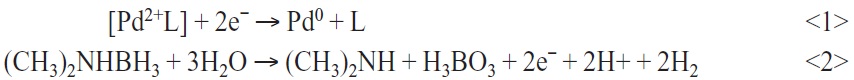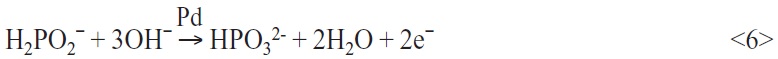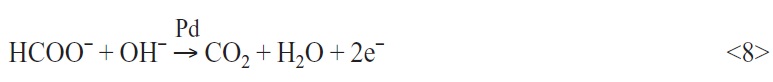Electroless copper plating processes have for some time constituted an irreplaceable part of the industrial printed circuit board manufacturing process. Without these processes, it is hard to see how through-hole or multilayer printed circuit boards might be manufactured. One main component of electroless copper plating solutions is the autocatalytic reducing agent which converts the copper from an ionic to a metallic form. Incorporation of such a reducing agent is essential to the working of the process, but brings several drawbacks with it. Many reducing agents have environmentally undesirable aspects and pose health and safety problems. At higher concentrations, they lead to undesirable side-reactions in the bath, apart from which, they can be quite expensive. Some electrolyte additives are discussed which enable copper to be deposited on a palladium-activated substrate, while allowing lower concentrations of a reducing agent in the bath to be used. Use of such an additive can bring advantages in terms of economic, environmental, and health aspects.
1 Introduction
Electroless copper plating is widely used for the fabrication of printed circuit boards and other electronic devices. The manufacture of printed circuit boards is an established technology, encompassing low cost, high yield whilst maintaining a high quality deposit. Conventional electroless copper plating solutions contain a copper salt for the delivery of the copper ions, one or several complexing agents for the copper ions, a copper ion reducing agent (usually formaldehyde), a pH adjusting agent as well as stabilizers and other additives.
In the process of electroless copper deposition, it is necessary to activate the surface of a non-conductive dielectric with palladium. The catalytic palladium deposit is used to overcome the kinetic barrier of electroless copper deposition [1]. The electroless copper plating process is initiated on the randomly distributed catalyst seeds on the substrate. This mechanism facilitates communication between electrical components and related conductors.
Though electroless copper plating will continue to have broad application potential in the future, the performances of electroless copper plating solutions still have some negative characteristics. The use of reducing agents in the electroless copper baths is necessary for achieving an autocatalytic plating process. Various common reducing agents lead to environmental and healths hazards or arevery expensive. In addition, high reducing agent concentrations can cause undesirable side reactions in the copper solution. Furthermore, other important issues need to be considered such as unstable copper baths with narrow temperature and pH windows, inefficient deposition rate and unsatisfied deposited film properties.
The aim of this study is to reduce the disadvantageous and injurious qualities of the autocatalytic reducing agents without affecting the quality of the copper deposits. This can be achieved with a low reducing agent concentration in the copper solution. To compensate for the resulting lack of reducing agent, and thus the lack of essential electrons for the copper deposition, additives are introduced. These additives support the startup process on palladium activated base material during the copper deposition.
The additional compounds operate only on the palladium and are not developed as a substitute for the traditional copper reducing agent. A standard reducing agent, such as formaldehyde, is still a fundamental requirement. This is to say, that an irreducible amount of reducing agent is necessary for the autocatalytic copper plating process.
This investigation demonstrates that additives can provide a significant benefit in terms of cost, health and environmental impact when compared to the traditional electroless copper process. In that way, a higher concentration of the mostly injurious and cost-intensive reducing agent can be avoided.
2 Reaction mechanism
Surfaces of epoxy resin substrates are easily metalized by the electroless copper plating technique. First, the base material is cleaned and etched to roughen the surface and make it hydrophilic. Thereby, a sufficient adhesion between substrate and metal is achieved. Afterwards the surface is activated with certain noble metals and brought into contact with an electroless copper bath. Then the chemical copper plating process initiates spontaneously from the catalytically activated surface.
Colloids of palladium/tin chloride or ionic palladium solutions are conventionally used as catalysts for reduction reactions of cupric ions in the presence of reducing agents.
In this study, an ionic palladium catalyst based on palladium sulphate is employed. The catalyst consists of an organic palladium complex [Pd2+L] in an alkaline solution. The palladium complex adsorbs onto the pre-treated substrate surfaces. For reducing the palladium complex to elementary, catalytic effective palladium seeds, a reduction step with dimethylamineborane ((CH3)2NHBH3) is applied. This process involves oxidation-reduction reactions:
Now metallic palladium adheres by adsorption on the surface and acts as a catalyst in electroless copper plating reactions. Electroless copper deposition reactions at palladium-catalyst-adsorbed surfaces consist of two partial reactions. At the anodic surface, the reducing agent is oxidized and at the cathodic surface, copper ions are reduced to a metallic state:
- Oxidation of the autocatalytic reducing agent (exp.: formaldehyde):
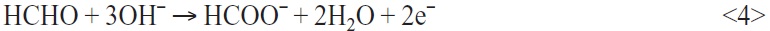
- Reduction of cupric ions:
 Both reactions occur simultaneously at the surface of the palladium catalyst. The emission of electrons is necessary for the copper reduction. Through the oxidation of the reducing agent, electrons are delivered and used for the autocatalytic copper plating process. In order to lower the disadvantageous qualities of the reducing agent, the reducing agent concentration in the copper solution has been reduced to a minimum. Slight concentrations of reducing agent cause only few electrons. The insufficient amount of electrons leads to an inhomogeneous and scant copper deposition on the base material. Therefore, in order to produce high-quality copper coatings with low reducing agent concentration, additional electrons must be provided.
Both reactions occur simultaneously at the surface of the palladium catalyst. The emission of electrons is necessary for the copper reduction. Through the oxidation of the reducing agent, electrons are delivered and used for the autocatalytic copper plating process. In order to lower the disadvantageous qualities of the reducing agent, the reducing agent concentration in the copper solution has been reduced to a minimum. Slight concentrations of reducing agent cause only few electrons. The insufficient amount of electrons leads to an inhomogeneous and scant copper deposition on the base material. Therefore, in order to produce high-quality copper coatings with low reducing agent concentration, additional electrons must be provided.
The supply of electrons for the electroless copper deposition can be increased by the application of electrolyte additives, which are able to discharge additional electrons. At the beginning of the chemical copper plating process, a sufficient supply of electrons is important for creating a defect free and uniform copper deposition despite the low concentration of the autocatalytic reducing agent. A locally inhibited deposition initialization will lead to a non homogenous deposit and thus is difficult to reverse.
For a perfect and defect free startup process of the electroless copper deposition on palladium-adsorbed surfaces, additives can be used which react with palladium. From the literature [6] the following additives were investigated: sodium hypophosphite, glycolic acid, sodium formate, and the salts of the citric acid, succinic acid and malic acid. Additional electrons are delivered by exploitation of the catalytic qualities of palladium. The electrons develop during the oxidation reaction of the electrolyte additives on the palladium seeds (exp.: hypophosphite):
The supply of additional electrons can be used for the copper reduction. Palladium interferes not directly in the plating process, but acts as an electron carrier. By use of such additions an accelerated copper deposition can be recorded. This is to say, the deposition rate is increased.
In the next steps, it should be clarified whether the accelerating reaction of the additives takes place exclusively on the palladium seeds or also on the chemically deposited copper, even if the catalytic palladium is covered with copper. Two test sequences were carried out:
- Additive without using palladium
- Variable amounts of additive and palladium
The electroless copper solution contains a diminished reducing agent concentration and, in addition, different amounts of additive 1 (sodium hypophosphite). The first experiment set-up consists of a two-electrode configuration with a working electrode and counter electrode. Both electrodes were immersed into the electroless copper solution with defined distance to each other (Fig. 1).
The working electrode and counter electrode are stainless steel. During a short, external current input (t = 1 min, I = 1 A) a thin copper layer is deposited onto the working electrode electrolytically. This allows us to exclude the influence of a palladium activated surface. After the initial electrical pre-contact, the electroless autocatalytic copper plating process keeps going on the thin copper layer. As previously stated, this method allows us to isolate the influence of the electrolyte additive in the copper solution and also on the deposit copper.
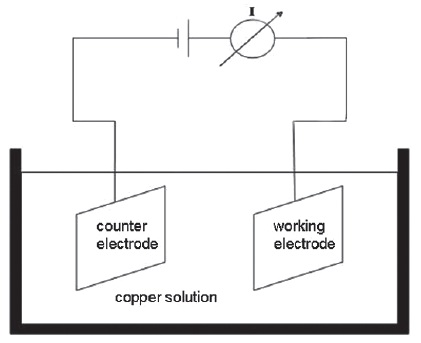
Fig. 1: Cell with two electrodes
The comparison of two copper baths with and without addition of sodium hypophosphite should show whether the expected accelerating effect takes place exclusively on the metallic palladium or also in the solution or on the plated copper (Tab. 1).
Because there is no difference in the copper deposition thickness, with or without the addition of the electrolyte additive 1 (sodium hypophosphite), the implication is that the impact of additive 1 is limited to a palladium activated surface. Without palladium activation, the additive has no influence on the copper plating process.
The second test sequence serves to characterize the reaction on the metallic palladium. Investigations were made to check the influence of the additive concentration and adsorbed palladium amount on the deposition speed. Standard epoxy resin material was activated with palladium and afterwards immersed into a copper bath. The sodium hypophosphite concentration in the electroless copper solution was varied between 0 g/L and 16 g/L. Moreover, modifications of the palladium activation process were carried out. The dwell time of the non-conductive material in the ionic palladium activator and the activator concentration were varied (Tab. 2).
Tab. 1: Comparison of copper layer thickness d with/without sodium hypophosphite

Tab. 2: Variation of the activation time and concentrations of hypophosphite (in the copper bath) and palladium (in the activator)
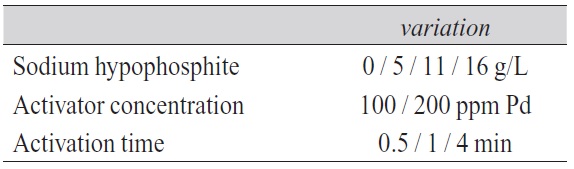
The different deposition tests indicate an increase in the deposition rate with raised concentration of sodium hypophosphite (additive 1) and high amount of adsorbed palladium on the surface (Fig. 2). This can be explained by increased emission of additional electrons which can be used for copper reduction. The palladium, with its catalytic qualities, acts as charge exchanger. It carries the electrons delivered from the additive oxidation process to the cupric ions to reduce them to metallic copper. The amplified supply of electrons leads to an enhanced copper reduction.
The maximum deposition speed is reached with a hypophosphite concentration in the electroless copper bath of 16 g/L and an activation process with an ionic activator containing 200 ppm Pd. The activation dwell time has no remarkable influence and can be between 1 and 4 minutes. A minimum copper deposition rate is experienced when the palladium concentration of the activator is reduced by 50 % to 100 ppm Pd and no additive is used.
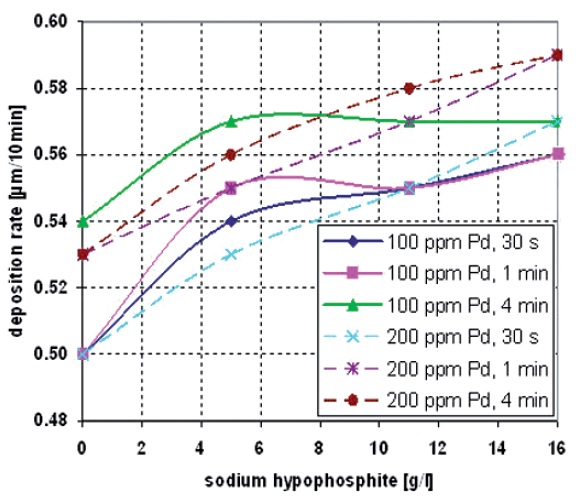
Fig. 2: The effect of sodium hypophosphite on the deposition rate at different activation conditions
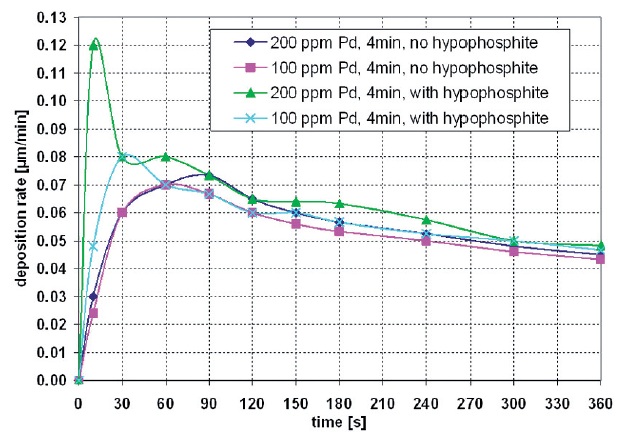
Fig. 3: Comparison of the deposition rate in copper solutions with and without hypophosphite
The comparison of the temporal change of the deposition rate in an electroless copper solution with and without sodium hypophosphite as the electrolyte additive shows some clear aspects: The maximum of deposition speed in copper solutions with an additive occurs in the initial phase of the electroless copper deposition. This peak is clearly greater than the deposition speed maximum in electrolytes without an addition (Fig. 3).
Decreasing the palladium adsorption on the surface causes a shift of the deposition rate maximum to smaller values and higher deposition times (Fig. 3, comparison between green and light blue curve).
The use of sodium hypophosphite in the copper bath stimulates a maximum deposition rate within 30 seconds. Although the maximum deposition rate is also achieved without the additive, the lag time is over 1 minute and is less dramatic or significant. The maximum in the curve shape originates from the change in copper deposition which first takes place at the catalytic active palladium seeds and then continues on the chemically deposited copper (Fig. 3, violet and deep-blue curve). The oxidation of the autocatalytic reducing agent (e.g. formaldehyde) is larger on the palladium than on the chemically deposited copper. Palladium is a better catalyst for the copper reduction than the copper itself. Thus the deposition rate decreases when all surfaces of the palladium are covered with deposited copper metal.
The curve shapes of the electroless copper solution with and without the sodium hypophosphite enhancement become nearly identical after a plating time of 90 seconds. After this, hypophosphite has no more influence on the electroless copper plating process.
In general, it can be assumed that a maximum deposition rate is achieved when a maximum activation and exposure of palladium is present. The use of an electrolyte additive (sodium hypophosphite) in conjunction with a high, homogenous concentration of palladium seeds on the surface can further enhance the deposition performance on the related substrate. The additives could be referred to as startup agents because they only react in the initial phase of the electroless copper deposition. At the beginning of the chemical plating process palladium seeds are active and affect the additive oxidation. The raised supply of electrons can subsequently induce an accelerated deposition.
3 Electrolyte additives
In order to characterize the reactivity on the palladium and to quantify the different electrolyte additives available, a quick test method was carried out [5]. The different additives were added to an aqueous palladium sulphate solution with a defined amount of palladium ions (1 g/L Pd). For checking the reactivity on palladium the colour change of the palladium sulphate solution after the addition is assessed visually. If the addition of an additive leads to color change from orange (source state) to black, metallic palladium precipitates (Fig. 4). The additive has an oxidizing potential and palladium ions can be reduced to metallic palladium.
If the original colour state of the palladium solution remains orange after addition of the chemical additive, no reactivity on the palladium is given. The palladium ions cannot be reduced by the additive and therefore remain in solution.
With addition of sodium hypophosphite, glycolic acid or sodium formate a color change of the palladium sulphate solution from orange to black could be detected. This is a visual indication that the additive is being oxidized and electrons are released. These electrons reduce the palladium ions contained in the orange solution. The resultant metallic palladium precipitates as a black solid. These additives are a potentially suitable reducing agent for palladium.

Fig. 4: Test to prove the reactivity on palladium: 1) Palladium sulphate solution in the source state (without addition); 2) Palladium sulphate solution with addition of sodium hypophosphite; 3) Palladium sulphate solution with addition of sodium formate; 4) Palladium sulphate solution with addition of glycolic acid
With help of the time interval required for this precipitation to occur, a general categorization can be made for the relative suitability of the reduction properties of the additives. This results in the following classification of the additives with regard to their reactivity strength on palladium: Sodium hypophosphite > Sodium formate > Glycolic acid.
The individual addition of the other substances under investigation (citric acid, succinic acid, malic acid) causes no colour change of the palladium sulphate solution. The solution remains in the origin state (orange). These additives cannot reduce palladium ions at temperatures up to 80 °C.
3.1 Sodium hypophosphite
The addition of sodium hypophosphite to the palladium sulphate, even at ambient temperatures, leads to the immediate formation of the black precipitation of finely distributed, metallic palladium. After a few seconds, sodium hypophosphite is oxidized. The generated electrons can be used for the reduction of the palladium ions.
Sodium hypophosphite is often used in electroless palladium electrolytes as the reducing agent. Also some electroless copper baths work on the basis of hypophosphite as a reducing agent. But this is disadvantageous because hypophosphite is not suitable for the autocatalytic copper reduction. Copper is not a good catalyst for the oxidation of hypophosphite.
After a thin deposited copper layer on the palladium activated substrate, the electroless copper plating process stops. Therefore, nickel or cobalt ions are added to the copper electrolyte to allow an autocatalytic copper plating process with hypophosphite [2, 3]. These metal ions are a much better catalyst for the hypophosphite oxidation.

Fig. 5: Initial phase of the electroless copper plating process on palladium activated basic material without addition of hypophosphite (on the left) and with hypophosphite (on the right)
While hypophosphite does not exhibit a reductive action on copper, it exhibits a highly reductive action on a catalyst metal such as palladium. It is effective at raising the initial plating reactivity via a catalyst metal. Since deposition uniformity at the start of plating is higher. The copper deposition starts homogeneous on the whole activated base material (Fig. 5).
Already after 1 minute of the copper plating process there is a clear difference in the copper distribution on the material. Although the concentration of reducing agent in the copper solution is very low (c < 0.05 mol/L), the copper deposition on the substrate is improved by the addition of hypophosphite (Fig. 5, right). Without additive and with a low concentration of reducing agent, the copper deposition is neither sufficient nor homogenous from the beginning (Fig. 5, left). After the initialization, the hypophosphite plays no further role in the subsequent autocatalytic process.
With the concentration of hypophosphite in the copper bath the copper deposition rate rises (Fig. 6). Similarly, the copper deposition quality is improved on the surface as well as in mechanically created physical structures (e.g. drilled holes in the printed circuit board, Fig. 7).
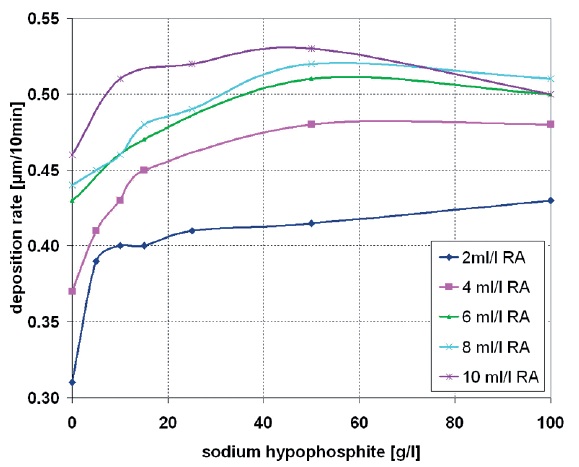
Fig. 6: Variation of the hypophosphite concentration in the copper bath and its effect on the deposition rate by simultaneous change of the reducing agent amount (RA)
The impact of the hypophosphite concentration on the copper deposition rate is immediate. The greatest increase in deposition rate is visible at lower hypophosphite concentrations. At a concentration of approx. 16 g/L hypophosphite, the optimum acceleration referring to copper coverage, bath stability and performance is achieved. This does not however correspond with the maximum value of the deposition rate. An increase of hypophosphite concentration above the optimum value causes no other positive effect. The deposition rate increases only relatively minimally and the copper deposition becomes qualitatively bad due to large amounts of electrolyte additives in the copper solution.
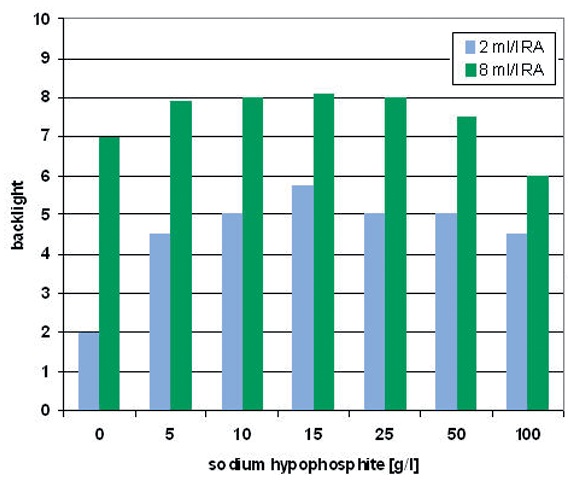
Fig. 7: Variation of the hypophosphite concentration and its effect on the drilled hole covering (backlight, 1: badly, 10: very well) with different amounts of reducing agent (RA)
The supporting effect of the electrolyte additive is most effective if the concentration of the copper reducing agent in the electrolyte is low (for example: formaldehyde). Figure 6 shows the biggest increase in deposition rate when the slightest reducing agent amount of 2 ml/L is used. With rising amount of reducing agent, the positive effect of the hypophosphite is overlaid by the autocatalytic deposition caused by the reducing agent. Too high hypophosphite amounts in the copper bath (from 50 g/L hypophosphite on) lead again to the decrease of the deposition speed (Fig. 6) and degradation in coverage of the drilled holes (backlight, Fig. 7).
As shown in Figure 7, an optimum copper coverage is to be observed with approximately 15 g/L to 20 g/L sodium hypophosphite. The comparison between low and high reducing agent concentration (2 ml/L and 8 ml/L) makes clear that with low reducing agent concentration, the effect of the additive (hypophosphite) is substantially stronger than with high reducing agent amount. In case of using electrolytes with 2ml/L reducing agent concentration, the degree of copper coverage in the drilled holes rises more than twice when the amount of hypophosphite is increased from 0 g/L to 15 g/L.
3.2 Glycolic acid
Beside sodium hypophosphite the copper deposition is also improved by using glycolic acid as a kick-starting agent. The copper deposition speed and the homogeneity of the copper coverage improve. The reaction mechanism is the same as with sodium hypophosphite. Metallic palladium which is adsorbed on the substrate surface by the activation process acts as a catalyst for the oxidation of glycolic acid and leads to an additive oxidation (exp.: glycolic acid):
Glycolic acid is oxidized in the presence of palladium. The released electrons can be used additionally in the electroless copper electrolyte to reduce copper ions. The copper plating process begins uniformly. As shown in Table 3, there is also an optimum of the glycolic acid concentration in the copper bath as already seen with copper solutions containing hypophosphite. A good bath performance (startup behavior, deposition rate, copper coverage) and stability is achieved with 18 g/L glycolic acid.
Tab. 3: Dependence of the deposition rate ν on the glycolic acid concentration with different amounts of reducing agent (RA)
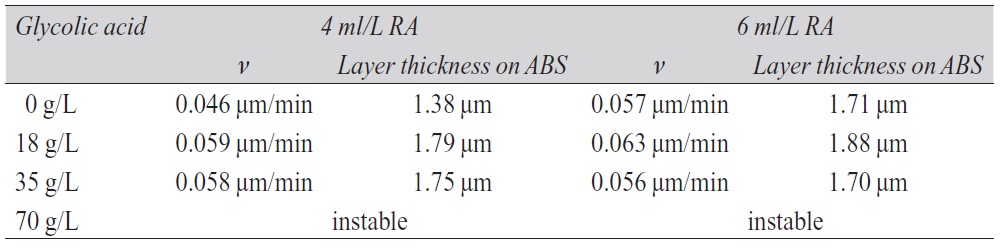
Raising the glycolic acid amount up to 18 g/L leads to an increase of the copper layer thickness. Using concentrations above 18 g/L, the deposition rate decreases and the electroless copper solution becomes unstable. Nevertheless, in contrast to copper baths with hypophosphite, electrolytes with glycolic acid in comparable concentrations (to approx. 20 g/L) are more stable during the electroless copper deposition. Raising the amount of glycolic acid in the bath up to 70 g/L or more has a negative impact on the bath stability. This instability can occur even before the process of the autocatalytic copper deposition begins on the substrate (Fig. 8, right).

Fig. 8: Electroless copper baths with glycolic acid (Left: 18 g/L glycolic acid → stable solution; Right: 70 g/L glycolic acid → unstable)
A combination of the additives sodium hypophosphite and glycolic acid in the copper bath leads to no improvement in the electroless copper deposition. Rather, the combination reduces even slightly the deposition speed and lowers the electrolyte stability.
3.3 Sodium formate
Another candidate which can support the start process of the autocatalytic copper formation as an additional electron source is sodium formate. Sodium formate is oxidized like sodium hypophosphite and glycolic acid in the presence of palladium. Hence, the sodium salt of the methane acid also often finds use as a reducing agent in chemical palladium electrolytes. Sodium formate delivers electrons by the oxidation reaction. The electrons can be used for the palladium ions reduction in the electroless palladium electrolyte. Reaction <8> shows the additive oxidation (exp.: sodium formate):
Electroless copper solutions that contain sodium formate as a reducing agent are to be taken from the literature [4] only sporadically and up to now also with nearly no success. This can be due to the fact that sodium formate is a very weak reducing agent for copper. Hence, very high activation energy is necessary to run the electroless copper plating process. If sodium formate is used as an addition in a copper electrolyte which already contains another, well functioning and autocatalytic active reducing agent, an accelerating effect can be observed during the copper deposition. The electrons that are additionally supplied by the sodium formate oxidation are used in the electroless copper plating process. With the help of sodium formate as an additive an increase of the copper layer thickness of about 25 % is registered after a deposition time of 10 minutes (Tab. 4).
Tab. 4: Comparison of the layer thickness with/without sodium formate

In electroless copper solution with formaldehyde as reducing agent, sodium formate accumulates by a side reaction (Cannizzaro reaction) as a reaction product:
Most electroless copper baths with formaldehyde show an increase of deposition speed with increasing bath age. A raised bath age is combined with a raise of the sodium formate concentration. So it seems as if sodium formate has a supporting and accelerating effect on deposition speed due to the delivery of additional electrons in the initial phase of copper plating.
Electroless copper solutions which contain sodium hypophosphite, glycolic acid or sodium formate as an additive allow a homogeneous copper deposition with low concentration of the autocatalytic working reducing agent on account of an additional electron-delivering source. The electrolyte additives work on a reduction-oxidation basis. By utilization of the catalytic qualities of palladium, they can be oxidized in the electroless copper solution.
3.4 Various additives
The other additives tested include the salts of citric acid, succinic acid and malic acid. These additives show only a very low positive supporting effect in the initial phase of the electroless copper deposition. They cause no reduction-oxidation reactions at the metallic palladium seeds. But they can form complexes with the copper ions dissolved in the copper bath. Citric acid, succinic acid and malic acid are complexing agents in electroless copper electrolytes. On account of different complex stabilities these copper complexes are easier to reduce than other complexes as for example EDTA complexes. Thereby, they can cause a slight acceleration effect of the copper deposition.
The kick-starting effect of these additives on the electroless copper deposition is very small. For this reason, these electrolyte additives represent no real alternative to the chemical additives that work on reduction-oxidation basis (sodium hypophosphite, glycolic acid, sodium formate).
4 Conclusions
In the initial phase of the electroless copper deposition, the palladium seeds adsorbed by the activation process of the non conductive substrate serve as an anode for the oxidation of the reducing agent. The electrons that are supplied by the oxidation reaction pass through the palladium and cause the copper reduction.
Electroless copper nucleates around the limited number of anodic palladium sites. The copper deposition is an autocatalytic process and copper continues to nucleate on other nucleated copper grains. As a result, the growth of the electroless deposited copper takes place in both lateral and vertical directions. If the reducing agent used in the copper bath does not work autocatalytically on copper, the copper plating process stops as soon as the palladium is covered with copper. Such deposits are self limiting with regard to deposition thickness.
In electroless copper solutions, the reducing agent is necessary to maintain the autocatalytic copper plating process. If the concentration of reducing agent is reduced in order to minimize injurious qualities and side reactions, the amount of delivered electrons by the oxidation of the reducing agent is not sufficient to generate a compact copper layer. Therefore, a high degree of copper coverage of the layer can not be achieved, especially at the beginning of the plating process. This leads to an inhomogeneous copper deposition. By the additional use of a chemical substance (electrolyte additive) which can be easily oxidized at a palladium surface, more electrons can be created. The amount of electrons consists of the electrons that are formed by the autocatalytic copper reducing as well as by the electrolyte additive. By that, the electrons coming from two sources permit, in sufficient amount, the deposition of a homogeneous and compact copper layer. Suitable additives are sodium hypophosphite, glycolic acid and sodium formate. They can be oxidized at catalytic active palladium and provide additional electrons for the electroless copper deposition. However, the additives are no reducing agents for the autocatalytic copper plating process.
The autocatalytic reducing agent has to be in the copper solution and cannot be substituted completely. The additives react exclusively with the palladium. Therefore, they can be considered as a sort of startup agent for the copper plating process on palladium activated base material. Palladium is not directly involved in the reduction-oxidation reactions, but works intermediary by its catalytic qualities as an electron carrier.
In the course of copper plating the palladium seeds are covered with electroless deposited copper after a short time and are no longer catalytically active. Then, the supporting effect of the used electrolyte additives is not given any more. Without palladium additional generated electrons are not available. In that case, the autocatalytic copper deposition continues steadily only due to the reducing agent in the electrolyte (e.g. formaldehyde).
The use of additives, to provide a chemical kick start, may provide the following benefits as there are indications that less traditional reduction chemistry is necessary to support a successful electroless plating mechanism: Lower health risks, lower costs due to non process relevant side reactions (e.g. Cannizzaro effect) and a better quality of copper deposition. By enhancing the plating reactivity in the initial phase, plating with lower concentrations of the reducing agent is realized.
References
- P. P. Lau, C. C. Wong, L. Chan; Improving electroless Cu via filling with optimised Pd activation; Applied Surface Science 253 (2006), p. 2357-2361
- J. G.Gaudiello, G. L. Ballard; Mechanistic insights into metal-mediated electroless copper plating employing hypophosphite as a reducing agent; IBM J. Res. Develop. Vol. 37 No. 2; 1993
- J. Li, P. A. Kohl; The Deposition Characteristics of Accelerated Nonformaldehyde Electroless Copper Plating; Journal of the Electrochem. Soc. 150 (2003)8, C558-C562
- L. Stein; Formaldehydfreies Bad zur stromlosen Abscheidung von Kupfer und Verfahren unter Verwendung dieses Bades; patent DE 4111558 C1 ; 1992
- L. Schoenberg; The Use of Organic Additives to Stabilize and Enhance the Deposition Rate of Electroless Copper Plating; Journal of the Electrochem. Soc. Vol. 119 (1972) No.11, p 1491-1493
- S. Chowdhury, M. Bauer; Electroless Plating Baths for High Aspect Features; patent US 7220296 B1; 2007
PDF Version of the article |
Epub Version of the article |
Flash Version of the article |
| [qr-code size=”2″] | ||