The electrodeposition of nickel-iron-alloys from a sulfate electrolyte with different additives ist described. As deposited alloys contained between 10 and 36 % of iron and were charaterized by hardness, grain size and lattize parameter. The thermal stability of the different alloys by exposing them up to 800 °C is investigated by cross sections and the mesurement of hardness in order to prove the suitability for usage in microfabrication.
1 Introduction
One of the most cost effective routes for the mass fabrication of microstructured parts or microcomponents from plastics is molding and embossing [1]. The molding tools or mold inserts can be manufactured by electrodeposition of a metal or alloy into a microstructured PMMA layer patterned by deep X-ray lithography because electrodeposition is one of the commonly applied techniques to produce dense films or coatings of metals and alloys. These mold inserts, thereafter, are hot embossed under vacuum into high performance plastics. The process temperatures of the embossing step is chosen with respect to the glass transition temperature of the plastic material which is in the range of 200 °C to 300 °C for high-performance plastics like polyamide (PA), polycarbonate (PC) or polyoxymethylene (POM).
The material mostly applied for mold inserts is electrodeposited nickel (Ni). The high process temperature, however, are not compatible with nickel mold inserts, because of the now recrystallization temperature of electrodeposited nickel of about 200 °C [1]. After several embossing processes the repeated heating cycles weaken the molding tool such that the forces during molding and demolding are sufficient to deform the microstructures on the tool [2] which cannot be used any longer for replication.
One possibility of improving the mold inserts is using a different material. Promising candidates are nickeliron (Ni-Fe) alloys which can be electrodeposited in a wide range of compositions.
2 Experimental Details
Nickel-iron alloys were deposited from a bath containing 45 g/l nickel sulfate (NiSO4 · 6H2O), 2.5 to 6.0 g/l ferrous sulfate (FeSO4 · 7H2O), 25 g/l boric acid (H3BO4), 0.5 g/l dodecyl sulfate ((C12H25O)2SO2) or a flourine-containing surfactant and more than 1500 mg/l saccharin or N-methyl saccharin as a stress-relieving agent dissolved in 500 ml or 3 l of distilled water (Tab. 1). The nickel-iron electroplating set-up consists of an electrochemical cell which is equipped with a Pt 100 thermocouple connected to a thermostat and an argon inlet and outlet. The electrolyte is pumped from the cell into a storage vessel and back to the cell passing a filter and a volume flow sensor.
Tab. 1: Composition of electrolyte used to deposit nickel-iron
A pH-electrode inside the storage vessel connected to a pH controlling unit regulates the pH value of the bath. Electrodepositions were carried out at 50 °C and at different current densities in the range of 50 A/m2 to 300 A/m2 (i.e. 0.5 A/dm2 to 3.0 A/dm2). The nickel-iron electrodeposits were highly adherent to the substrates, had a shiny surface and were free of microcracks for the investigated thickness ranging between 10 μm and 100 μm. Cathodes out of stainless steel or copper were used as substrate material. The anode was made of sulphur-depolarized nickel balls in a titanium basket.
The composition of the electrodeposits was investigated by standardless energy-dispersive X-ray spectroscopy (EDX) in a scanning electron microscope with a 30 kV-electron beam.
Microhardness values were determined on samples of a size of 10 mm × 10 mm and a thickness of about 100 μm perpendicular to the surface, i.e. parallel to the growth direction. In a few cases also hardness perpendicular to the growth direction was measured by embedding the specimens in epoxy resin and preparing a cross-section. In both cases a Vickers indenter and a proof load of 1 N were used. The reported microhardness values, HV0.1, are an average out of at least three different measurements with the standard deviation taken as error.
Quantitative X-ray diffraction (XRD) was carried out on a Siemens D500 X-ray diffractometer using Cu-Kα radiation (λ = 0.15418 nm, U = 45 kV, I = 40 mA). Apertures used in positions I to IV were: 1°, 0.3°, 0.3° and 0.15°, respectively. Cu-Kβ radiation was sup pressed by a thin nickel foil placed between the collimating apertures III and IV on the detector side. Standard θ-2θ scans with a step size of 0.02° and acquisition times between 5 s and 30 s were performed in Bragg-Brentano geometry. The specimens were fixed by pressing them into plasticine. The grain size was estimated from peak broadening of the XRD profiles using Scherrer’s formula [3, 4] and Warren’s correction for instrumental broadening [5]. The measured data were background subtracted and the Kα2 lines were stripped off by built-in functions of the software package Diffract AT, Release 3.3 (Siemens, 1993). The instrumental broadening was taken into account by measuring a XRD profile of a nickel-iron electrodeposit heat treated for one hour at 800 °C taken as a standard.
3 Results
3.1 Influence of deposition parameters on the microstructure of Ni-Fe deposits
One of the most important parameters for electroplating is the current density during deposition. To study its influence on the microstructure a series of nickeliron deposits about 10 μm in thickness was produced for varying current densities from 50 A/m2 to 300 A/m2 (i.e. 0.5 A/dm2 to 3.0 A/dm2). A bath composition with an iron concentration of 62.7 mmol/l (= 3.5 g/l), a pH value of 2.7, bath volume of 500 ml and use of a fluorine-containing surfactant (FT) were kept constant throughout all these depositions. Figure 1 shows the XRD profiles for scattering angles 2θ between 30° and 60°. The two peaks visible for each deposit correspond to Bragg reflections of {111} nd {200} lattice planes, respectively, of a face-centered cubic (fcc) lattice. No other peaks were detected throughout this study. Thus, the nickel-iron electrodeposits were entirely composed of a solid solution of iron in nickel.

Fig. 1: XRD profiles of a series of Ni-Fe electrodepositions for varying current densities from 50 A/m² up to 300 A/m² and constant pH value of 2.7. A fluorine-containing surfactant (FT) was used. For increasing current densities the peak maxima are shifted towards smaller values of 2θ indicating an increase of the lattice parameter and the {200}/{111} peak ratio is drastically changed.
Two further phenomena are obvious: firstly, the ratio of peak heights is drastically changed as a function of current density, and secondly, for decreasing current densities the peaks are shifted towards higher scattering angles indicating a shrinking lattice parameter.
In order evaluate the change in peak heights the background of the XRD profiles was subtracted and the heights of {111} and {200} peaks were determined using built-in functions of the Diffrac AT software package. The ratios of peaks heights were then calcuverlated and collected together with other data in the first five rows of Table 2.
Tab. 2: Ratio of {200}/{111} XRD peaks, lattice parameter and iron content of a series of nickel-iron alloys deposited with different current densities, pH values and two different surfactants

At the highest current density of 300 A/m² the {200}/ {111} peak ratio of 0.55 is close to that for a powder specimen of a fcc metal with randomly oriented grains [6]. The lower the current density the more {200}-oriented grains are deposited. Therefore, the orientation of the grains is changed from nearly random towards a preferential {200}-orientation. For comparison, a series of pure nickel deposits was produced using a bath identical to the one applied for the nickel-iron deposition but without addition of iron sulphate. In the case of pure nickel no severe change in texture was observed for current densities ranging from 50 A/m2 to 300 A/m2. All deposits exhibited a pronounced {200} peak and the {200}/{111} peak ratio ranged between 2.4 and 6.0. Therefore, the change in preferred grain orientation as a function of current density seems to be a special feature of the nickeliron electrodeposition at least for the range of current densities applied.
In addition, the influence of other deposition parameters like pH value and surfactant were also screened. The corresponding XRD profiles together with those already shown in Figure 1 are compiled in three plots of Figure 2: plot (a) for a constant current density, plot (b) for a constant surfactant and plot (c) for a constant pH value. The value of the parameters varied during deposition and the composition of the deposits determined by EDX are denoted directly at the respective XRD profiles in Figure 2. A complete list of the data can be found in Table 2. From Figure 2 it can be extracted that current density has a stronger influence on composition and texture, i.e. {200}/ {111} peak ratio, than any other parameter of the deposition process. Keeping all other parameters constant the two XRD profile for 50 A/m2 and 300 A/m² for a constant pH value (Fig. 2b) and constant surfactant (Fig. 2c) show significant differences concerning composition and also {202}/{111} peak ratio. Thus, current density during deposition proved to be the dominant factor influencing composition as well as texture of nickel-iron electrodeposited from the selected electrolyte.
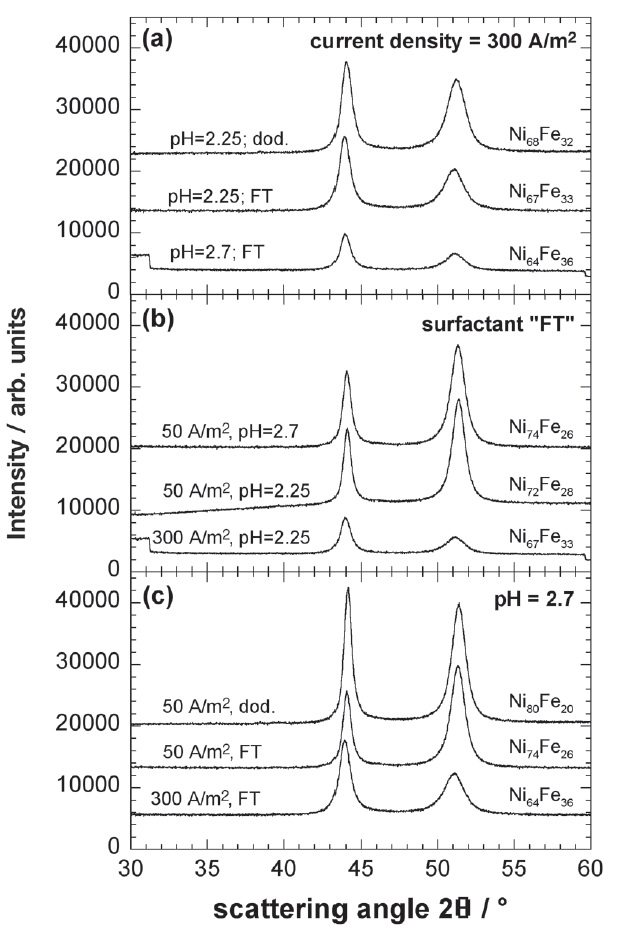
Fig. 2: XRD profiles of a series of Ni-Fe electrodepositions
As a consequence of different composition the nickeliron deposits exhibited also different lattice parameters as already mentioned. The values of the cubic lattice parameter of all nine nickel-iron deposits depicted in Figure 2 were calculated using both peaks of each XRD profile. The mean value of lattice parameter together with the standard deviation is also listed in Table 3. Comparing the composition of the deposits determined by EDX revealed that the increase in lattice parameter as a function of an increasing current density (Fig. 1) comes along with a rise in iron content. Plotting the lattice parameter of all nickel-iron deposits versus iron content illustrates the underlying linear relationship. The plot itself and the parameters of a linear regression fit are given in Figure 3. The average lattice parameter of the above-mentioned series of nickel deposits, aNi = 0.35222 nm ± 0.00003 nm, was taken as value for pure nickel in Figure 3.
Tab. 3: Lattice parameters for different compositions of deposits
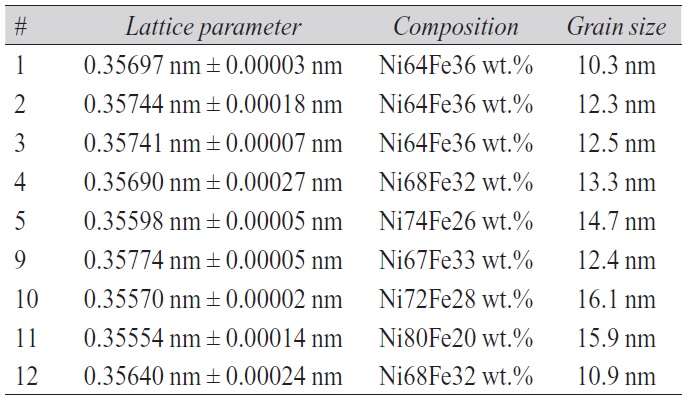
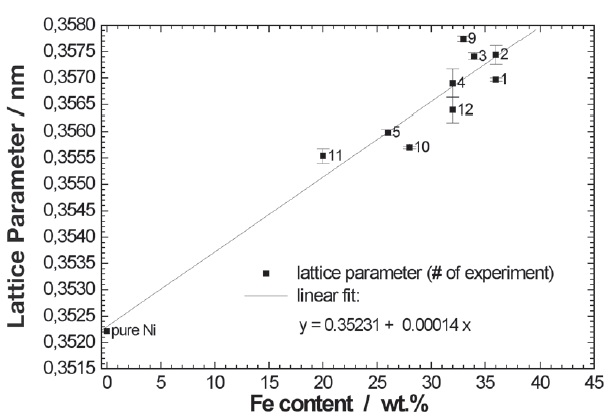
Fig. 3: Lattice parameters of a series of Ni-Fe electrodepositions as a function of iron content; linear fit A = 0.35231±0.00040, B = 0.00014±0.00001; R = 0.96598; SD = 0.00044, N = 10, P <0.0001
3.2 Thermal stability
In an earlier study, different bath compositions varied in Ni2+/Fe2+ ion ratio and surfactant were prepared and their influence on composition of the nickel-iron deposits was investigated [7]. It was found that, in the as-deposited condition, hardness of nickel-iron electrodeposits exhibited a maximum around the composition Ni80Fe20 [7] as also reported by Erb and coworkers [8]. Therefore, three compositions, namely Ni90Fe10, Ni80Fe20, and Ni70Fe30, were selected to study the hardness after thermal treatments. Since the fluorine-containing surfactant allowed the deposition of the highest iron concentration from an electrolyte with constant Ni2+/Fe2+ ion ratio [7], variation of the type of surfactant was also included in this investigation. Heat treatments for 1 h in air at various temperatures up to 550 °C were performed to test thermal stability. Thereafter, the microhardness HV0.1 was measured and plotted as a function of heat treatment temperature in Figure 4.
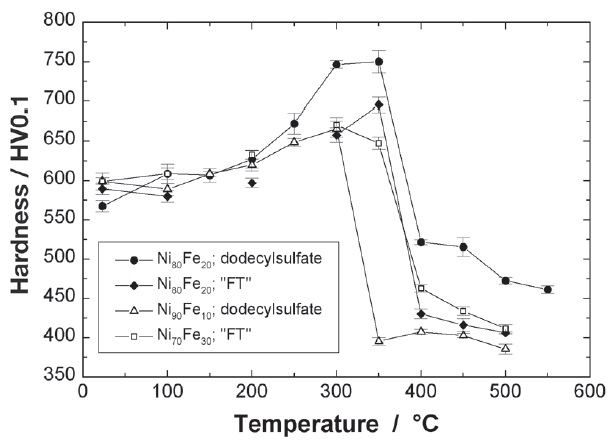
Fig. 4: Microhardness HV0.1 of different nickel-iron electrodeposits around the permalloy composition and for two different surfactants: dodecyl sulfate and a fluorinecontaining surfactant FT, after a 1h heat treatment in air for various temperatures.
Two main features can be observed in Figure 4. First of all, the hardness of all compositions covered by the present investigation is much higher than for electroplated pure nickel with HV0.1 ≈ 350 [1] even after heat treatments up to 550 °C. Secondly, there is a common feature for all four deposits studied: for temperatures up to 300 °C to 350 °C the hardness is more or less increasing before dropping within a temperature step of 50 °C. Ni80Fe20 deposited employing dodecyl sulfate as surfactant exhibits the highest hardness level of all nickel-iron electrodeposits investigated and will be the most promising candidate for the intended application.
Tab. 4: Hardness of deposits after thermal treatment
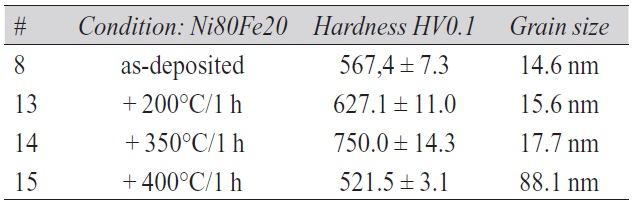
Therefore, the two nickel-iron compositions deposited applying dodecyl sulfate as surfactant, Ni80Fe20 and Ni90Fe10, were finally selected for a study of microhardness up to a temperature of 800 °C. The temperature range of heat treatments was expanded up to 800 °C since metallographic cross-section exhibited no traces of crystallization even for the highest temperatures shown in Figure 4, i.e. 500 °C and 550 °C, respectively. The electrodeposition was carried out at a bath temperature of T = 50 °C applying a current density of 50 A/m2 (0,5 A/dm2) and controlling the pH value to 2.7. Microhardness was measured parallel to the growth direction, i.e. perpendicular to the surface of the deposits, as well as perpendicular to the growth directions, i.e. in cross-section, in order to check for any difference. Both values for the two compositions selected are shown in Figure 5 as a function of heat treatment temperature (straight lines for Ni80Fe20 and dotted ones for Ni90Fe10). Error bars are omitted since the standard deviation of the hardness measurements is in most cases smaller than the size of the data symbols. It should be mentioned that heat treatments at temperatures of 550 °C and above were performed in a vacuum furnace to prevent excessive oxidation.

Fig. 5: Microhardness HV0.1 of Ni80Fe20 (squares) and Ni90Fe10 (triangles) electrodeposits as a function of heat treatment temperature; duration of the heat treatments was 1 h. Full symbols refer to hardness indentations parallel to the growth direction, open symbols refer to hardness measurements in cross-section
The hardness values determined perpendicular to the surface and in cross-section are quite close to each other for both of the nickel-iron deposits. The hardness values obtained in cross-section up to heat treatment temperatures of 800 °C clearly exhibit a two-stage behavior. After a first drop at 300 °C for Ni90Fe10 and at 350 ° for Ni80Fe20 a second decrease in hardness can be observed for both nickeliron compositions leading to a very low hardness after annealing for 1 h at 800 °C. Metallographic cross-sections viewed by optical microscopy revealed a fully crystallized microstructure with large grains after the second decrease in hardness, i.e. at 650 °C for Ni90Fe10 and at 800 °C for Ni80Fe20, respectively. Thus, massive grain growth and recrystallization dominated the development of the microstructure at high temperatures. But at a temperature corresponding to the first stage in hardness decrease no distinct the microstructure could be resolved in both nickel-iron compositions besides a few single grains (e.g. after 550 °C/1 h for Ni90Fe10) [7] indicating the onset of abnormal grain growth. Therefore, the reason for this first stage in hardness decrease of both nickel-iron deposits remains unclear after first metallographic investigation and will be treated in the following sections.
3.3 Grain size of deposits
The value of the medium grain size in the as-deposited condition is listed in Table 3 for all nine nickel-iron electrodeposits which were investigated in order to study the effects of variations of the electroplating parameters (Fig. 2, Tab. 1). The grain size of all nickel-iron deposits in the as-deposited condition lies in the range between 10 nm and 16 nm. The values for the nickel-iron deposites #1 to #5 which were produced by decreasing the current density clearly show that a decreasing current density is accompanied by an increasing grain size.

Fig. 6: Two series of optical micrographs of metallographic cross-sections of Ni90Fe10 (left column) and Ni80Fe20 (right column) for the heat treatments given in the middle
4 Discussion
Although electroplated nickel-iron alloys of a composition close to permalloy are not a new topic literature is scarce concerning information about thermal stability of those alloys. Data found in handbooks (e.g. [9]) mostly treat heat treatments as a way to render the electrodeposited nickel-iron alloys magnetically isotropic and to reduce internal stresses. Therefore, mechanical data like hardness are mainly determined in the as-deposited state and not after heat treatments. Recently, structural and also magnetic characterizations of electroplated nickel-iron alloys were presented which covered also thorough investigations of changes of texture after heat treatments, but unfortunately, no information on mechanical data was given [10, 11]. Nevertheless, influence of electroplating parameters on texture, composition, grain size and hardness will be discussed in the following as well as their thermal stability with respect to the scarce literature.
4.1 Influence of electroplating parameters
Due to the special requirements nickel-iron electrodeposits have to fulfill for applications as molding tools in microsystem technology the addition of organic additives like surfactants and saccharin cannot be avoided. Surfactants generally improve the wetting behavior of the electrolyte when pore-free electrodeposition into small and deep structures has to be achieved. To avoid microcracks of the deposits and bending of the whole molding tool the reduction of internal stresses is essential and accomplished by use of sulfur-containing additives like saccharin. Electrodeposition of iron-nickel alloys (e.g. Fe-14%Ni) from additive-free electrolyte was recently shown to be feasible but microcracks were always present [10].
Despite the fact that electrodeposition at somewhat higher temperature often results in better mechanical properties of the deposits the bath temperature was up to 50 °C. At higher temperatures of the electrolyte the patterned PMMA (polymethyl[meth]acrylate) layer would no longer be stable and thus, a reproduction of the desired microstructure with a precision on the micrometer scale could not be guaranteed.
As can be seen from Figure 2 and the corresponding data in Table 2 and Table 3 the current density exerts the strongest influence on composition and also peak ratio of the observed {111} and {200} XRD diffraction peaks of the electrodeposited nickel-iron alloys. The change of the surfactant and also a variation of the pH value from 2.7 to 2.25 do not influence the electrodeposition process that much. By reducing the current density from 300 A/m2 to 50 A/m2 the composition changed from Ni64Fe36 towards Ni74Fe26. To our knowledge such a composition variation was not reported in literature so far. On the contrary, Czerwinski [10] found that for iron-rich iron-nickel electrodeposits the current density did not significantly alter the nickel content. Since another electrolyte was used and also other plating parameters were not the same a direct comparison of both experiments seems to be inadequate.
As soon as the composition was changed by varying some of the electroplating parameters the lattice parameter was also affected. The plot of all the investigated nickel-iron electrodeposits shown in Figure 3 exhibits a linear relationship between the fcc lattice parameter and the iron content of the deposits. From the equilibrium phase diagram of the binary nickeliron system a solid solution of iron in the fcc nickel lattice is expected which directly leads to an increasing lattice parameter with increasing iron content at least up to 35 at.% [12]. Extracting graphically the lattice parameter for Ni64Fe36 (Fig. 8 in [12]) and comparing it to the value obtained from the linear regression in Figure 3 reveals a difference of only 0.1 %. Therefore, the lattice parameters of the electrodeposited nickel-iron alloys are in very good agreement with those reported for bulk binary nickel-iron alloys.
Besides the discussed changes of the lattice parameter the increasing current density led to a change in texture. In this study the texture was not explicitly studied by systematically measuring pole figures as performed by others [D10; 11]. However, already visual assessment of the results presented in Figure 2 and even more detailed comparison of evaluated {200}/ {111} peak ratios collected in Table 2 revealed the decrease of the {200} peak with increasing current density. The same change was observed for permalloy Ni-20%Fe alloys electroplated from a different electrolyte [11] but at higher current densities during deposition, i.e. 5 A/dm2 to 13 A/dm2 (corresponds to 50 A/cm2 to 130 mA/cm2 as it was originally given in [11]). In this study, lower current densities were intentionally employed because of the special demands given by the intended application for microsystem technologies, i.e. pore-free electrodeposition into small and deep structures. To meet this requirement the current density has to be extremely low in order to reduce hydrogen production during deposition as much as possible. Nevertheless, the effect of current density on the decrease of the {200} component of the texture is still present as reported for higher current densities [11].
Acknowledgement
The authors are grateful to Theo Stober and Uta Schanz for SEM studies and EDX measurements. Part of the Vickers hardness measurements were performed by Horst Zimmermann
References
- W. Bacher, K. Bade, B. Mattis, M. Saumer, R. Schwarz, Microsystem Technologies, 1998, 4, 117
- A. Fath, W. Leskopf, K. Bade, W. Bacher, Galvanotechnik, 2000, 91, 1690 (in German)
- P. Scherrer, Nachr. Ges. Wiss. Göttingen Math. Phys., 1918, K1, 98
- H. P. Klug, L. E. Alexander, X-ray Diffraction Procedures, 2nd edition (John Wiley & Sons, New York, 1974), p. 643
- B. D. Cullity, Elements of X-ray Diffraction (Addison-Wesley, Reading, 1978).
- J. V. Smith (ed.), X-ray Powder Data File, Set 1-5 (revised) (American Society for Testing Materials, Philadelphia, 1960): PDF #4-0787 for Al, Powder Diffraction File #4-0836for Cu and #4-0850 for Ni report values for the {200}/{111} ratio of 0.47, 0.46 and 0.42, respectively.
- A. Fath, W. Leskopf, K. Bade, W. Bacher, in: Microfabricated Systems and MEMS IV, P.J. Hesketh, S.S. Ang, W.E. Bailey, J.L. Davidson, H.G. Hughes, D. Misra (eds.) (Electrochemical Society, Pennington, 2000) pp. 7-16 (Proc. Electrochem. Soc. 2000-19, Phoenix, Arizona)
- C. Cheung, F. Djuanda, U. Erb, G. Palumbo, Nanostruct. Mater., 1995, 5, 513
- W. H. Safranek: The Properties of Electrodeposited Metals and Alloys (American Elsevier Publishing Comp., New York, 1974) pp. 300-308
- F. Cherwinski, Nanostruct. Mater., 1998, 10, 1363
- F. Cherwinski, J.A. Szpunar, U. Erb, J. Mater. Sci. Mater. Electron., 2000, 11, 243
- J. L. Swartzendruber, V. P. Itkin, C. B. Alcock, in: Phase Diagrams of Binary Nickel Alloys, P. Nash (ed.) (ASM International , Materials Park, 1991), pp. 110-132
PDF Version of the article |
Epub Version of the article |
Flash Version of the article |
| [qr-code size=”2″] | ||
