The composite technology allows the development of structures with a high degree of integration, where the number of elements and auxiliary means for their structural joining are minimised. This can only be achieved by the use of appropriate manufacturing and design processes. Some advantages of such an efficient integration would be the low installation and inspection efforts, shorter cycle times in the production, higher robustness and lower manufacturing costs. Despite this high potential, joining these highly integrated parts is indispensable because of restrictions concerning the components’ complexity, repair requirements as well as material specific limitations [1].
Context for hybridisation of composite materials
Especially in the composite technology, structure joining leads to an increase in complexity and causes peaks in the local stress conditions, which in sum have an impairing effect on the lightweight potential of composite materials. On the one hand, these local states of stress demand a precise knowledge of composites’ mechanics and their complex failure behaviour. On the other hand, they are the reason why appropriate reinforcement methods are needed [2]. Current connection techniques in composite technology are based on material connection, form closure or force closure. The adhesive technology as a material closure type has enormous advantages for composites due to the homogenised load distribution, the avoidance of drilling, the aerodynamic attained and the possible tolerance compensation. Yet, it represents a non-detachable connection sensitive to environmental conditions with challenging reproducibility, inspection and quality controls. This leads to a lack of reliability of the joined structure. Therefore, adhesively-bonded composite elements have found a restrained application field – mostly in the military sector [4]. Actually, form closure joining by means of rivets or bolts is the most applied connection technique for composites despite its fundamental mechanical incompatibility. Indeed, in a fibre-reinforced polymer, the matrix receives the load on the surface and transfers it to the reinforcing fibres. Fibres hence mainly contribute to the mechanical strength of composites so cutting them is detrimental to the performance of the final product. However, mechanical joining makes the structure reliable, easy to inspect, separable and, of course, meets the pronounced acceptance from the metal field. So the challenge consists in designing composites in order to answer the requirements of riveting and bolting. The low notch resistance, the poor bearing strength and the limited shear strength of fibre-reinforced polymers have, hence, to be improved to reach good connection efficiency. A conventional approach is to locally thicken the composite laminate with additional layers, which improves all three weaknesses. Yet, thickening results in an eccentric load transfer leading to secondary bending moments; the stress increase is high. Additionally, the increased bolt length impairs bolt bending and local stress concentration. Thus, thickening is not an optimal design although conventional mechanical fastening techniques are to be expected. Therefore, a compromise needs to be found to fully exploit the lightweight potential of composite materials. The use of hybridisation of composite materials with metal is one adequate and feasible design approach. It is meant to both, ally the beneficial properties and compensate the weaknesses of the sole materials, so that the intrinsically heterogeneous material allows an even more specific adaptation of the materials’ properties to the requirements provided than the use of its individual components [5]. Beside the material selection, important factors to consider for an optimised design are the metal volume fraction, the arrangement and binding to one another, the manufacturing process [6] and the consequent internal stress behaviours [7, 8]. Nonetheless, based on the extent of intrinsic hybridisation, it can be categorised as local or extensive reinforcement [9]. The former category generates composite-metal-hybrids locally in highly-loaded regions; the latter Fibre-Metal Laminates, short FMLs, used as structural materials that compensate fundamental weaknesses of the metal or composite part. GLARE, made of Glass Fibre Reinforced Polymer (GFRP) and aluminium, is the most known group of FMLs. It was designed at the Delft University of Technology, in the Netherlands, is patented and standardised. Thereby, the GFRP plies primarily bridge fatigue cracks occurring in the aluminium layer under cyclic loading and thus prevent the crack growth [10]. Secondarily, the glass fibre-epoxy layers play a barrier role and limit the extent of through-the-thickness corrosion of the aluminium alloy layers [11]. In the aircraft industry, aluminium structures mainly subjected to tension-tension cyclic loading are preferably replaced by panels of GLARE such as the upper fuselage shell of the Airbus A380-800 as shown in Figure 1.
Another extensive hybridisation is the reinforcement of carbon fibre reinforced plastic (CFRP) with aluminium layers and is named CARALL. Yet, it presents an increased risk of corrosion due to the electrically conductive carbon fibres and their potential difference with aluminium. In addition, the materials show a great difference in coefficients of thermal expansion. This combined with the hybrid’s manufacturing process induces internal stresses and hence represents a major problem so that CARALL could not be enforced so far [12]. A further category of fibre-metal laminates is based on CFRP and titanium and can be designated as TiGr (standing for Titanium Graphite) or HTCL (for Hybrid Titanium-Composite Laminate). It is mostly produced to fulfil high temperature requirements for supersonic flights and answer both, high weight specific stiffness and strength demands. In most cases, it has been investigated within the framework of cooperation between Boeing and NASA [13]. Similarly to the laminate shown in Figure 2, thin layer hybrids or UD-CFRP-steel-laminates are another application for extensively intrinsic hybrids.

Fig. 2: Drilled CFRP-steel-laminate manufactured at the Composite Design Department at DLR, Braunschweig
The adopted approach consists in using thin steel foils with a ply thickness under 0.08 mm and a metal volume fraction below 10 %. These metal layers complement the arranged composite layers in the load direction whilst keeping a similar stiffness and strength, reduce the notch and impact sensitivities and thus, increase the residual strength of a damaged laminate [9, 14, 15]. Of course, the extensive hybridisation of composite with metal plies permits the use of mechanically fastened joints but even greater connection efficiency can be attained by the local reinforcement of CFRP with metal, thus limiting the gain in mass introduced by the utilisation of metal. Two cases can be differentiated:
- a fibre-reinforced polymer is locally reinforced with metal,
- a composite material is applied locally to reinforce a metallic structure.
The local reinforcement of a metallic structure by composites can be found in bridges, where CFRP laminates are applied externally to the steel structure in order to strengthen deteriorated steel structures and thus, extend the overall service life [16]. Nonetheless, this promising method does not correspond to an intrinsic laminate. Kolesnikov [2] then Fink [1, 4] employ the local hybridisation technique in which composite plies are substituted by thin metal foils in the joint area, therefore bypassing the need for local thickening. In his work, Fink has set himself the task to combine CFRP with an adequate metal. At best, the metallic part should possess high fatigue strength, high stiffness, sufficient elongation at break, low density, low coefficient of thermal expansion, corrosion resistance, good adhesion properties, good machinability, electrochemical compatibility with carbon (thus excluding aluminium alloys, magnesium and non-corrosion resistant steel alloys), high yield strength (aluminium alloys, neat titanium, magnesium and low-strength steel alloys are hence inadequate), on top of being available in sheet shape. In view of these requirements to be fulfilled, only titanium alloys (here Ti-6-4 and Ti-15-3-3-3) and stainless steel alloys (here AISI 301) proved to be adequate. Throughout Finks work, titanium alloys prove themselves to be an optimal reinforcing material in light of their high specific strength and stiffness, their electrochemical compatibility to carbon fibres and their relatively low coefficient of thermal expansion. Yet, experiments show that neither the thickening of the metallic layer, nor the increase in metal volume can further increase the titanium-CFRP-laminate bearing strength. This is due to the inevitable degradation of the carbon composite ply. So the reinforcing effect of titanium alloys is limited. On the other hand, steel alloys are especially suitable as reinforcement due to their high stiffnesses, whilst having comparable outstanding strengths and acceptable elongation at break. Besides, the higher the metal strength, the greater the reinforcing effect. Therefore, higher bearing yield stress and bearing strengths can be achieved with stainless steel alloys in comparison with titanium alloys with the same robustness and comparable laminates’ densities. Finally, by reinforcing a neat composite with titanium or steel, its bearing strength is enhanced by a factor of 2 and 2.6 respectively. For the same resulting bearing strength, the composite-steel-laminate requires about half the metal volume needed for a composite-titanium-laminate, hence leading to manufacturing and cost benefits when using stainless steel (SST). Thus, the application field giving the most attention to hybridisation is the aircraft industry where the structures lightweight is a crucial design factor. Local hybridisation offers the best lightweight potential and shows its efficiency for conventional mechanical joining with bolts under standard conditions. Yet, manufacturing hybrid laminates with stainless steel foils is a first challenge to cope with.
Challenges in manufacturing hybrid laminates
Hybrid laminates consist of a variety of interfaces. Due to the different coefficients of thermal expansion, stresses are transferred by adhesion, resulting in two major goals: increase of adhesion properties and decrease of residual stresses induced during manufacturing. When using steel foils to build an FML, it may be used locally or in the whole part. In both cases, two major requirements to the surface treatment process can be deduced. Firstly, the aim is to reach a possibly thin surface treatment in order to avoid any additional weight or thickness. Secondly, the metal foils need to be as thin as a prepreg or a fabric single layer or even thinner. This increases the complexity of the metal foil handling during surface treatment. The surface treatment process can be subdivided into surface pre- and post-treatment. Following the standard Boeing sol-gel process, the material is degreased and then deoxidized by using a wet or dry grit-blasting method. Finally, an aqueous sol-gel system, a dilute solution of a stabilized alkoxyzirconium organometallic salt and an organosilane coupling agent, is applied. Typically, an adhesive coating is then applied to the treated surface to generate a durable bond. Thus, this method contains grit-blasting, which cannot be ap-plied on thin metal foils (thickness < 0.08 mm) due to the serious risk of damaging the substrate or inhomogeneous work-hardening. Therefore, an alternative pre-treatment needs to be investigated. Two promising alternatives, pickling and a more gentle form of grit-blasting which has been developed especially for the use on these thin foils, were therefore investigated as treatment for SST in an epoxy based CFRP laminate. To avoid the damaging of the metallic foils, the impact energy of the grit needs to be reduced. Due to health, sealing and handling issues, the particle size cannot be reduced below a certain value. Therefore, impact energy was reduced by adjusting impact velocity and angle. As a consequence, a process has been developed where the grits are conducted parallel to the metal foil surface. A turbulent motion of the grits achieves a mild abrasion and removal of the substrate. With the help of an automated facility, the material´s feed rate and the grit flow can be controlled to guarantee consistent process conditions. In the following, this novel approach is referred to as ‘vacuum blasting’ as the acceleration of the grits is achieved by a pressure difference. Both treatments show outstanding adhesion performance when tested at room temperature (RT), at 120 °C, –55 °C and on totally saturated specimens which have been stored in water for 1000 hours [17]. The above mentioned interply stresses may significantly lower the mechanical properties of the hybrid laminate, especially when the difference in thermal expansion is high or the fraction of a single constituent is low. Derived investigations show that the stress formation cannot be described as a thermo-mechanical relationship only because the mechanical interaction between the constituents acts in resin’s rubbery state as well. Ongoing work permits to measure, assess and modify the resultant stresses and achieve a fundamental understanding about the present mechanisms and their crucial parameters [18]. All in all, the manufacturing process being resolved and the internal stresses being investigated, the next step is to consider the intrinsic hybridisation under environmental conditions in order to predict its practical service life.
Need for a corrosion study
The environmental effects on metals are well-known as they have been encountered by industries as well as by the public. Called as the inversed metallurgy, the natural process of corrosion yet practically leads to the decrease of mechanical properties, impairment in surface quality (roughness, hardness…), leaks in tubing and containers, change in appearance of the material finally leading to economic losses, safety risks and loss of products. It is extremely crucial to study corrosion for four main reasons:
- Quest of scientific knowledge. Of course, corrosion is inherently a difficult phenomenon to understand, and its study is in itself a challenging and interesting pursuit.
- Cultural heritage conservation. Practically, understanding corrosion phenomena allows us to work for the conservation of historical monuments like the Statue of Liberty in New York.
- Human lives. Safety is a requirement as illustrated in Figure 3, where the cabin of a commercial airline flying from Hilo to Honolulu in Hawaii suddenly disintegrated on the 28th of April 1988. The cause was the combined effect of metal fatigue and corrosion.
- Economical costs. The United States Cost of Corrosion Study initiated by NACE International [19] estimates that the total annual direct cost of metallic corrosion reaches $276 billion for the U.S. Direct costs include cost of replacement, control costs (maintenance, repair, painting) and design costs (extra cost of utilising corrosion-resistant alloys, protective coatings and corrosion inhibitors). This means that the total direct cost of corrosion in the U.S. represent 3.1 % of their gross domestic product (GDP) yearly. Besides, there are indirect costs with amongst others the shutdown of an airplane, the loss of product due to leakage and overdesign.
Specific to aviation, the ageing of planes is a key problem: frequent inspections and maintenance make up most of the costs of older aircrafts. Using better corrosion-resistant metals, improved design and manufacturing, the costs lie in the incorporation of a corrosion monitoring system [19]. Indeed, an early detection and good prediction of the corrosion damages would permit to avoid safety breaches and massive costs. Although corrosion is an inevitable natural process, it should be avoided and prevented in order to maintain the materials’ mechanical properties. The number of studies available on the corrosion behaviour of intrinsic hybrid materials is small and almost non-existing for the specific combination of CFRP and stainless steel. Yet, it can be seen that most literature (especially focusing on hybrids with CFRP as composite) states a problem of galvanic corrosion between the carbon fibres and the aluminium sheet in moisture environment and conclude that more research has to be done [5]. Mandel and Krüger [21] investigate the localised corrosion behaviour of a coated steel rivet joint combining an epoxy-based-CFRP with an aluminium alloy with potentiodynamic polarisation, an electrochemical method. Thus, there is no previous work on this field apart from fragments. Consequently, the current solution well resumed by Tsai et al. [22] is to introduce glass fibre layers between the carbon fibres core and the overlaid metal – in discord with the lightweight and performance efficiencies. Since it is “a challenge to define the appropriate corrosion testing method” [21], the following intends to offer a specific approach to investigate the corrosion behaviour of a novel hybrid material. The hybrid material under focus is composed of CFRP and a corrosion-resistant stainless steel (SST).
A specific approach for a specific material combination
The prime objective of this work is to study the corrosion behaviour of a stainless steel-CFRP-hybrid. For this purpose, an abstraction of the system in its environment is needed and it can be divided into four interfaces. Two are external: composite/environment or metal/environment, both widely investigated. Two are internal: fibre/matrix (also greatly investigated) or composite/metal. The latter interface proves to be less studied so the focus is put on the mechanisms occurring at the CFRP-SST-interface (not being in direct contact with the environment). In other words, the aim is to comprehend the interactivity between both materials when moisture is present. Understanding the mechanisms at stake, it will be possible to evaluate the probable service life of an intrinsic CFRP-SST-hybrid in specific aircraft conditions. Thus, the data obtained for the first objective will be used to make a bridge between the hybrid’s electrochemistry and its mechanics. Finally, if the calculated lifetime is not satisfying, knowing precisely the corrosion mechanism(s) and its parameters will permit to specifically address one by one of these inducing or amplifying factors and determine the most economical means for refraining or slowing down corrosion at the interface CFRP-SST. To determine the adequate corrosion testing, one can identify the time and budget constraints. The typical time available for testing (hours to some months) is much less than the desired life (30 to 40 years for an aircraft). Therefore, the existing corrosion tests are divided into three groups:
- Field tests or simulated-service testing: it is the most reliable method to predict the corrosion behaviour of a system. Yet, the field tests mean here the application of coupons on an aircraft during 30 to 40 years. Thus, this testing is not further considered.
- Pilot-plant tests: it attempts reproducing the large-scale process at a smaller scale in a small-scale plant. Yet, the work framework being purely academic, there is no pilot-plant involved.
- Laboratory tests: especially adequate in cases such as material selection or the examination of the corrosion mechanisms. They are mostly accelerated tests employed to foresee the corrosion behaviour of a system with reliable information on the performance of the material system used. The premature changes undergone can be compared to the real time ageing. A particular attention is given to the tests results that should be transferable to the product’s service life. There are basically two types of laboratory tests, which can be divided in two groups:
- Exposure methods: accelerates the materials’ ageing through high temperatures, high concentrated solution, etc. Based on the hygrothermal conditions simulated, there are three classes: simulated atmospheric tests; salt spray tests; and simple immersion tests.
- Advanced electrochemical tests: consists in monitoring the flow of electricity produced during a corrosion process. Quick and industry suited [23]. Enable to determine the reactions’ kinetic and mechanisms.
Laboratory tests best fit the prime objective being the examination of the interaction mechanisms, are chosen. On the one hand, exposure methods could be used over a period of a few months and reveal corrosion rates (in mm/year). On the other hand, they would bring no further understanding of the redox reactions at stake, disallow studying the inner interface of a CFRP-SST-intrinsic hybrid and hence generate only limited data. In contrast, electrochemical tests allow the investigation of localised corrosion forms at best; permit to observe the mechanisms in detail; and enable quantifying the whole process. Therefore, electrochemical techniques are here favoured. To continue the selection of an appropriate test method, three criteria are considered:
- the service conditions: those of an aircraft but simulated at laboratory scale. Salts being present in the airplanes’ environment, it is decided to use 1 M NaCl(aq) in the electrochemical tests.
- the candidate alloys: the austenitic stainless steel AISI 301 (also X10CrNi18-8, see Table 1 for its chemical composition). It presents a high oxidation resistance under normal conditions: in air at ambient temperature, due to its passive behaviour, meaning the formation of an oxide layer of Cr2O3. Yet, in a chloride-containing environment, this stainless steel is prone to corrosion, especially pitting corrosion, a complex localised corrosion form. The second material is an epoxy-based-CFRP (HexPly® 8552 stacked as [0°,90°]s). With a polar behaviour, its absorption behaviour is described by Tsai et al. [22].
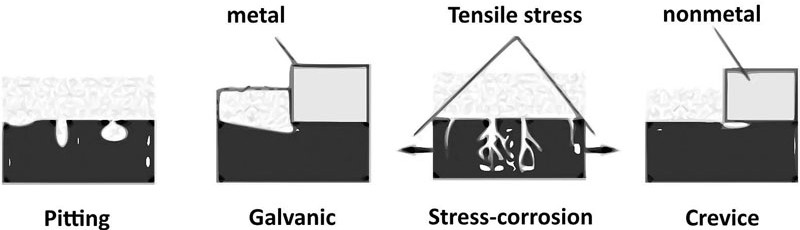
- the possible corrosion failure modes: There are mainly eight corrosion forms [23]. However, considering the stainless steel used and the electrolyte, only pitting and galvanic corrosion are to be expected. Crevice corrosion and stress-corrosion-cracking could be considered if there are defects or additional stress conditions. The expected corrosion forms are localised corrosion types schematized in Figure 4. It should be noted that localized forms are unstable and complex to foresee and monitor [25]. Besides, numerous corrosion cases are caused by several corrosion forms operating all at once [23].
Service conditions, candidate materials and probable corrosion forms having been defined, the adequate electrochemical techniques should be selected. They are divided into two groups:
- Direct current measurements using substantially three characteristics utilised to determine all the corrosion parameters: potential (to foresee the metal’s behaviour such as active, passive or immune), current and resistivity. Several techniques emerge from their combination [23].
- Alternating current measurements or electrochemical impedance spectroscopy (EIS) monitoring the response of a corrosion cell to small-amplitude alternating potential signals over a wide range of frequencies. Thus, EIS can provide several fundamental parameters relating to electrochemical kinetics such as resistance and capacitance elements [23].
The criteria to be fulfilled to sort methods are that it should be adequate for a strongly heterogeneous system and have already been used for both materials, especially the composite part.
Anodic and cathodic polarisation as an answer
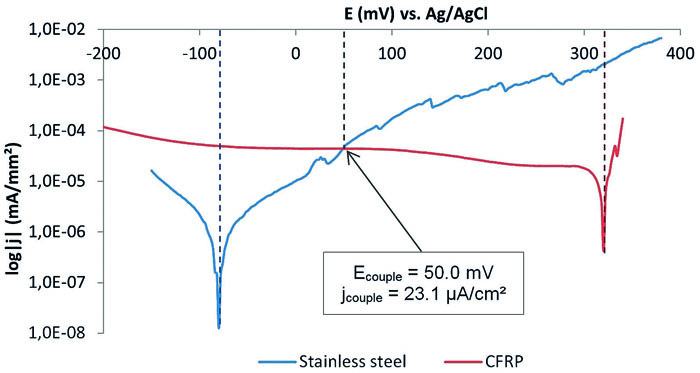
Polarisation tests enable to test the hybrid’s components separately in different corrosive media. This corrosion testing method requires electrical conductors, SST and CFRP here, and probes small enough to suit into the corrosion cell. Thereby, the potential (E, in volt) is varied at a specific rate (usually in mV/s) while the current (I, in ampere) is constantly measured. Given the sample’s geometry, the current density can be calculated and the polarization curve consists in current (or current density) as a function of the potential. Pre-tests consisting of potentiodynamic polarisation permitted to separately record the polarisation curves of the two ground elements of an intrinsic hybrid: CFRP and SST. Given the location of the intersection point of the curves, they indeed permitted to conclude that only the anodic branch of SST and cathodic branch of CFRP are needed to be recorded. The testing procedure has hence been optimised and performed. Besides, it is common to implement electrochemical tests with other techniques in order to supplement the results obtained. Therefore, weight loss, pH and temperature are monitored as useful additional operational data. Furthermore, the tested samples are optically and microscopically examined. Figure 5 displays the results, where the polarisation curves obtained for SST and CFRP are overlaid.
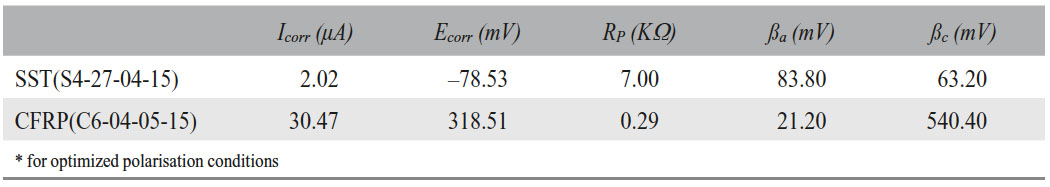
This enables an analysis, in which galvanic corrosion parameters are quantified for an intrinsic CFRP-SST-hybrid (the couple current Icouple and the couple potential Ecouple). Thus, several conclusions can be made from the diagram in view of the three essential parameters named before: potential, current and resistivity. First, the open circuit corrosion potentials are determined at the minimum of the current density. The carbon composite material possesses a more positive corrosion potential (319 mV vs Ag/AgCl) than the stainless steel electrode (–79 mV vs Ag/AgCl). Consequently, CFRP could form a galvanic corrosion cell with SST in which CFRP is the cathode and stainless steel the anode. In addition, the system displays a critical potential difference of 398 mV between both materials, which is a necessary but not sufficient condition for stainless steel to corrode. However and according to the prediction, the galvanic couple potential of 50 mV is closer to the corrosion potential of SST so the galvanic corrosion reactions in 1 M NaCl(aq) are anodically controlled. Therefore, one can conclude that the stainless steel of a CFRP/stainless-steel-hybrid would oxidise if carbon composite also reacts. Secondly, the couple current density (jcouple) has a value of 23.1 μA/cm2. Yet, its calculation presents a certain source of error since the Tafel branches’ slopes are not clear-cut. Besides, although galvanic corrosion between SST and CFRP can contribute to a detrimental decrease in mechanical properties, this corrosion rates might be too low to cause the type of failures encountered in the field. To clarify this, the measured corrosion densities have to be compared to calculations based on Faraday’s law, which indicate how much SST can be removed in one year for 1 μA/cm2 corrosion current density. Thirdly, the polarisation resistance is calculated from the corrosion current through the Stern-Geary equation and the needed parameters calculated are given in Table 2:
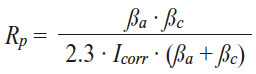
The polarisation resistance also called corrosion resistance of the stainless steel electrode (7 kΩ) is higher than the one of the CFRP sample (390 Ω). This may be the consequence of the stainless steel’s passivation. Yet, chloride ions can break down this passivity leading to actively corroding steel at several orders of magnitudes higher rates.
Based on the optical observation of the polarised SST, the Cr2O3 passive oxide film did fail and pits appeared locally, which indicates that the corrosion form at stake is pitting corrosion. In addition, the pH increases from neutral (pH = 7) to basic pH (pH = 10.5) during the experiment and corrosion products with a characteristic dark brown colour are massively generated. This is probably due to ferrous hydroxides. For a more precise determination, the solid products have been filtered and will be analysed.
Conclusion and outlook
The use of hybridisation of composites with metal is an adequate and feasible design approach to generate a material with better mechanical properties than its elements taken separately. The application field taken into consideration is the aircraft industry for which local intrinsic hybridisation offers the best lightweight potential and shows great connection efficiency for conventional mechanical joining with bolts under standard conditions. In view of the lack of research, the first step defined is the development of a corrosion test program to understand the mechanisms at stake. Accelerated test methods based on electrochemical techniques enabled to study the corrosion behaviour of the hybrid material under focus. The first results consisted in anodic and cathodic polarisation of the two elements of an intrinsic CFRP-SST-hybrid, respectively for carbon composite and stainless steel AISI 301. So far, with the anodic and cathodic polarisations, it can be shown that the combination of stainless steel and CFRP could interact in a chloride-containing solution and the parameters allowing delimiting the corrosion extent were defined: galvanic couple potential, galvanic couple current density and the corrosion resistivity. Nevertheless, the study is not yet finished and some points to be clarified include:
- The polarisation curves of the carbon fibre element will be recorded in order to clarify the (non)-favouring role of the epoxy resin in the CFRP in view of the potential difference with SST. Moreover, as an exemplary test, a polarisation curve will be taken for an intrinsic CFRP-steel-hybrid to verify the extent of the prediction.
- The analysis of the filtered corrosion products.
- The zero-resistance ammeter method will be employed to better determine the corrosion current density of each material. Then the quantity of SST removed will be defined for 1 μA/cm2 corrosion current density and then compared with the measured corrosion current densities.
As an outlook, corrosion of CFRP-SST-hybrid laminates is possible in a harsh and corrosive environment but its extent for real structures should be further investigated.
References
- A. Fink: Lokale Metallhybridisierung zur Effzienzsteigerung von Hochlastfügestellen in Faserverbundwerkstoffen. PhD thesis, Technische Universität Braunschweig, Germany, April 2010
- B. Kolesnikov; L. Herbeck; A. Fink: CFRP/titanium hybrid material for improving composite bolted joints, 83(2008)4, Pages 368–380,
- Flight International, 2005, Retrieved on 03.12.2015, https://www. flightglobal.com/assets/getAsset.aspx?ItemID=9116
- A. Fink; P.P. Camanho; J.M. Andrés; E. Pfeiffer; A. Obst: Hybrid CFRP/titanium bolted joints: Performance assessment and application to a spacecraft payload adaptor, Composites Science and Technology, 70(2010)2, 305–317
- T. Sinmazçelik; E. Avcu; M.Ö. Bora; O. Çoban: A review: Fibre metal laminates, background, bonding types and applied test methods, Materials and Design, 32(2011)7, 3671–3685
- S.Y. Park; W.J. Choi; H. S. Choi: A comparative study on the properties of GLARE laminates cured by autoclave and autoclave consolidation followed by oven postcuring, The International Journal of Advanced Manufacturing Technology, 49(2010),5-8, 605–613
- S.U. Khan; R.C. Alderliesten; R. Benedictus: Post-stretching induced stress redistribution in Fibre Metal Laminates for increased fatigue crack growth resistance, Composites Science and Technology, 69(2009)3-4, 396–405
- Y. Yu; I.A. Ashcroft; G. Swallow: An experimental investigation of residual stresses in an epoxy-steel laminate, International Journal of Adhesion and Adhesives, 26(2006), 511–519
- B. Kolesnikov; D. Stefaniak; J. Wölper; C. Hühne: Adaptive, Tolerant and Efficient Composite Structures, M. Wiedemann and M. Sinapius (eds.), chapter Payload Adapter – Fibre-Metal-Laminate- Laminate Struts (2012), 263–274, Springer-Verlag Berlin Heidelberg
- A. Vlot; J.W. Gunnink: Fibre Metal Laminates: An Introduction, Kluwer Academic Publishers, 2001
- L.B. Vogelesang; A. Vlot: Development of fibre metal laminates for advanced aerospace structures, Journal of Materials Processing Technology, 103(2000)1, 1–5
- W.-X. Wang; Y. Takao; T. Matsubara: Galvanic Corrosion-Resistant Carbon Fiber Metal-Laminates, In 16th International Conference on Composite Materials, Kyoto, Japan (2007), 1–10
- D.F. Silva; E.C. Botelho; A.C. Ancelotti; C.A. Damato: Environmental Conditioning Effects on the Mechanical Properties of Titanium Fiber-Metal-Laminates, In ICCM19 – The 19th International Conference on Composite Materials, 2013
- B. Kolesnikov; A. Fink; C. Hühne; D. Stefaniak; H. Borgwardt: DE102010035324.8-16, Strukturelement aus einem Hybridlaminat, 2010
- D. Stefaniak; A. Fink; B. Kolesnikov; C. Hühne: Improving the mechanical performance of CFRP by metal-hybridisation, In ICCS16 – 16th International Conference on Composite Structures, Porto, 2011
- S. P. Chiew; Y. Yu; C. K. Lee: Bond failure of steel beams strengthened with FRP laminates – Part 1: Model development, Composites Part B: Engineering, 42(2011)5, 1114–1121
- D. Stefaniak; E. Kappel; B. Kolesnikov; C. Hühne: Improving the mechanical performance of unidirectional CFRP by metal-hybridization; ECCM15 – 15th European Conference on Composite Materials, Venice, Italy, June 2012
- R. Prussak; D. Stefaniak; C. Hühne; M. Sinapius: Residual Stresses in Intrinsic UD-CFRP-Steel-Laminates – Experimental Determination, Identification of Sources, Effects and Modification Approaches, Materials Science Forum, Vols. 825-826, pp. 369–376, 2015
- Corrosion Costs and Preventive Strategies in the United States, 2012, Retrieved on 22.10.2015, https://www.nace.org/uploadedFiles/Publications/ccsupp.pdf
- This day in aviation, Important Dates in Aviation History, Retrieved on 03.12.2015, http://static.thisdayinaviation.com/wp-content/uploads/ tdia//2013/04/Boeing-737-297-N73711-Aloha-Airlines-Flight-243- right-side-view.jpg
- M. Mandel; L. Krüger: Electrochemical corrosion studies and pitting corrosion sensitivity of a self-pierce rivet joint of carbon fiber reinforced polymer (CFRP) – laminate and EN AW-6060-T6, Materialwissenschaft und Werkstofftechnik, 43(2012)4, 302–309
- Y.I. Tsai; E.J. Bosze; E. Barjasteh; S.R. Nutt: Influence of hygrothermal environment on thermal and mechanical properties of carbon fiber/fiberglass hybrid composites, Composites Science and Technology, 69(2009)3-4, 432–437
- J.R. Davis: Corrosion: Understanding the Basics, ASM International, 2000
- DIN EN 10088-1:2005(D), Nichtrostende Stähle – Teil 1: Verzeichnis der nichtrostenden Stähle, Technical report, DIN Deutsches Institut fuer Normung e. V., 2005
- D. A. Jones: Principles and prevention of corrosion, Prentice Hall, 1996
PDF Version of the article |
||
| [qrcode] | ||




![Fig. 1: A380-800 material mix [3]](https://www.jept.de/wp-content/uploads/2017/04/kleinViandierBild1.jpg)
![Fig. 3: Fuselage of Aloha Airlines Flight 243, 1988 [20]](https://www.jept.de/wp-content/uploads/2017/04/kleinViandierBild3.jpg)
![Fig. 4: Possible corrosion forms [23]](https://www.jept.de/wp-content/uploads/2017/04/kleinViandierBild4.jpg)