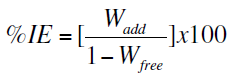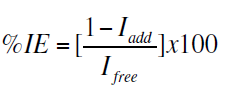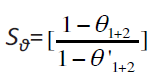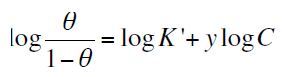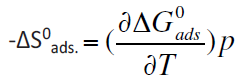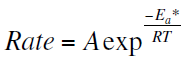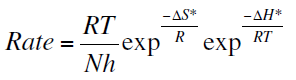The inhibiting effect of some coumarin derivatives toward the corrosion of Zinc in 0.1M HCl solution was studied using weight loss and galvanostatic polarization techniques. Addition of KI to acidic medium containing the coumarin derivatives increases the inhibition efficiency of the system. The obtained results showed that the inhibition efficiency of these compounds increased by increasing their concentrations and decreased by rising the temperature, so that the adsorption of these compounds is physically adsorbed on the zinc surface. Temkin’s adsorption isotherm fits the experimental data for the studied compounds. Some thermodynamic parameters for the adsorption and activation process were computed. The values of Tafel slopes indicate that these compounds act as a mixed type inhibitors but cathode is more polarized when an external current was applied. The inhibitors are explained in terms of adsorption on the zinc surface. The order of inhibition efficiency are interpreted on the basis of the molecular structure, the subsistent groups and their charge densities of the coumarin derivatives.
1. Introduction
Zinc and their alloys are exposed to attack by acids especially hydrochloric acid. For scale removal and cleaning surfaces with acidic solutions, it is become necessary to added corrosion inhibitors to minimize metal loss and reduce acid consumption. Several organic compounds containing nitrogen, oxygen and sulphur atoms are used to inhibit the corrosion of zinc in acidic solutions [1-10]. The inhibiting power of these compounds is attributed to the interaction between zinc surface and inhibitors molecules through their adsorption on the metal surface. The adsorption influenced by factors such as nature and surface charge on the metal, the type of aggressive media, the structure of the inhibitor, and the nature of its interaction with the metal surface [11].
The aim of this study is to investigate the effect of 4-coumarin derivatives inhibitors on the corrosion of zinc in HCl solution using weight loss method (chemical method) and galvanostatic polarization method (electrochemical method) at 303 K. Investigate the synergistic effect by addition of KI to the investigated 4-coumarin derivatives in 0.1M HCl solution. The effect of the temperature on the corrosion of Zn in free and inhibited acid solution was studied. Adsorption and activated thermodynamic parameters were computed.
2. Experimental methods
The zinc (BDH grade) used in this investigation has chemical composition listed in Table 1. For weight loss measurements, the reaction basin used in this method was a graduated glass vessel having a total volume of 100 ml. 100 ml of the test solution was employed in each experiment. The test pieces were 20 x 20 x 2 mm. The samples were first mechanically polished with a fine grade emery paper in order to obtain a smooth surface, followed by ultra-sonically degreasing with acetone and then rinsed with distilled water, dried between two filter papers and weighed. The test pieces were suspended by suitable glass hooks at the edge of the basin, and under the surface of the test solution by about 1 cm.
Weight loss measurements were carried out as described elsewhere [12]. The percentage inhibition efficiency (% IE) and a parameter (θ) which represents the part of the metal surface covered by the inhibitor molecules were calculated using the following equations:
where, Wfree and Wadd are the weight losses of Zn sample in free and inhibited acid solution, respectively.
For galvanostatic polarization measurements, a cylindrical rod embedded in araldite with exposed surface of 0.5 cm2 was used. The electrode was polished with different grades emery paper, degreased with acetone and rinsed by distilled water. Galvanostatic polarization studies were carried out using zinc rod of the same compositions used in weight loss. E vs. log I curves were recorded at temperature 30 oC. A constant quantity of the test solution (100 ml) was taken in the polarization cell. Galvanostatic polarization studies were carried out using EG&G model 173 potentiostat/galvanostat. for accurate measurements of potential and current density. Three compartment cell with a saturated reference calomel electrode and a platinum foil auxiliary electrode was used. Solutions were not deaerated to make the conditions identical to weight loss measurements. All the experiments were carried out at 30±1 oC by using ultra circulating thermostat.
The percentage inhibition efficiency (% IE) was calculated from corrosion current density values using the equation
where, Ifree and Iadd are the corrosion current densities in absence and presence of inhibitors, respectively.
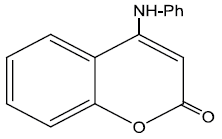
Chemical structure of coumarin derivatives
3. Results and discussion
3.1. Weight – loss measurements
Figure 1 show the weight loss-time curves for zinc in 0.1M HCl solution in absence and presence of different concentrations of compound (b) as an example. Similar curves were also obtained for other two compounds. As shown from Figure 1, it is clear that, as the concentration of these derivatives increase, the weight loss of zinc samples are decreased. This means that the presences of these derivatives retard the corrosion of zinc in 0.1M HCl solution or in other words, these compounds act as inhibitors. The linear variation of weight loss with time in uninhibited and inhibited 0.1M HCl solution indicates that the absence of insoluble surface films during corrosion. In the absence of any surface films, the inhibitors are first adsorbed on to the metal surface and there after impede corrosion either by merely blocking the reaction sites (anodic and cathodic) or by altering the mechanism of the anodic and cathodic partial processes.

Fig. 1: Weight-loss vs. time curves for the dissolution of zinc in 0.1 M HCl in absence and presence of different concentrations of compound (b) at 30 oC.
1) Blank, 2) 3×10-6 M, 3) 5×10-6 M, 4) 7×10-6 M, 5) 9×10-6 M, 6) 11×10-6 M
The percentage inhibition efficiencies (% IE) of these compounds were determined using the equation (1). The calculated values of % IE are given in Table 2, from the values of % IE, the order of the inhibition efficiencies of 4-coumarin derivatives decreased as following: (b) > (c) > (a). This order will be discussed later.

Tab. 2: Inhibition efficiency obtained from weight loss of zinc in 0.1M HCl at different concentrations of the 4-coumarin derivatives after 180 min immersion at 30 oC.
3.2. Synergistic effect of KI
The effect of KI, on the corrosion inhibition of zinc in 0.1M HCl solution in absence and presence of different concentrations of these additives was studied by the weight loss method. Similar curves were obtained to Figure 1 not shown. The percentage inhibition efficiency (% IE) for specific concentrations of KI (1×10-2 M) in the presence of various concentrations of 4-coumarin derivatives is given in Table 3.

Tab. 3: Inhibition efficiency obtained from weight loss of zinc dissolution in 0.1 M HCl at different concentrations of the 4-coumarin derivatives with addtion of 1X10-2 M KI after 180 min immersion at 30 oC.
It is observed that % IE of the inhibitors increases by increasing the concentrations of 4-coumarin derivatives, also the order of inhibition efficiency increased as following: (b) > (c) > (a). The strong chemisorption of iodide ion on the metal surface is responsible for the synergistic effect of this ion leads to greater surface coverage and, therefore, greater inhibition.
The synergistic inhibition effect was evaluated using a parameter, Sθ, obtained from the surface coverage values (θ) of the anion, cation and both. Aramaki and Hackerman [13] calculated the synergism parameter Sθ using the following equation
where: θ1+2 = (θ1 + θ2) – (θ1θ2);
θ1 = surface coverage by anion;
θ2 = surface coverage by cation;
θ’1+2 = measured surface coverage by both the anion and cation;
The synergism parameters was calculated from equation (4). The plot of the synergism parameter (Sθ) against various concentrations of 4-coumarin derivatives given in Figure 2 and the corresponding values are shown in Table 4. As can be seen from this Table, the values of Sθare nearly equal to unity, which suggests that the enhanced inhibition efficiencies caused by the addition of KI to 4-coumarin derivatives is due mainly to the synergistic effect.

Fig. 2: Plots of synergism parameter (Sθ) vs. the concentration of 4-coumain derivatives for dissolution of zinc in 0.1 M HCl in presence of 1×10-2 M KI at 30 oC.

Tab. 4: Synergism parameter (Sθ) for different concentrations of the 4-coumarin derivatives with addition of 1×10-2 M KI.
Synergistic adsorption is classified into two types plus a mixture of the two:
(i) specific co-adsorption of anions and cations, and
(ii) ionic or physical overlap adsorption of cations over the anion covered zinc surface.
3.3. Adsorption Isotherm
Organic molecules like 4-coumarin molecules inhibit the corrosion process by the adsorption on metal surface. Theoretically, the adsorption process can be regarded as a single substitution process between the inhibitor molecule in the aqueous phase I(aq) and the water molecules adsorbed on the metal surface H2O(ads) [14, 15] according to the following equation
where, x is the size ratio and simply equals the number of adsorbed water molecules replaced by a single inhibitor molecule. The adsorption depends on the structure of the inhibitor, the type of the metal and the nature of its surface, the nature of the corrosion medium and its pH value, the temperature and the electrochemical potential of the metal-solution interface. Also, the adsorption provides information about the interaction among the adsorbed molecules themselves as well as their interaction with the metal surface. Actually an adsorbed molecule may make the surface more difficult or less difficult for another molecule to become attached to a neighboring site and multilayer adsorption may
take place. Attempt were made to fit surface coverage (θ) values to various adsorption isotherms including Langmuir, Freundlich, Frumkin and Temkin. By far, the best results were obtained fitted Temkin isotherm according the following equation:
where, C is the inhibitor concentration in the bulk of the solution, a is the interaction parameter and K is the equilibrium constant of the adsorption process which is related to the standard free energy of adsorption (ΔG°ads.) by:
where, R is the universal gas constant and T is the absolute temperature.
Plots of θ vs. log C (Temkin adsorption plots) for adsorption of 4-coumarin derivatives on the surface of zinc in 0.1M HCl acid at 30 °C is shown in Figure 3. The data gave straight lines indicating that Temkin’s isotherm is valid for these systems. Temkin’s isotherm is applied for ideal case of physical and chemical adsorption on a smooth surface with no interaction between the adsorbed molecules.

Fig. 3: Curve fitting of the corrosion data for zinc in 0.1 M HCl in presence of different concentrations of 4-coumain derivatives to the Temkin adsorption isotherm at 30oC
On the other hand, it is found that the kinetic-thermodynamic model of El Awady et al [16].
is valid to operate the present adsorption data. The equilibrium constant of adsorption K = K’ (1/y), where 1/y is the number of the surface active sites occupied by one 4-coumarin molecule and C is the bulk concentration of the inhibitor. Plotting log θ/(1-θ) against log C at 30 °C is given in Figure 4. Straight line relationships were obtained suggesting the validity of this model for all cases studied. The calculated values of 1/y, K and ΔG°ads. are given in Table 5. Inspection of the data of these Tables shows that the large values of ΔG°ads. and its negative sign, indicate that the adsorption of 4-coumarin compounds on the zinc surface is proceeding spontaneously and is accompanied by a highly-efficient adsorption. It is worth noting that the value of 1/y is more than unity. This means that the given inhibitor molecules will occupy more than one active site. In general, the values of ΔG°ads. obtained from El-Awady et al., model are comparable with those obtained from Temkin isotherms.

Fig. 4: Curve fitting of the corrosion data for zinc in 0.1 M HCl in presence of different concentrations of 4-coumarin derivatives to the kinetic model at 30oC

Tab. 5: Inhibitor binding constant (K), Free energy of binding ΔGads, number of active sites ( 1/y) and later interaction parameter (a) for 4-coumarin derivatives at 30oC
The entropies of adsorption ΔS°ads. were calculated from the relation between ΔG°ads. and T:
For calculating the values of heat of adsorption of the various inhibitors, plots of log (θ/1-θ) vs. 1/T. Figure 5. The heat of adsorption (Q) are calculated from the slopes of these plots. The values of ΔS°ads. and Q are given in Table 6. From these results it may be generalized that the more efficient inhibitor has more negative ΔG°ads. value and less values of ΔS°ads. and Q. So that from the tabulated values of ΔG°ads. ΔS°ads. and Q the order of inhibition efficiencies is as following: (b) > (c) > (a). This trend will be interpreted latter.

Fig. 5: Log θ/(1- θ) vs. 1/T plots of for the dissolution of zinc in 0.1 M HCl in presence of 11×10-6 M of 4-coumarin derivatives at 30 oC.

Tab. 6: Thermodynamic parameters for the adsorption of 4-coumarin derivatives in 0.1 M HCl on zinc surface.
3.4. Effect of Temperature
The effect of rising temperature from 30° to 55°C on the corrosion of zinc electrode in 0.1M HCl solution in the presence of 11×10-6 M of 4-coumarin derivatives was studied using weight-loss measurements. Similar curves to Figure 1 were obtained (not shown). As the temperature rises the dissolution rate of zinc increase and the percentage inhibition efficiency decrease. This behavior indicated that the adsorption of 4-coumarin derivatives on the zinc surface is physically adsorption.
Activation parameters for corrosion of zinc were calculated from Arrhenius-type equation [17, 18]
and transition-state equation:
where, A is the frequency factor, h is the Plank’s constant, N is Avogadro’s number and R is the universal gas constant. Ea is the apparent activation energy, ΔH* is the enthalpy of activation and ΔS* is the entropy of activation.
Figure 6 represents the relation between the logarithm of corrosion rate (logRate) versus 1/T. The values of activation energy Ea can be calculated from the slope of the straight line in Figure 6. The values of Ea are listed in Table 7. It obvious that the presence of the studied compounds increased the values of Ea. This indicate that the 4-coumarin derivatives acted as inhibitors through increasing the activation energy of metal dissolution by making a barrier to mass and charge transfer by their adsorption on metal surface [19].

Fig. 6: Log corrosion rate vs. 1/T plots of for the dissolution of zinc in 0.1 M HCl in presence of 11×10-6 M of 4-coumarin derivatives at 30 oC.

Tab. 7: Activation parameters of the dissolution of zinc in 0.1M HCl in the absence and presence of 11×10-6 M 4-coumarin derivatives.
Figure 7 shows a plot of log (Rate/T) vs. 1/T. A straight lines are obtained with slope equal to (-ΔH*/2.303R) and an intercepts of (log R/Nh + ΔS*/2.303R) from which the values of ΔH* and ΔS* are calculated and listed in Table 7.

Fig.7: Log corrosion rate / T vs. 1/T plots of for the dissolution of zinc in 0.1 M HCl in presence of 11×10-6 M of 4-coumarin derivatives at 30 oC.
The increasing in the temperature has a reverse relationship with the percentage efficiency this means that the adsorption of 4-coumarin derivatives on the metal surface is physically. This behavior can be explained on the basis that the increase of the temperature leads to desorption of the adsorbed molecules of the inhibitors from the metal surface.
The calculated values of the apparent activation energy, Ea, activation entropies, ΔS* and activation enthalpies, ΔH* are given in Table 7. The almost similar values of Ea suggest that the inhibitors are similar in the mechanism of action and the order of efficiency may be related to the preexponential factor A in equation (11). This is further related to concentration, steric effects, metal surface characters. The order of the inhibition efficiencies of 4-coumarin derivative as gathered from the increase in Ea* and ΔH* values and decrease in ΔS* values is as following: (b) > (c) > (a)
3.5. Galvanostatic Polarization
The effect of 4-coumarin derivatives on the anodic and cathodic polarization curves of zinc electrode in 0.1M HCl solution at 30 °C was studied.The effect of increased concentrations of compound (b) Figure 8 as an example. Similar curves were also obtained for other two compounds (not shown). The corrosion parameters such as the corrosion current density (Icorr.), corrosion potential (Ecorr), anodic and cathodic Tafel slopes (βa and βc) and inhibition efficiency (% IE) are calculated and given in Table 8.

Fig. 8: Galvanostatic polarization curves for dissolution of zinc in 0.1 M HCl in absence and presence of different concentrations of compound (b) at 30 oC.
1) Blank, 2) 1×10-6 M, 3) 3×10-6 M, 4) 5×10-6 M,
5) 7×10-6 M, 6) 9×10-6 M, 7) 11×10-6 M

Tab. 8: Electrochemical parameters obtained from galvanostatic polarization of Zn in 0.1M HCl solution containing different
concentrations of inhibitors at 30˚C.
Inspection of Table 8, it is clear that, the corrosion current density (Icorr) was determined by the intersection of the extrapolation anodic and cathodic Tafel lines with the steady state (corrosion) potential Ecorr.
Inspection of Table 8 reveals that:
i) The increase in concentration of the additives showed that, the cathodic and anodic curves obtained exhibit Tafel-type behavior. Addition of 4-coumarin derivatives increased both cathodic and anodic overvoltages and caused mainly parallel displacement to the more negative and positive values, respectively.
ii) Ecorr was almost constant and the values of the corrosion current density (Icorr.) decreases with increasing the concentration of 4-coumarin derivatives indicating that the presence of these derivatives retards the dissolution of zinc in 0.1M HCl and the degree of inhibition depends on the concentration and type of the inhibitor present.
iii) The values of anodic (βa) and cathodic (βc) Tafel slopes are nearly constant indicating the inhibitor acted by blocking the reaction sites of the metal surface without changing the anodic and cathodic reaction mechanism [20]. However, the data suggested that these compounds act mainly as mixed-type inhibitors.
iv) The inhibition efficiencies of the three tested compounds increased in the following order (b) > (c) > (a).
3.6. Chemical Structure of Inhibitors and Corrosion Inhibition
Inhibition of corrosion of zinc in 0.1M HCl solution by the investigated 4-coumarin derivatives as measured by chemical and electrochemical measurements was found to depend on both the concentration and the nature of the inhibitor. The observed corrosion date in presence of the inhibitors, namely. The decrease of corrosion rate with increasing the concentration of the inhibitors. The linear variation of weight loss with time. The parallel shift in Tafel lines to higher potential values, and the decrease in corrosion inhibition efficiency with increasing temperature.
These observations indicate that the corrosion inhibition is due to adsorption of the inhibitors at the solution/interface [21, 22]. The nature of inhibitor interaction on the metal surface during corrosion inhibition can be explained in terms of its adsorption characteristics. However, inhibition efficiency of additive compounds depends on many factors, which include the number of adsorption active centers in the molecule and their charge density, molecular size, mode of adsorption, heat of hydrogenation and formation of metallic complexes.
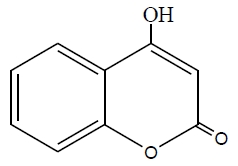
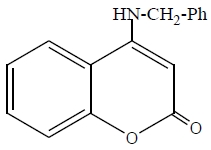
The inhibition efficiency of the three studied derivatives: (a), 4-hydroxy coumarin, (b), 4-benzyl amino coumarin, and (c), 4-phenyl amino coumarin, for zinc in 0.1M HCl solution decreases in the order: (b) > (c) > (a). The extent of inhibition depends on the molecular size of the molecule and the electron density (donating or with drawing) of the subsistent groups. Skeletal representation of the mode of adsorption of the tested derivatives is shown in Figure 9. Compound (b) exhibits excellent inhibition power due to: (i) its larger molecular size that may facilitate better surface coverage, (ii) its adsorption through two active centers as shown from Figure 9, and (iii) the benzyl group which is highly electron releasing group which enhance the delocalized π-electrons on the active centers of the compound. Compound (c) comes after compound (b) in inhibition efficiency in spite of it has two active centers, because it has lesser molecular size. Compound (a) has the lowest inhibition efficiency; in spite of it has two active centers. This is because it has the lowest molecular size.
4. Conclusions
- 4-coumarin derivatives act as inhibitors for the corrosion of zinc in 0.1M HCl solution.
- The inhibition efficiencies of 4-coumarin derivatives is increase in concentration of these inhibitors but decreases with increase temperature as following (b) > (c) > (a).
- Investigate the synergistic effect of some anions such as KI improve the values of the inhibition efficiency due to synergistic effect.
- This behavior can be explained on the basis that the increase of the temperature leads to desorption of the adsorbed molecules of the inhibitors from the zinc surface.
- The desorption of the adsorbed molecules of the inhibitors from the zinc surface followed Temkin desorption isotherm.
References
- Y. K. Agrawal, J. D. Talati, M. D. Shah, M. N. Desai, N. K. Shah: Schiff bases ofethylene diamineas corrosion inhibitors of zinc in sulphuric acid, Corrosion Science. , vol. 46, p. 633., 2004.
- A. A. Abdel Fattah, M. E. Mabrouk, R. M. Abd El-Galil, M. M. Ghoneim, Hetrocyclic Compounda as Corrosion Inhibitors for Zn in HCl Acid Solutions, Bulletin Society Chemistry France., vol. 127, p.48, 1991.
- K. Olusegun, A. Abiola, O. James: The effects of Aloe vera extract on corrosion and kinetics of corrosion process of zinc in HCL solution, Corrosion Science, vol. 52, p. 661, 2010.
- M. Abdallah, I. Zaafarany, A. S. Fouda, D. Abd El-Kader: Inhibition of zinc corrosion by some benzaldehyde derivatives in HCl solution, J. Materials Engineering and Performance, vol. 21, p. 995, 2012.
- M. Abdallah , A. Y El-Etre, M. F. Moustafa: Amidopoly ethylamines as corrosion inhibitors for zinc dissolution in different acidic electrolyte, Portugaliae Electrochimica. Acta, vol. 27, p. 15, 2009.
- M. S. Abdel-Aal, Z. A. Ahmed, M. S. Hassan: Inhibiting and accelerating effects of some quinolines on the corrosion of zinc and some binary zinc alloys in HCl solution, Journal of Appied Electrochemistry, vol. 22, p. 1104, 1992.
- A. Y. El-Etre, A. Y. M. Abdallah: Z. E. El-Tantawy, .Corrosion Inhibition of Some Metals Using Lawasonia Extract, Corrosion Science, vol 47, p. 385, 2005.
- M. Abdallah: Ethoxylate Fatty Alcohols as Corrosion Inhibitor for Dissolution of Zinc in Hydrochloric Acid, Corrosion Science, vol. 45, p. 2705, 2003.
- M. N. Desai, J. D. Talati, N. K. Sheh: Schiff bases of ethylenediamine / triethylenetetramine with benzaldehyde/cinnamic aldehyde/salicylaldehyde as corrosion inhibitors of zinc in sulphuric acid, Anti–Corrosion Methods and Materials, vol. 55, p. 27, 2008.
- A. S. Fouda, M. Abdallah, S. T. Atwa, M. M Salem: Corrosion inhibition of zinc in 0.4 M HCl solutions using some teterahydrocarbazole derivatives, Modern Applied Science, vol. 4, p. 41, 2010.
- A. J. Szyprowski: Relationship between chemical structure of imidazoline inhibitors and their effectiveness against hydrogen sulphide corrosion of steels Br. Corros. J., vol. 35, p. 155, 2000.
- P. B. Mathur, T. Vasudevan: Reaction Rate Studies for the Corrosion of Metals in Acids. Part 1, Iron in Mineral Acids, Corrosion, vol. 38, p. 171, 1982.
- K. Aramaki, N. Hackerman: Inhibition Mechanism of Medium-Sized Polymethyleneimine, Journal of Electrochemical Society, vol. 116, p. 568, 1969.
- S. S. Abd El Rehim, M. A. Amin, S. O.Moussa, A. S. Ellithy: The corrosion inhibition of aluminum and its copper alloys in 1.0 M H2SO4 solution using linear-sodium dodecyl benzene sulfonate as inhibitor, Materials Chemistry and Physics., vol. 112, p. 898, 2008.
- E.-McCaaffery: Corrosion Control by Coating, Science press, Princeton, 1979.
- Y. A. El-Awady, S. A. I. Ahmed: Effect of Temperature and inhibitor on the corrosion of four primary aliphatic amines on mild steel in 2M HCl solution, Journal of Indian Chemistry, vol. 24, p. 601, 1985.
- K. J. Laider: Chemical Kinetics, McGraw-Hill Publishing Company Ltd., 1965.
- S. S. Abd El-Rehim, M. A. Ibrahim, K. F. Khaled: 4-Aminoantipyrine as an inhibitor of mild steel corrosion in HCl solution, Journal of Applied Electrochemistry, vol. 29, p. 593, 1999.
- A. S. Fouda, M. Abdallah, T. Y. Mahmoud, E. Fouad: Phenazone and aminophenazone as corrosion inhibitors for aluminum in HCl solution, Protection of Metals and Physical Chemistry of Surfaces, vol. 47, p. 803, 2011.
- M. H. Hussi, M. J. Kassim: Electrochemical, Thermodynamic and Adsorption Studies of (+)-Catechin Hydrate as Natural Mild Steel Corrosion Inhibitor in 1 M HCl, International Journal of Electrochemical Science, vol. 6, p. 1396, 2011.
- M. Abdallah: Tetradecyl-1, 2-Diol Propen Oxylates as Inhibitors for Corrosion of Aluminium in Hydrochloric Acid. Bull of Electrochem., vol. 16(6), p. 258, 2000.
- I. B. Obotand, N. O. Obi-Egbedi: Adsorption properitis and inhibition of mild steel corrosion in sulphuric acid solution by ketoconazole:experimental and theoritcal investigation, Corrosion Science , vol. 52, p. 198, 2010.
PDF Version of the article |
Epub Version of the article |
Flash Version of the article |
| [qr-code size=”2″] | ||