Oxidation-reduction reactions also known as redox reactions for a shorthand term are typified by any chemical process that involves transfer of electrons between two species. The ubiquitous nature of redox reactions occurs over diverse systems ranging from industrial processes to chemical reactions encountered in everyday basic life functions. Based primarily on electrochemistry, the present article reviews the science of redox reactions with applications in some specific industrial and real life cases.
1 Introduction
In the realm of chemical arrangements, a novel reaction system can occur where both oxidation and reduction reactions occur simultaneously termed as redox process. Such a system may sound dangerous but it operates in many industrial and real life situations. The binary concept of chemical reaction process of present interest, involves simultaneous oxidation and reduction mechanisms utilizing oxidizing and reducing agents. In simple terms, reduction is promoted via donation of electrons by the reducing agent which changes its ionic form during the process to an oxidized state. In contrast, an oxidizing agent removes electrons from a system to promote oxidation whereby it is reduced, for example, from ionic to metallic state.
Electrochemistry plays an important part in the present context. Accordingly, oxidation (anodic) and reduction (cathodic) reactions spatially separated from each other, undergo different electrochemical reactions. Ions are allowed for charges to carry between the anode and cathode immersed in an electrolyte. The reaction mechanism allows electrons to move around an external circuit to assure electroneutrality. Primarily based on the principles of electrochemistry, a comprehensive account is presented of a remarkable phenomenon of simultaneous occurrence of oxidation and reduction reactions in several interesting industrial cases as well as some mundane domestic situations.
2 Historical background of the redox concept
Of the various chemical processes involving redox
reactions, combustion was the earliest focus of philosophical and scientific attention. A German chemist G. E. Stahl articulated a Phlogiston theory first in 1697 when he noted that matter releases an elementary constituent called phlogiston during combustion. The burning of charcoal, for example, was interpreted as the loss of phlogiston from carbon to the air i.e. charcoal (phlogiston) → ash where charcoal is presumed to be a compound. A notable consequence of the phlogiston theory was the notion that a compound, for example, an oxide of a metal such as mercury oxide (HgO) was apparently a chemically simpler substance than the metal itself which can be obtained only from the oxide by the addition of phlogiston (oxide + phlogiston → metal). The phlogiston theory despite enjoying scientific popularity in more modern times, could not, however, provide any acceptable explanation of the gain in weight when an oxide is formed from a metal presumably with the addition of phlogiston.
Late in the 18th century, an interrelated work between English chemist Joseph Priestley and French chemist Antoine-Laurent Lavoisier led to overthrow the phlogiston theory. Lavoisier saw Priestley’s discovery of oxygen in 1774 as the key to the weight gain known to accompany the burning of sulphur and phosphorus and the calcinations of metals (forming oxide). In his Traité élémentaire de chimie, he clearly established that combustion consists of a chemical combination between oxygen from the atmosphere and combustible matter. By the end of the century, his ideas were widely accepted and had been successfully applied to the more complex processes such as respiration and photosynthesis. Reactions in which oxygen was consumed were classified as oxidation, while those in which oxygen was lost were termed reduction.
3 Principle of redox reaction
Background
The law of conservation of charge stipulates that if one species loses electrons, there must be another species that gains them. The term oxidation refers to a process that loses electrons – this definition may appear tricky because oxidation sounds like it might only involve oxygen. In truth, oxygen is undoubtedly an important element in many oxidation-reduction reactions by virtue of its large electro negativity which causes many other elements to give up its electrons. Since oxygen often „forces“ other elements to lose electrons, the process of losing electrons became known as oxidation. Likewise, reduction is also a troubling word, since it refers to a process that gains electrons. In this case, the word „reduce“ has nothing to do with the quantity of electrons, but rather the change in charge. Since the addition of electrons will add more negative charges to a species i.e. reduce the number of positive charges, it is in its charged form considered to be reduced.
Most oxidation-reduction (redox) processes involve the transfer of oxygen atoms, hydrogen atoms or electrons – all these three involvements share two important characteristics:
- they are coupled i.e., in any oxidation reaction, a reciprocal reaction namely reduction occurs, and
- they involve a characteristic net chemical change – i.e., an atom or electron goes from one unit of matter to another.
Both reciprocity and net change are fundamental aspects of redox reactions.
Basic definitions
There are three ways one can define redox reactions:
- A simpler non-electrochemical definition would be to consider cases of loosing or gaining oxygen such as in the production of pure iron from iron oxide in a blast furnace:
3 C + 2 Fe2 O3 → 4 Fe + 3 CO2
In this reaction, iron oxide (Fe2 O3 ) gives away its oxygen to carbon (C). In chemical terms, the carbon is said to be oxidized because it has gained oxygen. At the same time, the iron oxide is said to be reduced because it has lost oxygen. Iron oxide here is called an oxidizing agent by virtue of its ability to give away oxygen. Carbon, on the other hand, is acted as a reducing agent because of its ability to take on oxygen. Thus a redox reaction system involves simultaneous occurrence of oxidation and reduction where one substance gives away oxygen (reduction) to a second substance which must be present to take on that oxygen (oxidation). - An electrochemical definition would involve ionic equations where reduction occurs via gain of electrons while oxidation is characterised by loss of electrons. In the following example of a redox reaction in an electrochemical cel, both copper(II) oxide and magnesium (II) oxide are ionic, while the metals with zero valencies are obviously not:
CuO + Mg0 → Cu0 + MgO
Besides involving a simple oxygen transfer, the above reaction can be re-written in terms of ionic equation as
Cu 2+ +2 e → Cu0 and Mg 0 – 2 e → Mg 2+
with the overall reaction as
Cu 2+ + Mg0 → Cu0 + Mg 2+
where Cu 2+ ion is reduced with gain of electrons, while Mg0 in its metallic state is oxidised with loss of electrons. Thus a chemical substance undergoes oxidation by taking on oxygen or loosing electrons, but gives off oxygen or takes on electrons in reduction as schematically shown. - A third interesting definition of redox reactions may not be too obvious from the outset, because it does not necessarily involve any oxygen. It encompasses some degree of electrochemistry. In this context, two elements reacting with each other, involve exchange of electrons only. For such redox reactions, the gain (reduction) and loss (oxidation) of electrons are more important than the gain and loss of oxygen. For example, sodium metal (Na) reacts with chlorine gas [Cl2(g)] whereby sodium atoms lose one electron each to chlorine atoms:
2 Na + Cl 2 → 2 NaCl
Sodium is considered to be oxidized because it loses electrons, while chlorine is said to be reduced by gaining electrons in the reaction.
Assigning oxidation numbers
The oxidation number (also called oxidation state) of a participating chemical species (molecule, atom, or ion) changes by gaining electrons (reduction process) or donating electrons (oxidation process) to form a chemical bond with another species/atom. Each atom that participates in a redox reaction is assigned with an oxidation number that reflects its ability to acquire, donate, or share electrons. These numbers represent formal but not necessarily the real charges which can be much lower for individual elements that make up an ion or a compound. The sum of all the individual oxidation numbers must be equal to the charge of the ion, making it zero if it is a compound. For example, an iron ion such as Fe3+ has an oxidation number of +3 because it misses three electrons to acquire its elemental state to form a chemical bond. In contrast, an oxygen ion O2− has an oxidation number of −2 because it can donate two electrons. In an electronically neutral substance, the sum of the oxidation numbers is zero. Thus, for example, in haematite (Fe2O3) the sum of oxidation number of the two iron atoms (+6 in totals) balances the sum of negative oxidation number of the three oxygen atoms (−6 in total).
4 Application of redox reactions in industry
4.1 Metallurgical extraction of metals from ores
Background
Metals are rarely found free in nature, but occur in ores mostly in their oxidized form which must be reduced to the metallic state for normal usage when the oxidation number becomes zero. The method of extraction of a metal from an ore depends on its reactivity. Some metals are easily reduced. For example, a less-reactive metal such as mercury can be produced from a mercury sulphide ore simply by heating the ore in air. On the other hand, copper from chalcopyrite ore (CuFeS2) or iron from its oxide ore (haematite, Fe2O3) is produced by pyrometallurgical reduction technique. In the case of iron, the oxide ore is reduced by heating with coke (impure carbon) and oxygen. The coke reduces the iron in the ore to the metallic state. It is actually the CO gas which is in equilibrium with CO2 gas and C (Boudouard reaction):
C + CO2 → 2CO
Such reaction runs quite fast as one of the reactant is a gas. More about the pyrometallurgical reductions of iron and copper ores are described later on.
There are other metals which are highly reactive such as aluminium that are difficult to reduce, and thus cannot be extracted using pyrometallurgical reduction technique. In the case of aluminium, corrosion/oxidation will occur as long as oxygen is available which would reverse the reduction of aluminium during the application of a reduction process. Such a metal can only be extracted from its ore after electrons are pumped into their ores using electricity i.e. a technique based on electrolysis. Redox reactions involved are described below. It is worth mentioning that even the electrolytic method would not necessarily be useful for extraction of all metals. Examples include noble metals such as gold and platinum which are most resistant to electrochemical corrosion during electrolysis due to their high standard reduction potentials. It is worth pointing out that these noble metals do already occur in their elemental state in nature because of their characteristic corrosion resistance.
4.1.1 Pyrometallurgical process
1) Iron from haematite oxide ore (Fe2O3)
Iron is produced via reduction of its ore by heating it with coke (impure carbon) which acts the reducing agent in a blast furnace. The coke reduces the ore while itself undergoing oxidation during the process:
Fe2O3 (s) + 3C(s) → 2Fe (s) + 3CO (g)
2) Copper from chalcopyrite ore (CuFeS2)
Chalcopyrite /copper pyrite ores typically contain low percentages of copper and thus have to be first concentrated, for example, by froth flotation before refining. The concentrated ore is next exposed to strong heating in a furnace with silicon dioxide (silica), calcium carbonate and air or oxygen. The copper(II) ions in the chalcopyrite are reduced to copper(I) sulphide which is further reduced to copper metal in the final stage. The iron in the chalcopyrite ends up as iron(II) silicate slag (FeSiO3) which is removed. The overall redox reactions can be written as:
2CuFeS2 + 2 SiO2 + 4O2 → Cu2S + 2 FeSiO3 + 3SO2
Copper(I) sulphide thus produced is converted to copper with a final blast of air:
Cu2S + O2 → 2Cu + SO2
Most of the sulphur in the chalcopyrite is oxidised to sulphur dioxide gas which is used to make sulphuric acid via the Contact Process. In contrast, copper is reduced from Cu 2+ to metallic Cu 0.
4.1.2 Thermal heating method
Most of the world‘s mercury is obtained from its sulphide (HgS) ore called cinnabar or vermillion by heating the ore in a current of air and condensing the mercury vapour formed. Due to its relatively low boiling point, mercury can be easily purified by vacuum distillation. The redox reaction of mercury sulphide with oxygen would be as follows where mercury and sulphur undergo reduction and oxidation respectively:
Hg 2+S 2- + O2 → Hg0 + SO2
4.1.3 Cementation process
This is a heterogeneous precipitation process in which ions are reduced to zero oxidation states at a solid metallic interface. The process is often used to refine leach solutions. Cementation of copper is a common example. Copper ions often from an ore leaching process, are precipitated out in the presence of solid iron. Iron oxidizes while the copper ions are reduced through the transfer of electrons. The reaction is spontaneous because copper is higher on the galvanic series than iron.
Cu2+(aq) + Fe(s) → Cu(s) + Fe2+(aq)
This was a historically useful process for the production of copper, where the precipitated solid copper metal was recovered as flakes or powder on the surface of scrap iron. One can refer to the unwanted cementation process which occurs while trying to electroplate copper on low alloy iron alloys from an acidic electrolyte containing Cu 2+ ions.
4.1.4 Electrolytic method
Background
There are metals for which reduction of the corresponding ores by any of the above methods, would not only be very expensive but would find it difficult to operate efficiently. In such a case, extraction involves pumping electrons into their ores using electricity. Aluminium once a precious metal proved to be extremely difficult to separate it from oxygen. Unlike rust on iron discussed below, aluminium with high affinity for oxygen oxidizes quickly forming a strongly adherent coating of aluminium oxide (Al2 O3) on the substrate. Thus aluminium cannot be extracted by pyrometallurgical method from its oxide ore (bauxite) because oxidation will act to reverse the metallurgical reduction to the metallic state. Also, unlike iron extraction process, carbon cannot be used as a reducer to produce aluminium from its oxide ore because aluminium is too reactive to carbon. Thus in the case of aluminum, the electrolytic technique is the only option.
Aluminium
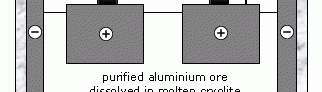
In the Hall-Heroult process of electrolysis, the use of electric current produces a chemical change by reducing Al 3+ ions to neutral aluminium atoms and thereby overcaming the problem for extraction of aluminium from its ore.
Initially aluminium ore (bauxite) is purified to yield a white powder (aluminium oxide) from which aluminium is extracted. The extraction is done by electrolysis for which aluminium oxide must be in molten stage to allow electricity to pass through. But such a process involving melting would be expensive because aluminium oxide has a very high melting point (over 2,000 °C). Thus it is first dissolved in molten cryolite which is an aluminium compound with a lower melting point than aluminium oxide. The use of cryolite reduces some of the energy costs involved in the extraction of aluminium.
In the above diagram, both the negative (cathode) and positive (anode) electrodes are made of graphite (a form of carbon). Aluminium metal is formed at the negative electrode and sinks to the bottom of the tank, where it is tapped off, while oxygen is evolved at the positive electrode. This oxygen reacts with the carbon of the positive electrodes, forming carbon dioxide, and they gradually burn away. Consequently, the positive electrodes have to be replaced frequently, which, however, adds to the cost of the process. The process involves redox reactions with metal deposition at the cathode (-ve) terminal via reduction process while an oxidation reaction takes place at the anode (+ve) terminal.
In the United States today, 4.5% of the total electricity output is used for the production of aluminium via electrolysis. The foregoing statistic is staggering, considering just how much electricity Americans use, and it indicates the importance of this once-precious Electrochemical cell for aluminium extraction by fused salt electrolysis metal. Thus although aluminium is the most abundant metal on Earth, it is expensive largely because of the amount of electricity used up in the extraction process.
Besides aluminium, there are other metals such as, lithium, sodium, potassium, magnesium, calcium, etc. which are produced commercially by the electrolytic methods. Also, many chemicals of our daily needs such as caustic soda, chlorine, fluorine, etc., are produced by electrolysis based on redox reactions.
4.2 High pressure technology
It is important to point out that oxygen is not an essential prerequisite for oxidation-reduction reactions. For example, a commonly produced chemical namely ammonia (NH3) does not contain oxygen. To produce ammonia by a redox reaction (Haber process), nitrogen and hydrogen are combined together under pressure about 200 atmospheres (200 times normal pressure) at a temperature around 932°F (500°C) using iron as catalyst. The nitrogen is oxidized and the hydrogen is reduced. The redox reaction in the Haber process can be summarised as follows:
nitrogen + hydrogen ⇆ ammonia, or
N2 + 3H2 ⇆ 2 NH3
The resulting ammonia, of course, can be used to make fertilizers, dyes, explosives, cleaning solutions, and polymers.
4.3 Corrosion/rusting of metals
Rusting of iron
Most metals react with oxygen to form compounds known as oxides. Rust is the name generally given to the oxide of iron. The process by which rusting occurs is also known as corrosion by exposure to oxidizing agents – primarily oxygen itself. Most metals react with oxygen and might corrode so quickly that they become useless. This is because the oxide tends to flake off and thereby exposing fresh surfaces to further oxidation. Every year, businesses and governments devote millions of pounds to protecting iron and steel from oxidation by means of painting and other measures, such as galvanizing with zinc. In fact, oxidation-reduction reactions virtually define the existence of the world of iron.
When water comes into contact with, for example, an iron pipe, some of the oxygen in the water oxidizes iron to the ionic state, yielding free hydrogen ions. The iron ions combine further with oxygen of the ambient air to form water, and the process starts again at the oxidation-of-iron step. As a result, there is an increasing amount of iron in a more oxidized state that would thus carry more and more positive charge. These iron atoms combine with hydroxyl groups which are negatively charged oxygen-hydrogen pairs (OH1-) to form hydroxy compounds such as iron(II) hydroxide [Fe(OH)2] and iron(III) hydroxide [Fe(OH)3]. With some drying, what ultimately remains is hydrated iron (III) oxide:
Fe + O2 (from air) + H2O →
Hydrated iron (III) oxide, Fe2O3. XH2O and also
FeO(OH)
The result is the familiar orange-brown rust seen on rusty iron and steel objects such as a rusty bike left outside in damp weather too many times. As a result, the iron in the metal parts has rusted.
Iron rusting is an example of a redox reaction whereby iron reacts with the oxygen of the air to form rust and can be explained in terms of transfer of electrons. During the initial stages of rusting, iron is oxidised by loosing electrons:
Fe – 2e– → Fe2+
while oxygen is reduced by gaining electrons:
1/2O2 + 2e– + H2O → 2OH–
Combining the above two reactions, one obtains reaction for initiation of rust:
Fe + 1/2O2 + H2O → Fe2+ + 2OH– (rust)
There are also some everyday examples of redox reactions involving atmospheric oxygen. One can highlight examples of tarnishing of silver wares and formation of bluish-green encrustation called verdigris patina (derived from the French for green grey) on copper objects.
Tarnishing of coinage metals
Silver and copper are termed as „coinage metals“, because they have often been used to mint coins. Both these metals respond to oxidation by corroding in a different way by changing colour compared to the rusting of iron. The surface of a silver cup/goblet if not polished regularly, would tarnish via oxidation by forming silver sulphide, Ag2S on the surface. This tarnishing phenomenon on silver ware occurs because of a chemical reaction between silver and sulphur-containing chemicals in the air. Metallic silver is oxidised to ionic state while oxygen is reduced by combining with hydrogen as shown below illustrating redox reactions:
4 Ag0 + 2H2S (g) + O2 → 2Ag2S (tarnish) + 2H2O (l)
In case of copper, verdigris patina is formed during exposure to atmospheric elements such as oxygen, carbon dioxide, rain, acid rain, sulphur-bearing compounds etc. A blue green colour is developed which is mainly copper hydroxide Cu(OH)2 and copper carbonate, CuCO3. It is typically formed when copper reacts with water vapour, oxygen and carbon dioxide of the air:
2Cu(s) + H2O(g)+CO2(g)+O2(g) →
Cu(OH)2(s) +Cu(CO)3 (s)
4.4 Commercial electrochemical process
4.4.1 Batteries
Electrochemistry by definition is the study of the relationship between chemical and electrical energy. An important application of the concept of oxidationreduction reactions lies in the development of electrochemical cells (these types of cells should not be confused with biological cells; the word cell comes from cella which is a Latin word for chamber or small room). In an electrochemical cell as described above for the Hall-Heroult process, the oxidation reaction is physically separated from the reduction reaction. Electrons for the reactions pass via electrolyte through a semi-permeable porous conductor. Oxidation occurs at the anode while reduction occurs at the cathode. A simple Daniel cell shown above illustrates the following characteristic electrochemical cell reactions:
Anodic reaction:
Zn (s) → Zn 2+(aqueous) + 2 e –
and Cathodic reaction:
Cu 2+ (aq) + 2 e– → Cu (s)
Hence, the overall cell reaction:
Zn (s) + Cu +2 (aq) → Zn 2+ + Cu (s)
where zinc is oxidized and copper is reduced – a perfect case of redox reaction.
The creation of batteries and dry cells based on the above principle, uses electrochemical redox reactions to convert chemical energy into electrical energy which ultimately generates DC electric current for use in our day-to-day life by running a number of small to big gadgets and equipments. For example, storage cells are used to supply all the electrical needs in cars, trucks, buses, trains, aero planes, etc. A very familiar type of battery is the lead storage battery of a car where lead itself is the reducing agent, while lead (IV) oxide (PbO 2) acts as the oxidizing agent. Another example includes application in the space capsule where electrical energy needed is provided by the reaction of hydrogen and oxygen in fuel cells which are electrochemical cells using oxygen and hydrogen electrodes. In galvanic cells, spontaneous chemical reactions take place in the electrolytic cells involving redox reactions.
4.4.2 Electrodeposition
Based on redox reactions, a variation on the theme of electrolysis involve a] electroplating and b] participation of electrodes leading to purification of metals (such as copper). In both cases, the electrochemistry involved is similar to that discussed above in section 4.4.1 on batteries.
a) Electroplating
Electroplating is used to make jewellery, reflective /shiny object to act as mirrors, deposition on surfaces to make them resistant to abrasion, tarnishing and corrosion. There are many applications of the technique in modern world. The principles of electrochemistry involved are similar to that described above for batteries. And thus the corresponding electrochemical reactions involved illustrate another case of redox reactions.
In electrodeposition/electroplating, cells that use electricity are used to deposit metals onto the surface of another metal. Metal salts in a solution called the plating bath are used for reduction to metal at the cathode of an electrochemical cell. Any metal lower than hydrogen in the electrochemical series i.e. with a positive electrode potential, is likely to act as an anode to dissolve in an electrolyte and eventually be deposited on the same or other metal acting as cathode during electrolysis. However, this simple approach needs to be looked at a bit carefully. It is well known that metals like zinc which despite being electronegative is not necessarily heavily corroded in an electrolyte because of characteristic high hydrogen overvoltage on zinc.
b) Participating electrodes
When non-inert electrodes are used in electrolysis, they can interact with the solution being electrolysed. This is especially important when electrolysis of ions of relatively unreactive or impure metals are electrolysed using the same metal as electrode. For example, in the case of purification of copper bar, the impure copper is used as anode that would participate in the reaction by losing atoms in the form of ions in the copper(II) sulphate solution used as electrolyte, leaving behind their own electrons. Thus during the electrolysis of copper(II) sulphate solution using impure copper as anode and pure copper as cathode, the following reactions takes place:
At anode Cu(s) – 2e → Cu 2+ (aq) (oxidation)
At cathode Cu 2+(aq) + 2e→ Cu (s) (reduction)
– a perfect case of redox reactions.
4.4.3 Dry cell batteries
There is a tremendous growth of dry cell battery which as its name implies, involves no liquid components. Instead, it utilizes a range of combinations of various elements such as zinc, magnesium, mercury, silver, nickel, and cadmium. The last two are applied in the nickel-cadmium battery, which is particularly useful because it can be recharged over and over again by an external current. The current turns the products of the chemical reactions in the battery back into reactants. In general, the dry cell batteries can power all kinds of portable appliances, from flashlights to boom boxes.
4.4.4 Vanadium flow redox batteries to store and release energy
The latest utility-scale commercial storage battery in the market involves the technology of vanadium flow (V-flow) redox battery. It acquires its name because the process involves flow of active materials from the tanks to the electrodes within the cells through piping and pumps. In this electrochemical storage system, energy is allowed to be stored in two solutions containing different redox couples (which can be apparently soluble in a solvent). The idea was first conceived by Skyllas-Kazacos and co-workers in 1985.
Basic principle
Most batteries use chemicals that change charge/valency i.e. redox state in response to electron flow which would convert chemical energy to electrical energy and vice versa. In V-flow batteries, multiple valence states of vanadium are used to store and release charges. Vanadium can exist in solution in several ionic forms with different charges, namely V2+, 3+, 4+ or 5+ with each charge state having different number of electrons around the nucleus. System with fewer electrons is associated with a higher positive charge. Energy is stored by providing electrons forming V (2+and 3+), and energy is released by losing electrons to form V(4+and 5+). Such battery will actually have V2+/V3+ on one side while V4+/V5+ on the other side. As a result, cross contamination is less of a problem in such a cell. On charging up, the ratio of V2+/V3 in the cell would increase on the anode side, while the ratio of V5+/V4+ increases on the cathode side. In Appendix-1, a brief description of the battery along with its current status is presented (Technological reviews such as J. Mater. Chem. A, 2015, 3, 16913-16933 by K. J. Kim et.al., of electrodes and performance mechanisms of vanadium redox flow batteries will not be discussed here because it is not relevant in the present context).
4.4.5 Galvanic stripping of waste stream
Background
A major problem in the minerals industry arises from huge tonnages of environmentally unacceptable oxidized zinc residues containing high iron and zinc along with copper, lead, cadmium, and other heavy metals. Normally, they are precipitated in chemical forms acceptable for standard landfills. However, under current laws, this practice will no longer be allowed, and costs of compliance are expected to rise dramatically. Any application of the conventional metallurgical and chemical processes suffers from a serious drawback due to their inability to separate ions of iron and copper from zinc economically in aqueous hazardous wastes. This technical impediment with aqueous phase has now been overcome with the development of galvanic stripping process conducted in an organic liquid such as D2EHPA [di-(2-ethylhexyl) phosphoric acid].
(U.S. Patent 5,228,903, 1993, http://www.engg.ksu.edu/HSRC/96Proceed/chang.pdf, Separation Sci.& Tech., Vol.34(12), 2395,1999; Minerals & Metallurgical Processing,Vol.17(1) 17, 2000)
Also, on a laboratory scale, the recovery of gold as Au 3+ ion has been reported from its aqueous solution by extraction into an organic strong-base anionic extractant of a quaternary amine salt, ALIQUAT336 ®, in xylene.
(D.F.C.Lacerda, Braz. J. Chem. Eng. vol. 18 no.2 São Paulo June 2001; http://dx.doi.org/10.1590/S0104-66322001000200008)
Mechanics of performance
A galvanic technique by definition involves spontaneous reaction between reactants without the need of any applied current as a driving force. The process of galvanic stripping in the present context is carried out in an organic solvent which extracts the desired positively charged metal cations (+ve ions) namely Fe 3+, Cu 2+, Au 3+ and Zn 2+ from an aqueous phase. There are two basic stages of the technique namely solvent extraction and electrochemical redox reaction.
Stage 1: This is a prerequisite state for subsequent electrochemical operation. The aim here is to remove Fe 3+, Cu 2+ Au 3+ or Zn 2+ ions from aqueous phases into an organic liquid. In a typical solvent extraction also known as liquid-liquid extraction or partitioning method, aqueous industrial waste containing one or more dissolved metal cations is mixed with an organic liquid capable of extracting the desired metal cations. A conventional solvent extraction method is carried to separate the two liquids. Mass transfer of cations namely Fe 3+, Cu 2+, Au 3+ and Zn 2+ takes place across the boundary interface between the two contacting insoluble liquids namely the aqueous phase and an organic liquid. The organic liquid thus acts as an extractant of the desired ions from the aqueous phase.
Stage 2: After the phase separation, the loaded organic phase containing the desired cations is contacted with a suitable solid metal such as zinc as a reducer to provide spontaneous electrochemical reduction of the oxidation state of the ions from higher to lower (Fe 3++ e ⇆ Fe 2+) or to zero valency (metallic) (Cu 2++ 2e ⇆ Cu0 or Au 3+ +3e ⇆ Au0) states of oxidation. A brief electrochemistry of reduction effectiveness of zinc is described in Appendix-2.
Involvement of electrochemical process
The redox reaction in galvanic stripping although not normally attainable with a standard chemical driving force, it is, however, possible with the following generic electrochemical reaction pattern:
R-M1 n+(org) + M02 (s) ⇆ R-M1(n-1)+(org) + R-M2 1+(org)
where M1 represents cations namely Fe 3+, Cu 2+ and Zn 2+ to be recovered from the aqueous phase by an organic extractant, M02 is the reducing metal (zinc powder here) and R-M1 or 2(org) is the organic phase containing ions M1 or 2, and n is the number of ions/electrons involved in M n+. The organic solvent denoted by R could be D2EHPA or organic strongbase anionic extractant of a quaternary amine salt, ALIQUAT336®, in xylene as the case may be. One can expect some degree of possible cementation of M1 on the redactor powder M02 with a possible reaction:
R-M1 (org) + M02(s) ⇆ R-M2(org) + M01(s)
In terms of specific ions, cations are reduced either partially to a lower ionic state (Fe3+→ Fe2+) (reaction 1) or to a metallic state resulting in cementation of copper or gold on zinc (reactions 2 and 3) as shown below. The Zn 2+ ion in the organic solvent will, of course, remain non-responsive to the zinc powder.
- 2R – Fe3+(org) + Zn0(s) → 2R – Fe2+(org)+ R – Zn2+(org):
Redox reaction producing Fe 2+ ion - R – Cu2+(org) + Zn0(s) → Cu0 + R – Zn2+(org):
Redox reaction producing Cu0 (metallic copper) - 2R – Au 3+(org) + Zn0(s) → 2Au0 + 3R – Zn2+(org):
Redox reaction producing Au0 (metallic gold)
Reduction of Fe 3+ to the Fe 2+ state, makes it possible for easy removal of iron using a very low pH dilute acid solution such as 6N HCl that would only strip Fe2+ and neither Zn 2+ nor Fe3+ from the organic phase:
R-Fe2+(org) + 2H+(aq) → Fe2+(aq) + 2R-H+(org).
This feature of stripping Fe2+ with a dilute acid without affecting Fe3+ is feasible because of lower stability of ferrous ions in D2EHPA.
4.5 Energy from thermal reaction
4.5.1 Fuelling space shuttle
Oxidation-reduction reactions also fuel the most advanced form of transportation known today namely the space shuttle. A combination of ammonium perchlorate (NH4 ClO4) and powdered aluminium acts as a solid rocket booster via oxidation-reduction reactions to provide the shuttle an enormous amount of extra thrust. The actual orbiter vehicle is relatively small compared to its external power apparatus which surrounds it on either side and consists of two solid rocket boosters along with an external fuel tank that powers the rocket. The tank contains super-cold gases such as liquid hydrogen fuel and liquid oxygen as oxidizer which burned together to provide the fuel for the space shuttle. These two gases are kept in separate compartments because they are extremely explosive. When they react, water is, of course, formed, but in doing so, a vast quantity of energy is released. The chemical equation for this redox reaction can be simply written as:
2H 2 + O 2 →2H 2 O +energy.
(On January 28, 1986, something went terribly wrong with this arrangement of the fuel tank on the space shuttle Challenger. Cold weather had fatigued the O-rings that sealed the hydrogen and oxygen compartments, and consequently the gases fed straight into the flames behind the shuttle itself. This produced a powerful and uncontrolled oxidationreduction reaction. As a consequence, an explosion occurred that took the lives of all seven astronauts aboard the shuttle).
4.5.2 Combustion of fuels
The word “combustion” (supplying energy) means burning that involves oxidation-reduction reaction. The energy required for our daily needs is obtained by combustion/oxidation of fuels. In everyday life, one would experience combustion every time one strikes a match, burns a candle, builds a fire, or lights a grill. The process of combustion combines energetic molecules with oxygen to produce carbon dioxide and water.
Perhaps a common misconception of burning, or
combustion is that it is perceived more as a physical process than a hemical one. Nevertheless, combustion of hydrocarbons in fossil fuels, or the burning of organic material in wood represents quintessential redox reaction. In each case, carbon in the compound being burned bonds with oxygen atoms in the air, while some oxygen bonds to the hydrogen forming moisture. Thus the compound being burned is oxidized while oxygen is reduced, and carbon dioxide and water vapour are emitted as combustion products with emission of energy.
In simple cases of burning a fuel such as coal (which is carbon), or petrol/gasoline (a long chain organic compound, C8 H18 ), the fuel is oxidized and oxygen is reduced. Redox reaction occurs with reduction of oxygen and oxidation of carbon. In terms of oxidation numbers, the oxidation number of carbon jumps from 0 to 4, while that of oxygen is reduced to a pair of two negative charges. As they burn, these two elements form carbon dioxide or CO2, in which the pair of two negative charges of the oxygen atoms cancel out the four positive charge of the carbon atom to yield a compound (CO2) that is electrically neutral:
For coal: C + O2 → CO2+ energy↑;
For petrol: 2 C8 H18 + 25 O2 →
16 CO2 + 18 H2 O +energy ↑
Thermodynamically, combustion reactions are strongly exothermic, meaning liberation of heat energy. Due to the large number of moles of gaseous products, a large positive entropy change is also associated with combustion reactions. For these reasons, combustion reactions are strongly spontaneous.
Combustion of fuels such as wood, gas, kerosene, petrol, etc. produces a large amount of energy which is utilized for various purposes in our daily life that including heating homes and buildings, driving cars, trucks, ships, airplanes, and trains, operating industrial processes and numerous other purposes.
Explosions and explosives
The concept of combustion can be extended to “explosion” which in simple terms, is a fast form of combustion and hence involves redox reaction. As with any type of chemical reaction, combustion takes place when chemical bonds are broken and new bonds are formed. It so happens that combustion can particularly result in a dramatic type of redox reactions. Whereas one cannot watch the slow process of rusting of iron, combustion is a noticeable event. A more dramatic form of combustion can take place at a rate so rapid that it results in an explosion. The first such explosives were invented by the Chinese during the Middle Ages, and these included not only fireworks and explosive rockets, but also gunpowder. In modern times, one can refer to an internal combustion engine which does not simply burn fuel but rather, by the combined action of the fuel injectors (in a modern vehicle) in concert with the pistons, cylinders, and spark plugs, it actually produces small explosions in the molecules of petrol/gasoline. These result in an output of power that becomes essential to turn the crankshaft, and ultimately the wheels.
4.5.3 Combustion and human life
Combustion has been a significant part of human life ever since our prehistoric ancestors learned how to harness the power of fire to cook food and light their caves. In fact, our modern age is even more combustion-driven than our forebears. An example is the development of steam engine in the period from the late seventeenth century to the early nineteenth century. For centuries, burning animal fat for uses in torches, lamps, and eventually in candles provided light for humans. Wood fire supplies warmth, as well as a means to cook meals. All these examples illustrate some of the good uses of combustion.
In real life example, human body needs energy for functioning properly. This is provided by the oxidation of glucose in our body to CO2 and water:
C6H12O6 (glucose) + 6 O2 → 6 CO2 + 6 H2O + energy
4.5.4 Combustion and Human Health
Combustion, though do much good, can also result in some harm which may not be too obvious initially. For example, by burning fossil fuels or hydrocarbons, the excess carbon (in the form of carbon dioxide and carbon monoxide) is released to the atmosphere that would damage the environment. In fact, oxidationreduction reactions are intimately connected with the functioning of the natural environment. For example, redox reactions in photosynthesis produce two essentials of human life namely oxygen and carbohydrates.
In a typical cellular respiration, an oxidation-reduction reaction takes place in which living things break down molecules of food to produce energy, carbon dioxide, and water.
Enzymes in the human body regulate redox reactions. These complex proteins, of which several hundred are known, act as catalysts, speeding up chemical processes in the body. Oxidation-reduction reactions also take place in the metabolism of food for energy, with substances in the food broken down into components the body can use.
5 Application of redox reactions in domestic cases
Redox reactions are among the most common and important chemical reactions in everyday life. The great majority of these reactions can be classified on the basis of how rapidly they occur. Compared to combustion discussed above where redox reactions occur very rapidly producing noticeable heat and light, reactions involving photosynthesis, corrosion (highlighted earlier), decay and some biological processes progress so slowly that no noticeable heat and light are produced.
5.1 Photosynthesis
Photosynthesis (a reaction to make food in plants) takes place in the green leaves of plants and a few microorganisms whereby carbon dioxide (CO2) and water are transformed under the influence of sunlight to provides the energy to form
- carbohydrates (glucose) that they use to store energy, and
- oxygen that they release into the environment.
During photosynthesis, water (H2O) is oxidized to molecular oxygen implying loses electrons, while CO2 is reduced to carbohydrates meaning gain of electrons. The plant uses the glucose produced as fuel for its metabolic process.The reaction of photosynthesis is sensitized by chlorophyll. The overall reaction for photosynthesis can be expressed as:
6CO2 + 6 H2O + Sunlight/Chlorophyll →
C 6H12O6 (carbohydrate/ glucose) + 6 O2
The above reaction is one of the most common everyday chemical reactions and also one of the most important since this is how plants produce food for themselves and animals and convert carbon dioxide into oxygen. It also maintains a constant supply of 21% of O2 by volume in the atmosphere needed for combustion of fuels and breathing of all the living creatures in the world.
5.2 Decay
The compounds that make up living organisms, such as plants and animals, are very complex. They consist primarily of carbon, hydrogen and oxygen. When a plant or animal dies, their composition in terms organic compounds begin to react with oxygen. A simple way to represent such compounds is to use the letters x, y, and z to show that many atoms of carbon, hydrogen, and oxygen are present in the compounds. The reaction is similar to the combustion of petrol in a car, but it occurs much more slowly. The process is known as decay, and it is another example of a common oxidation-reduction reaction. The equation for the decay (oxidation) of a compound that might be found in a dead plant can be represented as
C x H y O z + O 2 → CO 2 + H 2 O
5.3 Respiration
Cellular respiration allows organisms to liberate the energy stored in the chemical bonds of glucose. The balanced redox reactions can be written as
C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + 36 ATP,
where ATP is adenosine triphosphate, a simple energy-supplying compound that drives various metabolic processes. In this reaction, glucose is oxidized and oxygen is reduced. A compound losing hydrogen atoms means it has been oxidized, while gaining them would imply reduction.
This aerobic cellular respiration is the opposite process of photosynthesis in that energy molecules are combined with the oxygen we breathe to release energy needed by our cells plus carbon dioxide and water. Energy used by cells is chemical energy in the form of ATP.
5.4 Biological processes
Live system
Many of the changes that take place within living organisms are due to redox reactions. For example, digestion of food is an oxidation process. Food molecules react with oxygen in the body to form carbon dioxide and water. Energy is also released in the process. The carbon dioxide and water are eliminated from the body as waste products, but the energy is used to make all the chemical reactions possible that keep an organism alive and help it to grow.
Although oxidation process is very important to humans via multiple reactions that would keep our bodies going, such a process is linked to aging, cancer, hardening of the arteries, and rheumatoid arthritis in humans. It appears that oxygen molecules and other oxidizing agents with high electronegativity are always hungry for electrons which are extracted from the membranes in human cells. Over time, this can cause a gradual breakdown in body‘s immune system. To forestall the effects of oxidation, some doctors and scientists recommend antioxidants which are reducing agents such as vitamin C and vitamin E that are water/fat-soluble vitamins. These antioxidants might inhibit damages caused by the by-products of oxidation reactions that can occur in the human body after exposure to some toxic chemicals.
Food and dietary products
In everyday life, oxidation-reduction reactions are responsible for preventing, for example, spoiling of food. The culprit here is oxygen from the air which, of course, oxidizes the food. Research has been carried out evaluating the benefits of antioxidants in foods and dietary supplements. To prevent spoilage, manufacturers of foods add preservatives/antioxidants which act as reducing agents. For example, if lemon juice is squirted on a cut apple, the vitamin C (a reducing agent) in the juice will prevent the browning of the apple that is caused by oxidation of the cut apple by air.
5.5 Redox reactions in chemistry
Titration
Redox reaction in titration is an important component of quantitative chemical analysis involving both inorganic and organic reactions.Titration involves reactions between oxidizing and reducing agents and help in estimating the amount of unknown substances in solutions. Potassium permanganate and cerium (IV) solutions can be used as strong oxidizing agents in the analysis of iron, tin, peroxide, vanadium, molybdenum, titanium and uranium. Potassium dichromate is an oxidizing agent used in the analysis of organic materials in water and wastewater.
Bleaching and sanitization
Oxidation-reduction reactions can be used for bleaching materials and sanitizing water. Sodium hypochlorite (in solution) is used as a laundry bleach liquid as well as a solid component of dishwasher powders and cleansers. Calcium hypochlorite is often used for swimming pool sanitation. The hypochlorite kills bacteria in water by oxidizing them. Ozone is a powerful oxidizing agent that can also be used to purify water.
The natural ability of algae and other water plants to oxidize harmful materials in sewage has been used in sewage lagoons, also known as oxidation pond systems. Small volumes of raw sewage can be treated simply by directing the sewage into shallow ponds containing algae and other water vegetation. In Belgium, nitrates are removed from wastewater by bacteria that reduce the nitrates to nitrogen which can be safely released into the atmosphere.
6 Final comments
The present article reflects the notion that redox reactions are uniquely everywhere. Human body uses redox reactions to convert food and oxygen to energy plus water and CO2 which is then exhaled. Their applications in both industrial and domestic sectors are highlighted.
Redox reactions can be harnessed as a source of energy. When hydrogen and oxygen are carefully fed into an electrochemical cell namely a fuel cell, the subsequent oxidation-reduction reactions would operate to provide vast amount of energy. The only by-product of the reaction between hydrogen and oxygen is non-polluting water. Combustion of hydrogen to power vehicles is a latest development where application of the hydrogen/oxygen reaction is used. Currently, hydrogen is produced from water using electricity and it takes more energy to make the hydrogen than is obtained from combustion of water. In future, hydrogen might be made from solar energy and would provide a non-polluting fuel.
Appendix 1
Brief description of the Vanadium flow battery
A schematic illustration of the battery is shown below. It consists of two tanks of liquid (anolyte and catholyte) which simply sit there until needed. When pumped into a reactor, the two solutions flow adjacent to each other past a membrane, and generate a charge by moving electrons back and forth during charging and discharging i.e. typical displays of oxidation (at anode) and reduction (at cathode) reactions (see sec. 4.4.4).
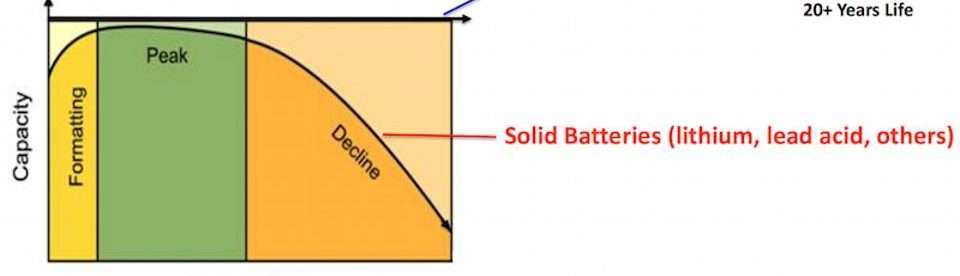
The battery can provide almost unlimited energy capacity simply by using larger electrolyte storage tanks. It can be left completely discharged for long periods with no ill effects, making simpler maintenance and hence with reduced cost of storage compared to other batteries. However, these batteries are rather large and best suited to industrial and utility scale applications. Although V-flow battery could never fit in an electric car (so the Tesla battery is safe for now), it out competes Li-ion, and any other solid battery for utility-scale applications. These batteries are safer, more scalable, longer lasting and cheaper (apparently less than half the cost per kWh compared to standard batteries).
Present status
UniEnergy Technologies (UET) of the U.S., amongst others, have developed fully containerized, non-flammable, reusable commercial batteries. Presently, the largest V-flow battery in the U.S. is a 2MW/8MWh (power/total dischargeable energy in a single full charge) installed by UET at the Snohomish County Public Utility District’s Everett Substation (SnoPUD), Everett Substation in Washington State that is scheduled to come online in January 2017. This type of vanadium battery can keep the lights on in 1,000 homes for eight hours. A V-flow battery system planned for Dalian China by UET’s sister company Rongke will soon be the largest battery in the world at 200MW/800MWh.
Unlike solid batteries, like lithium-ion or lead-acid that begin to degrade after a couple of years, V-flow batteries, according to UET, are fully reusable over semi-infinite cycles and do not degrade, thus giving them a long life. Such batteries apparently offer the best deployable large battery storage technology developed thus far.
Appendix 2
a) Effectiveness of zinc as a reducer – electrochemical redox criteria
Some relevant data on standard electrochemical reduction potentials are presented below for M n+/M systems of current interest where n is the number of ions/electrons. It is of paramount importance to strictly follow the sign convention of the electrode potential. Based on the data below, it is obvious that the reduction potentials of Au 3+→Au0, and Fe 3+→Fe 2+ and Cu 2+→Cu0 systems are much higher and more electropositive than for Zn 2+ →Zn0 systems. This would imply that Zn metal being more electronegative is capable of effectively reducing more electropositive, noble systems either reducing to a lower ionic state (Fe 3+ →Fe +2), or to a metallic state namely Cu 2+→ Cu0 or Au +3→Au0 by absorbing/gaining electrons. Cations reduced to the metallic state would deposit onto the solid metal reductor namely zinc powder (a generic cementation reaction) while reduction of metal cation namely Fe 3+→ Fe +2 is essential for easy removal of iron. See Table 1.
Electrons gained by Au 3+, Fe 3+, and Cu 2+ ions are available from the metallic reducer Zn which will be ionised in the process:
Zn0(s) → Zn 2+ + 2e.
Thus zinc which is less noble i.e. more electronegative than Au 3+, Fe 3+ or Cu 2+ ions, would act as an excellent reducer when added in powder form.
b) Final comments on galvanic stripping
The feasibility of galvanic stripping process has demonstrated interesting chemical process options for the treatment of a variety of hazardous metal bearing wastes. There is a commercial interest in the process because direct, short-range spontaneous electrochemical redox reactions are used to accomplish separations that are impossible using conventional hydrometallurgical methods. Such reactions are not normally expected because the organic solutions are essentially non-conductors, at least orders of magnitude worse than comparable aqueous electrolytes. However, various works have demonstrated that galvanic stripping involving redox reactions make the process a technically feasible option to treat waste residues.
Resources
- Atkins, P.W.; Beran, J.A.: General Chemistry, 2nd ed. New York,
Scientific American Books, 1992 - Shreir, L.L.: ed. Corrosion – Volume 1 and 2, London, Newnes-
Butterworths, 1976 - Kostiner, E.: Oxidation-Reduction Reactions and Electrochemistry,
Study Keys to Chemistry, Barron‘s Educational Series, Inc,
1992 - Lide, D.R.: ed. CRC Handbook of Chemistry and Physics, Boca
Raton, CRC Press, 2001 - Raven, P.H.; Johnson, G.B.: Oxidation-Reductio,: The Flow of
Energy in Living Things, and The Nitrogen Cycle, Biology. 3rd ed.,
Dubuque, Wm. C. Brown Publishers, 1992 - Knapp, B.J.: Oxidation and Reduction, Danbury, CT, Grolier Educational,
1998 - Oxidation-Reduction Reactions: Redox, UNC-Chapel Hill Chemistry
Fundamentals - Yount, L.; Lavoisier, A.: Founder of Modern Chemistry, Springfield,
NJ, Enslow Publishers, 1997 - Zumdahl, S.S.: Introductory Chemistry: A Foundation, fourth edition,
Boston, Houghton Mifflin, 2000 - Final Report: Design and Development of an Innovative Industrial
Scale Process to Economically Treat Waste Zinc Residues – EPA
Grant Number: R825549C055, Missouri University of Science and
Technology - http://www.shodor.org/unchem/advanced/redox/
- http://www.scienceclarified.com/Oi-Ph/Oxidation-Reduction-
Reaction.html#ixzz4bCwxwRM3 - https://www.thoughtco.com/examples-of-chemical-reactions-ineveryday-
life-604049, - http://science.jrank.org/pages/4963/Oxidation-Reduction-Reaction-
Current-future-uses.html - http://science.jrank.org/pages/3465/Hydrogenation-Hydrogenation-
in-industry.html - http://www.chemistry-assignment.com/applications-of-redoxreactions
- http://sciencing.com/oxidation-reduction-used-everydaylife-
10020550.html - http://www.britannica.com/science/oxidation-reduction/Historical-
origins-of-the-redox-concept - https://www.heraldnet.com/news/pud-invest-in-11-2-million-inenergy-
storing-units/ - https://www.chemguide.co.uk/inorganic/redox/definitions.html
- http://www.worldcat.org/title/chemlab./oclc/37315246Appendix – 1