NickFor post treating anodising layers on aluminium, typically two different technologies are applied, the hot water sealing at 96-100 °C and the cold sealing using reactive salts to plug the pores of the anodic coating. Both applications show major disadvantages. Whereas the hot water sealing is extremely energy consuming due to the mandatory hot process temperature, the low temperature sealing processes typically apply nickel compounds being harmful to the environment. Nickel salts are toxic and carcinogenetic, having irreversible effects on the human body and health. Furthermore, nickel containing waste waters are difficult to treat, especially when also aluminium is present [1]. New nickel-free technologies have been developed accordingly, enabling a low temperature application yielding in major energy savings. The deposition of antisoluble compounds in the pores of the anodizing layer leads to best stability and corrosion protection, exceeding the performance of hot water sealing. The new process solutions as being non-toxic are less risky to store and to handle, assisting the safety at work. Implementing a new photometrical method for analysing the ingredients, process stability and production quality can be improved [2]. In some cases, the pH-resistance of the anodised surface can be enhanced, extending the application field of anodised aluminium. Moreover, the waste water treatment of the rinses is carried out at pH 9–10, hence, can be done mutually with aluminium containing effluents.
Introduction
The sealing is one of the most sensitive steps during the anodising process as it presents the final surface treatment, defining most important properties like appearance and corrosion protection. Since long, the hot sealing technology at the boiling temperature of water and the cold sealing technology applying nickel salts are state of the art.
The advantage of the cold sealing is its low application temperature. The disadvantage is the use of toxic nickel compounds. Obviously, the question is how to avoid nickel and nevertheless keep low application temperatures.
Nickel Cold Sealing – State of the Art
Because of the low application temperature of typically 28 °C and the short application time, nickel containing cold sealings are excellent for saving energy and simultaneously providing a good resistance of the anodising layer. Additionally, in case of best maintenance, the sealing bath has a long life time, minimising the chemical consumption. The cold sealing usually is based on nickel fluoride and it can be used as stand alone sealing as well as in combination with successive hot water sealing.
Mechanism
During the cold sealing nickel hydroxide is deposited in the pores of the anodising layer and the fluoride reacts with the aluminium hydroxide forming aluminium fluoride. Simplified, the reaction can be described as in equation 1.
Al2O3 + 3 NiF2 + 3 H2O → 2 AlF3 + 3 Ni(OH)2
The nickel hydroxide does not fill the complete pore, but plugs the outer area, just as the cork of a bottle (Fig. 1).
![Fig. 1: Scheme of the anodising layer [3]](https://www.jept.de/wp-content/uploads/2017/04/WeigeltBild1.jpg)
Fig. 1: Scheme of the anodising layer [3]
Nickel is Hazardous
Despite the well known fact that nickel compounds are toxic and are causing cancer, they still are used for the sealing because the process is robust and the sealing quality is good. Especially decorative surfaces coloured with organic dyes often are treated with nickel containing sealings. During the subsequent handling of such surfaces it cannot be ruled out completely that traces of nickel are released from the pores, for instant in case of skin contact. Some people in turn can develop nickel contact allergies causing itching and reddened skin (Fig. 2). Critical fields of application in this respect are of course the medical sector but also costume jewellery, containers in the cosmetic industry and items like camera objectives or electronic equipment.
Furthermore, the concentrates of the nickel cold sealing as well as the process solutions are hazardeous to the environment and harmful (Fig. 3). Storing and handling these substances request special precautions and work protection.
Cold Sealing(R) – Evolution without Nickel
The novel cold sealing SurTec 350 works free of nickel, cobalt or chromium. Based on zirconium salts, the concentrate is not hazardous to the environment and it does not cause serious health effects on exposure. The working bath needs no risk label at all (Fig. 3).
Simplified Waste Water Treatment
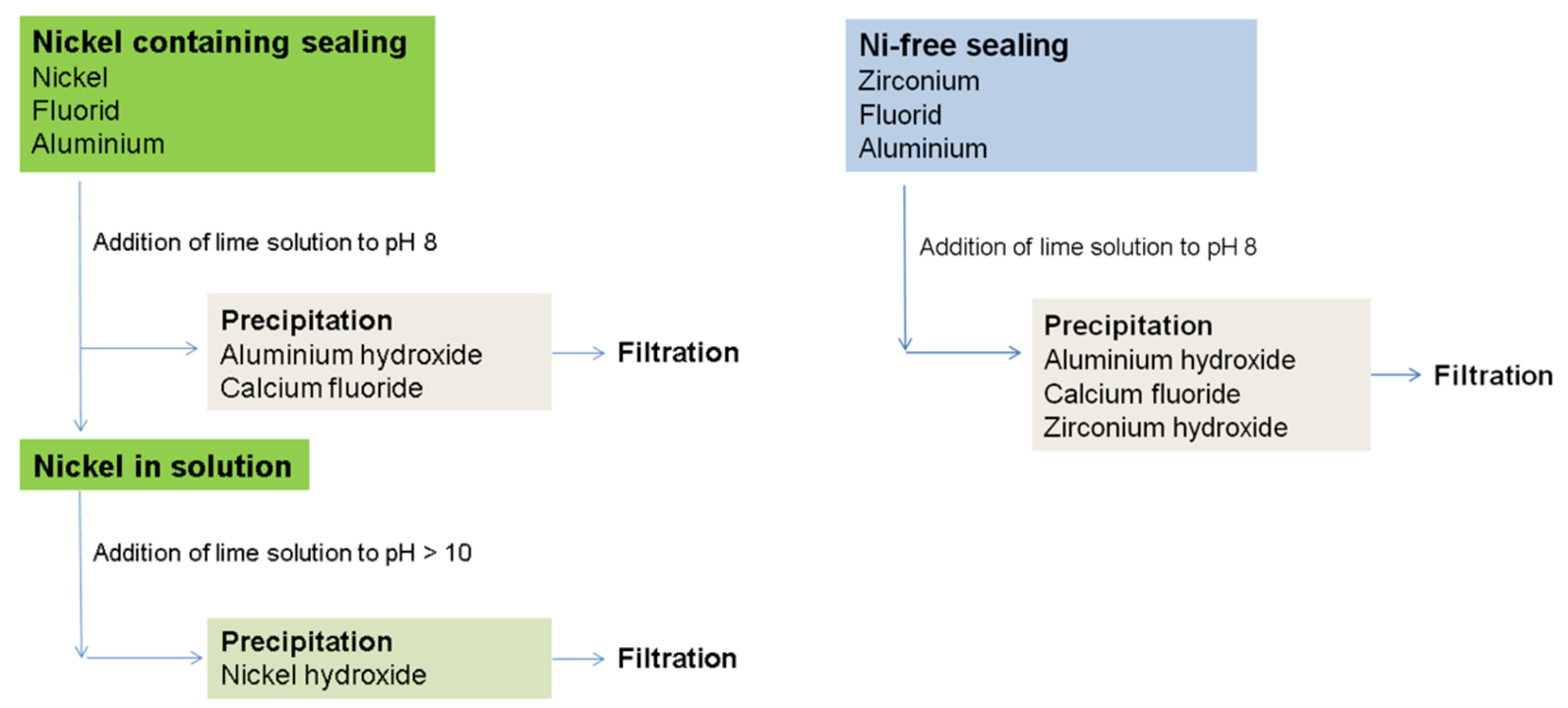
Besides the excellent ecotoxicology also the treatment of waste waters, e.g. rinsing waters from the anodizing plant, is easy. Whereas nickel and aluminum containing waste needs a 2-step treatment to guarantee the very low limit value of nickel in the sewage, the Ni-free cold sealing can be treated in a single step together with aluminum. During the effluent treatment of the Ni-free cold sealing, the zirconium precipitates simultaneously with the aluminum at pH 8. Not to mention that presently no limit value is defined for zirconium in the effluent. Figure 4 shows and compares the waste water treatment of a nickel containing and the Ni-free process.
Process Parameters
The Ni-free cold sealing SurTec 350 typically is applied in DI-water at 3–7 %v/v. The pH-value is adjusted to 5.4–5.8 and the application temperature is possible in a rather wide range from 15–30 °C. The treatment time depends on the thickness of the anodising layer and takes 1 min per μm, whereupon the minimum treatment time should be 5 min and the maximum treatment time should be 10 min.
Analytical Control, Maintenance and Mode of Action
The analytical control of the process solution is fast and easy. In order to maintain the process, either a photometric analysis of the zirconium or the control of the free fluoride by measuring it with a fluoride sensitive electrode is applicable. The photometric determination can be done with the test kit LCK 364 of Hach Lange (Fig. 5) that had been developed mutually by Hach Lange and SurTec [5].
The effective components of the process are consumed evenly, therefore, either one or the other method is sufficient for the analytical control. Also because of the even consumption, the replenishment can be done with a single concentrated additive that also is used in case of a fresh bath make up. In order to evaluate the mode of action, Ni-free cold sealed surfaces were analysed by Scanning Electron Microscopy (SEM) and Energy Disperse X-ray Spectroscopy (EDX). Figure 6 shows a fraction of the anodising layer at a magnification of 10 000 times.

Fig. 6: Left: Fraction of the Ni-free cold sealed anodising layer; Right: Magnification of the upper area of the anodising layer showing a dense appearing and completely sealed surface
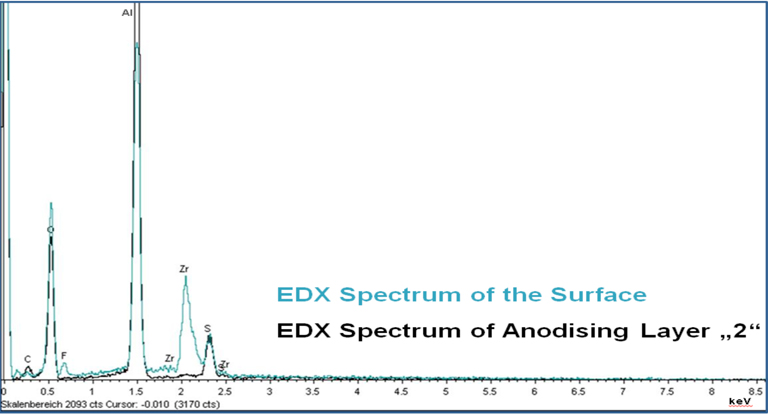
In the upper part of the anodising layer, a more dense appearing area is noticeable. The thickness of this area was measured at different sites and it was determined to be approximately 180–200 nm thick. An EDX analysis in this thin and completely sealed area technically was impossible since the electron beam of the SEM excites a far bigger area of about 1 μm. In order to estimate how deep the pores are filled with zirconium, at first, a spectrum of the surface was taken (blue curve in Fig. 7).
This spectrum sums up all elements present within the first 1 μm of the anodising layer. Besides aluminium, oxygen and sulphur from the anodising process, significant amounts of zirconium and fluoride are detectable. A second spectrum was taken of the middle of the anodising layer fraction marked with “2” on the left picture of Figure 6. This spectrum is shown in black colour in Figure 7 and does not exhibit zirconium or fluoride, hence, indicating that the pores are not sealed completely. Just as in case of the nickel containing cold sealing, also the Ni-free cold sealing plugs the upper area of the anodising layer, whereas the inner area is not sealed completely.
Performance
The nickel-free cold sealing matches the values typically demanded for cold sealed aluminium. Typical results for Ni-free treated EN-AW 5005 and EN-AW 6060 are shown in Table 1.
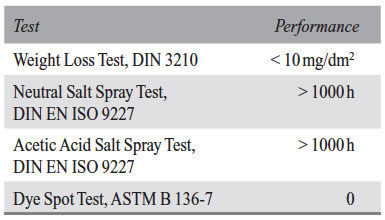
Table 1: Typical performance for EN-AW 5005 or EN-AW 6060 sealed with the Ni-free cold sealing SurTec 350
Applying the Ni-free cold sealing, also alkaline resistant layers can be produced. Combined with a successive hot water sealing, a resistance against pH 12.5 (VOLVO VCS 1026,8198) can be provided. In Figure 8 the effect of the Ni-free process as cold pre-sealing is shown for matt and polished surfaces that had been anodised with 10 μm.
To indicate the effect of the alkaline test solution at pH 12.5, the lower areas of the parts were immersed into it. The regular hot water sealing at > 96 °C, 30 min was not sufficient to obtain an alkaline resistant surface and the surface appears dull. The combination of the Ni-free cold sealing SurTec 350 (3 %v/v, pH 5.6, 25 °C, 10 min) and a subsequent hot water sealing (> 96 °C, 30 min) yielded in alkaline resistant anodising layers and the tested area appears nearly unchanged.
Conclusions
A novel cold sealing was developed being free of nickel, cobalt or chromium. The important quality criteria like weight loss or corrosion protection are fulfilled and combined with a successive hot water sealing also alkaline resistant anodizing layers can be generated that comply with VOLVO VCS 1026,8198. Applying zirconium it is possible to cold seal anodizing layers at a broad temperature range of 15–30 °C. During the processing, the Ni-free cold sealing is easy to handle and it is not hazardous to the environment and also does not cause serious health effects on exposure. Indeed, the working bath needs no labelling at all. The process works stable and can be maintained by measuring the concentration of fluorides, or most easy by measuring the zirconium content photometrical. As the reactive components are consumed evenly, only one liquid concentrate is necessary for maintenance.
References
- W. Hübner; C-T. Speiser: Die Praxis der anodischen Oxidation des Aluminiums, Aluminium Verlag, Düsseldorf, 1988
- C. Weigelt: Kontrolle von Konversionsschäden mittels Farbstoffnachweis, Pulversymposium Dresden 2014
- http://www.alutecta.de/alutecta_home/0_p/alugrafikeloxal.jpg (11.12.2014)
- http://www.hautarzt-hoerner.at/0336kontaktallergie.htm (11.12.2014)
- C. Weigelt: JOT, Springer fachmedien Wiesbaden GmbH (2013)8, 46–47
PDF Version of the article |
||
| [qrcode] | ||


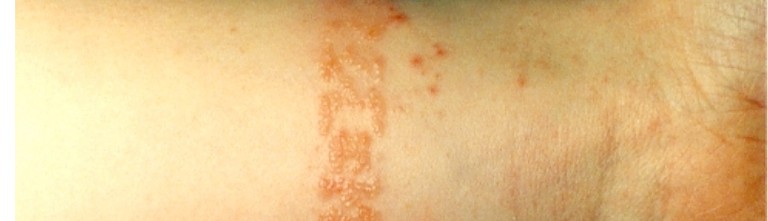


![Fig. 2: Eczema caused by nickel containing wrist band [4]](https://www.jept.de/wp-content/uploads/2017/04/WeigeltBild2.jpg)