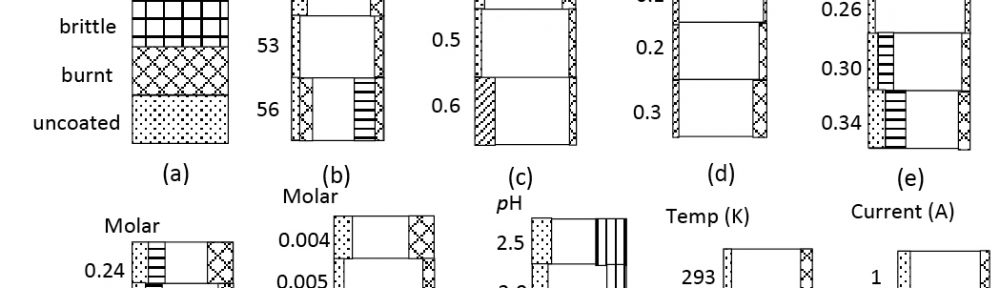
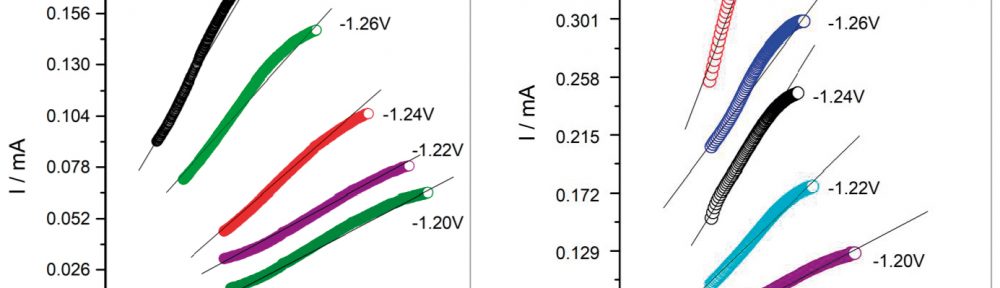
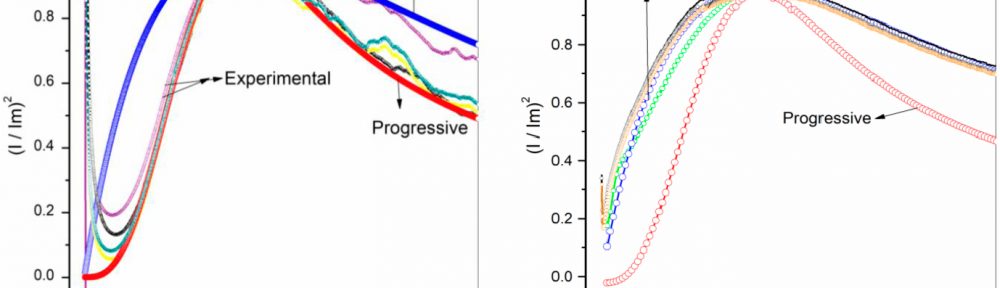
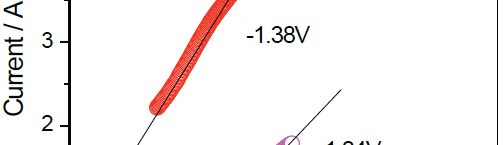
In present investigation, a new brightener was synthesized by condensation of 3, 4, 5-Trimethoxy benzaldehyde and Glycine (TG). Hull cell experiments were conducted to optimize the plating bath components and operating parameters. To examine the influence of TG on nucleation mechanism of Zn-Co alloy electrodeposition, cyclic voltammetry and chronoamperometry study was carried out. Schariffker and Hills model was used to analyze current transients, which in presence of TG confirmed instantaneous nucleation. Corrosion studies were done using potentiodynamic polarization and electrochemical impedance spectroscopic technique, in 3.5 wt. % NaCl for bright and dull zinc-cobalt alloy coatings. Phase structure, surface morphology and brightness of the deposit were characterized by X-ray diffraction analysis, scanning electron microscopy and reflectance studies. These studies revealed the role of TG in modifying the nucleation mechanism and surface morphology of zinc-cobalt alloy crystallites and thereby producing a bright corrosion resistant Zn-Co alloy coating on mild steel substrate.
Author Archives: Ass. Prof. Jyoti Shankarrao Kavirajwar
Electrochemical studies of the bright Zn-Ni alloy electrodeposit from acid sulphate bath
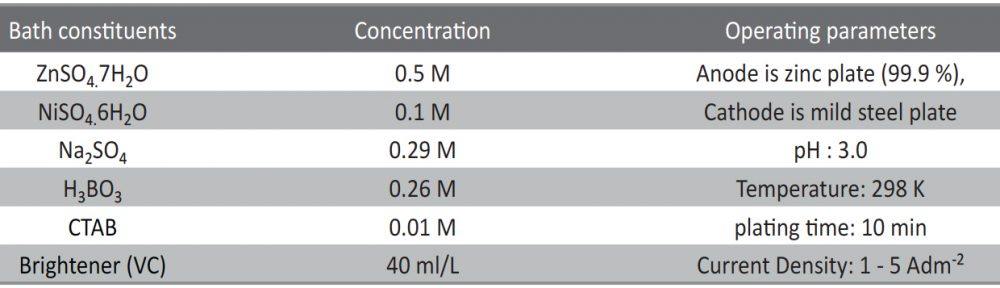
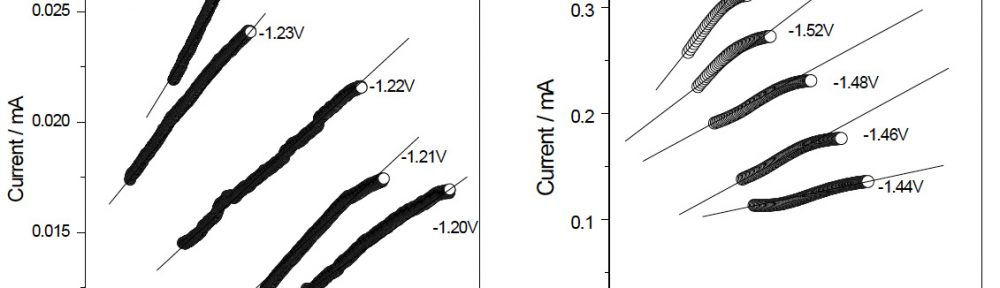
The condensation product of Vanillin and Cysteine Hydrochloride (VC) was used as an additive for the electrodeposition of Zn-Ni alloy on mild steel substrate. The bath constituents and operating conditions were optimized by Hull cell experiments. The electrochemical behaviour and nucleation mechanism was studied using cyclicvoltammetry and chronoamperometric techniques. The electrochemical studies revealed that electrocrystallisation process of zincnickel alloy coating was governed by three-dimensional (3D) nucleation process, controlled by diffusion. The model of Schariffker and Hills was used to analyze the current transients and it revealed that, in bright zinc-nickel alloy coating, the electrocrystallization process is regulated by instantaneous nucleation mechanism. The electrochemical impedance spectroscopy and Tafel polarization studies were used to study corrosion nature of Zn-Ni electrodeposits. Corrosion studies showed an improved corrosion resistant nature of bright Zn-Ni alloy coatings on mild steel substrate. The scanning electron microscopy (SEM) and X-ray diffraction (XRD) studies depicted smooth, compact and fine-grained structure of Zn-Ni electrodeposit in presence of VC, in plating bath solution.
A study into the effect of a new brightener on electrodeposition and corrosion resistance of zinc
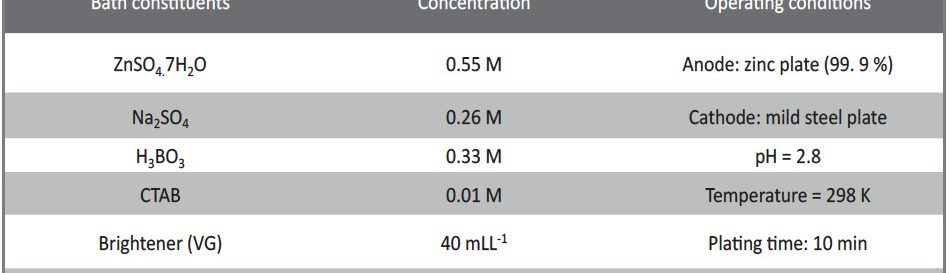
The electrodeposition of zinc on steel was obtained from an acid sulphate bath containing condensation product formed between Vanillin and Glycine (VG). The bath constituents and operating parameters were standardized by Hull cell experiments. The investigation of electrodeposition and nucleation mechanism was carried out on graphite electrode using cyclic voltammetric and chronoamperometric techniques. The corrosion studies were carried out by Polarisation and Electrochemical impedance techniques, which helped to explore the good protection ability of the zinc coating in presence of VG. The surface morphology of the deposit was characterised by scanning electron microscopy. Increase in brightness of the zinc coating obtained on mild steel substrate was confirmed by reflectance studies. The phase structure and the preferred orientation of the zinc crystallites were studied by X-ray diffraction analysis. These studies revealed the influence of VG in enhancing the brightness and corrosion resistance of the zinc electrodeposit on mild steel substrate.