allen Bai, Chi-Chang Hu, Composition controlling of Co–Ni and Fe–Co alloys using pulse-reverse electroplating through means of experimental strategies, Electrochimica Acta, (2005) v. 50, p.1335, https://doi.org/10.1016/j.electacta.2004.07.055
PDF Version of the article |
Flash Version of the article |
|
| [qrcode] | ||