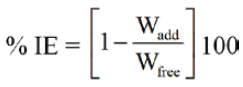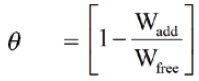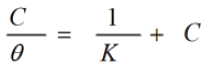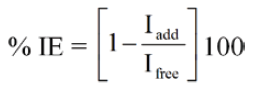Inhibition of aluminum corrosion in 1M HCl in absence and presence of three compounds of non ionic surfactants compounds derived from phenol was investigated using hydrogen evolution reaction, weight loss galvanostatic polarization and electrochemical impedance spectroscopy techniques. It was found that the percentage inhibition increases with increasing the concentration of inhibitor, amount of ethylene oxide unit and with decreasing temperature. The inhibitive action of non ionic surfactant compounds was explained in terms of blocking the electrode surface by adsorption process. The adsorption process follows Langmuir isotherm. The polarization measurements showed that these inhibitors are acting as mixed inhibitors for both cathodic and anodic reaction. Electrochemical impedance spectroscopy technique exhibit one capacitive loop indicating that, the corrosion reaction is controlled by charge transfer process. Some activated thermodynamic parameters are calculated and explained.
1. Introduction
Corrosion of aluminum and its inhibition is very important to many researchers due to their high technological value and wide range of industrial applications especially in aerospace, motor vehicle, house hold industries, pipes, chemical batteries and other studies [1-2]. Hydrochloric acid is often used for electrochemical etching and acid pickling of Al. Several studies were made to decrease the rate of corrosion of Al in HCl by adding of corrosion inhibitors. Most of the efficient acid inhibitors are organic compounds contained heterocyclic atoms (nitrogen, oxygen, sulphur and phosphours), aromatic rings or multiple bonds [4-17]. These compounds are adsorbed at metal surface and they block the active site and consequently, decreasing the corrosion rate and consequently increase the inhibition efficiency [19]. The strength of adsorption process depends on the chemical structure of the inhibitors and its prosperities such as aromaticity, functional groups, electron density at the donor atoms-orbital‘ s characters of donating electrons and the number of aromatic rings [20].
The aim of the present work is to study the inhibitive action of three non ionic surfactants derived from phenol compounds toward the corrosion of Al in 1.0 M HCl solution using hydrogen evolution, weight loss and galvanostatic polarization measurements. The percentage inhibition efficiency was calculated using the above techniques. The adsorption isotherms, the effect of temperature and the inhibition mechanism of these compounds was explained.
2. Experimental Methods
The working electrode used in this study was aluminium with purity 99.99 % provided by „aluminum Company of Egypt, Nagh Ammady“. For weight loss measurements and hydrogen evolution reaction. The test Al sheets having dimension 2.0 x 1.0 x 0.2 cm3. The sheets were first polished with different grades of emery paper in order to obtain a smooth surface, and then degreasing with acetone and then rinsed with distilled water, dried between two filter papers. The procedure methods of weight loss and hydrogen evolution reaction measurements were carried out as described elsewhere [21].
For galvanostatic polarization measurements, a cylindrical rod embedded in araldite with exposed surface of 0.54 cm2 was used. Galvanostatic anodic and cathodic polarization measurements were carried out using a PS remote potentiostat with PS6 software to calculate the corrosion parameters. Three compartment cell with a saturated calomel reference electrode and platinum foil auxiliary electrode was used. The polishing, degreasing and washing of the Al electrode as explained in the above techniques. Solutions were not deaerated to make the conditions identical to hydrogen evolution measurements and weight loss. All the experiments were carried out at 25±1oC by using ultra circulating thermostat. Three non ionic surfactants derived from phenol compounds were prepared using a method described elsewhere [22]. Electrochemical impedance spectroscopy (EIS) were carried out at open circuit potential (Eocp) in the frequency
range from 10 kHz to 100 MHz with signal amplitude perturbation of 5 mV by using a computer potentiostat (AutoLab 30 Metro).
3. Results and discussion
3.1. Hydrogen evolution reaction measurement
Figure 1 represents the relation between the volume of hydrogen evolved with time during dissolution of Al in 1.0 M HCl solution in absence and presence of different concentrations of compound III as an example of the tested non ionic surfactants derived from phenol compounds. Similar curves were obtained for other two compounds I and II (not shown). Inspection of Figure 1, reveals that the volume of hydrogen evolved increases linearly with the time. The rate of the evolution is very small at the beginning of the reaction and after certain time called induction period, the volume of H2 evolved increases. During the induction period, the breakdown of pre-immersion oxide film on Al surface takes place before the start of metal attack. After induction period the dissolution of Al is linearly related to the reaction time according to the following equation:
where, V is the volume of H2 evolved due to the corrosion of Al in 1.0 M HCl solution at time (t) and K is the rate constant of the corrosion reaction. The corrosion rate was obtained from the slope of the straight portion of curves after induction period. As the concentration of non- ionic surfactants compounds increases the rate of hydrogen evolution reaction decrease and hence the rate of corrosion decreases and the inhibition efficiency increases. This indicates that the inhibiting effect of non ionic surfactants compounds.
The percentage inhibition efficiency (% IE) was calculated using the following equation:
where, Rfree and Radd are the corrosion rate of Al coupons in absence and presence of inhibitors, respectively.
The values of inhibition efficiency were calculated and given in Table 1. At one and the same inhibitors concentration the values of IE decreases in the following order:
Compound III > Compound II > Compound I
This order will be discussed later.
3.2. Weight loss measurements
Figure 2 shows the weight loss- time curves for the corrosion of aluminum in 1.0 M HCl solution in absence and presence of different concentrations of compound III at 25±1oC as an example of the studied inhibitors. Similar curves were obtained (not shown). The weight loss of Al are decreased. This means that the presence of these compounds retards the corrosion of aluminum in 1 M HCl solution. These compounds act as an inhibitors.
The linear variation of weight loss with time in uninhibited and inhibited 1 M hydrochloric acid indicates the absence of insoluble surface films during corrosion. In the absence of any surface films, the inhibitors are first adsorbed onto the metal surface and thereafter impede corrosion either by merely blocking the reaction sites (anodic and cathodic) or by altering the mechanism of the anodic and cathodic partial processes.
The percentage inhibition efficiency (% IE) and a parameter (θ) which represents the part of the metal surface covered by the inhibitor molecules were calculated using the following equations:
where, Wfree and Wadd are the weight loss of Al coupons in absence and presence of inhibitors, respectively.

Fig. 1: Volume of hydrogen evolved-time curves for the corrosion of aluminum in 1.0 M HCl in the absence and presence of different concentrations of compound III at 25oC.
(1) 0.00 ppm compound III, (2) 100 ppm, (3) 200 ppm, (4) 300 ppm, (5) 400 ppm, (6) 500 ppm

Fig. 2: Weight loss-time curves for the corrosion of aluminum in 1.0 M HCl in the absence and presence of different concentrations of compound III at 25oC.
(1) 0.00 ppm compound III, (2) 100 ppm, (3) 200 ppm, (4) 300 ppm, (5) 400 ppm, (6) 500 ppm

Tab 1.: Effect of compounds on rate of corrosion (Rcorr.), inhibition efficiency (% IE) and surface coverage (θ) obtained from weight loss and hydrogen evolution measurements for dissolution of Al in 1.0 M HCl solution.
The calculated values of % IE at 30oC are listed in Table 1. The order of decreasing inhibition efficiency of the investigated compounds is as follows:
Compound III > Compound II > Compound I
This sequence will be discussed later in the inhibition mechanism section.
3.3. Adsorption Isotherm
Non ionic surfactants derived from phenol compounds inhibit the corrosion of Al in acidic solutions by its adsorption on the aluminum surface. Theoretically, the adsorption process can be regarded as a single substitution process in which an surfactant molecule, in the aqueous phase substitutes an „x“ adsorbed on the metal surface
where x is the size ratio that is, the number of water molecules replaced by one organic inhibitor. The adsorption depends on the structure of the inhibitor, the type of the metal and the nature of its surface, the nature of the corrosion medium, the pH value, the temperature and the electrochemical potential of the metal-solution interface. Also, the adsorption provides information about the interaction among the adsorbed molecules themselves as well as their interaction with the metal surface.
Attempts were made to fit the surface coverage (θ) to various isotherm including Langmuir, Freundlich, Frumkin, Temkin and Flory-Huggins. By far the best results is obtained with Langmuir adsorption isotherm and can be represented using the following equation [23]:
where K and C represent the equilibrium constant of adsorption process and additive concentrations respectively. Plotting C/θ against C gave straight lines with unit slope (Fig. 3) suggesting that the adsorption of non ionic surfactants derived from phenol compounds on the aluminum surface follows Langmuir isotherm. This indicates that there are no interaction or repulsion forces occurred between the adsorbed molecules.

Fig. 3: Langmuir adsorption isotherm for aluminum electrode in 1.0 M HCl solution in absence and presence of non ionic surfactant compounds
The values of K is obtained from the intercept of Figure 3. The values of K is related to the free energy of adsorption according to the following equation:
where R is the universal gas constant is the absolute temperature and 55.5 in the concentration of water in bulk solution M-1. The values of K are equal to 0.02, 0.05 and 0.11 for compounds I, II and III, respectively. The high value of K indicates that the strong adsorption of non ionic surfactants compounds on the Al surface.
The calculated values of ΔGoads for non ionic surfactants compounds derived from phenol adsorbed on the aluminum surface are equal to –22.56, –23.48 and –24.62 kJ mol–1 for compounds I, II, and III, respectively.
The free energy change of adsorption is associated with water adsorption/desorption equilibrium which forms an important part in the overall free changes of adsorption. The negative value of ΔGoads obtained indicates that the adsorption process of non ionic surfactants on the aluminum surface is spontaneous one.
3.4. Effect of temperature
The effect of increasing temperature on the corrosion rate of Al in free 1 M HCl and containing 500 ppm of three non ionic surfactants compounds was tested in the temperature range of 25-55 °C using weight loss measurements. Similar curves to Figure 1 were obtained (not shown). The effect of temperature on the Rcorr and % IE are listed in Table 2. It is obvious from the data listed in Table 2. The values of Rcorr increases and the inhibition efficiency decreases. This indicates that the adsorption of non ionic surfactant on the Al surface is physically.
The activation energy (Ea) of the corrosion process was calculated using Arrhenius equation [24]
and the logarithmic form is
where Rcorr is the rate of corrosion obtained from weight loss, A is Arrehenius constant, R is the gas constant and T is the absolute temperature.
Figure 4 represents the plots of logarithm of corrosion rate (log Rcorr), with reciprocal of absolute temperature (1/T) for aluminum 1.0 M HCl containing 500 ppm of the studied surfactants compounds. The values of Ea can be obtained from the slope of the straight lines were found to be 20.4 KJ mol-1 in 1.0 M HCl and 22.8, 24.6 and 25.9 kJ mol-1, for compounds I, II and III, respectively.

Fig. 4: Relation between log Rcorr and the reciprocal of temperature of Al electrode in
a) 1.0 M HCl
b) 1.0 M HCl + 500 ppm of the studied compounds
The increase of the activation energy in the presence of inhibitors is attributed to an appreciable decease in the adsorption process of the inhibitors on the metal surface with increase of temperature and a corresponding increase in the reaction rate because of the greater area of the metal that is exposed to the acid.
The entropy of activation (ΔS*) and the enthalpy of activation (ΔH*) for dissolution of Al in 1 M HCl in presence of 500 ppm of each used compound were obtained by applying the transition state equation [24].
where N is Avogadro‘s number, h is Planck‘s constant.
Figure 5 represents the relation between the logarithm of (log Rcorr/T), with reciprocal of absolute temperature (1/T) for aluminum 1.0 M HCl containing 500 ppm of the studied compounds. Straight lines were obtained with a slope of (-ΔH*/2.303 R) and an intercept [log (R/Nh -ΔSo/2.303 R)]. The values of ΔH* obtained from the slope of the straight line equal 17.46 KJ mol-1 in 1 M HCl and equal 19.77, 20.45 and 22.66 KJ mol-1 in presence of compound I, II and III, respectively.

Fig. 5: Relation between log Rcorr/T and the reciprocal of temperature of Al electrode in
a) 1.0 M HCl
b) 1.0 M HCl + 500 ppm of the studied compounds
The values of ΔH* are different for studied compounds which mean that their structure affect the strength of its adsorption on the metal surface. The increase in the activation enthalpy (ΔH*) in presence of the inhibitors implies that the addition of the inhibitors to the acid solution increases the height of the energy barrier of the corrosion reaction to an extent depends on the type and concentration of the present inhibitor. The values of ΔS* calculated from the intercept of the straight line were found to 185.77 J mol-1 k-1 in 1.0 M HCl and 276.14, 285.23 and 298.76 J mol-1 K-1 for compound I, II and III, respectively.

Tab. 2: Effect of rising temperature on the corrosion rate and percentage inhibition efficiency for the corrosion of Al in 1.0 M HCl solution devoid and containing 500 ppm of non
ionic surfactants using weight loss measurements.
The negative values of ΔS* in the absence and presence of the inhibitors implies that the activated complex is the rate determining step and represents association rather than dissociation. It is also reveals that an increase in the order takes place in going from reactants to the activated complex. This means that the activated molecules are in high order state than that at the initial stage [25].
3.5. Galvanostatic polarization
The effect of addition of three non ionic surfactant compounds on the galvanostatic polarization curves of Al in 1.0 M HCl solution was studied. Figure 6 shows the glvanostatic polarization curves of aluminum in 1.0 M HCl solution in the absence and presence of different concentrations of compound III as an example of the studied non
ionic surfactants compounds. Similar curves were obtained for other two compounds (not shown). The corrosion parameters such as, anodic Tafel slope (βa), cathodic Tafel slope (βC), corrosion potential (Ecorr), corrosion current density (Icorr), and inhibition efficiency (I.E.) were calculated and given in Table 3.

Fig. 6: Anodic and cathodic polarization curves of aluminium electrode in 1 M HCl solution in the absence and presence compound III.
(1) 0.00 ppm compound III, (2) 100 ppm, (3) 200 ppm, (4) 300 ppm, (5) 400 ppm, (6) 500 ppm

Tab. 3: Corrosion parameters obtained from potentiodynamic polarization technique for aluminum electrode in 1.0 M HCl in the
absence and presence of non ionic surfactant compounds.
The percentage inhibition efficiency (% IE) was calculated from corrosion current density values using the equation.
where, Ifree and Iadd are the corrosion current densities in absence and presence of inhibitors.
Inspection of Table 3 reveals that, as the concentration of non ionic surfactants compounds increases, it is clear that,
- Values of anodic (ba) and cathodic (bc) Tafel
slopes are approximately constant indicating
that these compounds are of mixed type
inhibitors. The constant values of Tafel slopes
indicates that the adsorption of non ionic surfactants
at the aluminum surface according to
blocking adsorption mechanism. - Values of Ecorr is shifted to more negative
potentials, the values of Icorr decreases and
values of IE increases indicating the inhibiting
effect of these compounds. - The inhibition efficiency of non ionic surfactants
decreases in the following order:
compound III > compound II > compound I
The efficiencies obtained from the galvanostatic technique were in good agreement with those obtained from the weight loss and hydrogen evolution reaction measurements. This proves the validity of this tools in the measurements of the investigated inhibitors.
3.6. Electrochemical impedance studies (EIS)
Electrochemical impedance measurements were carried out over the frequency range from 10 kHz to 100 MHz at open circuit potential. The sample equivalent Randle circuit for the studies is shown in Figure 7, where Rs (Ω cm2) represents the solution and corrosion products film in series with the parallel combination of resistor Rct (charge transfer resistance) and capacitor Cdl (double layer capacitance) which represents the corroding interface.

Fig. 7: Electrical equivalent circuit (Rs = uncompensated solution resistance, Rct = charge transfer resistance, and Cdl = double layer capacitance).

Fig. 8: The Nyquist plots for Al in 1.0 M HCl solution in absence and presence of compound III at 30oC.
(1) 0.00 ppm, (2) 100 ppm, (3) 200 ppm, (4) 300 ppm, (5) 400 ppm, (6) 500 ppm.
The effect of addition of three non ionic surfactant compounds on the electrochemical impedance spectroscopy technique of Al in 1.0 M HCl solution was studied. Figure 8 shows the complex- plane impedance plots (Nyquist plots) for Al in 1.0 M HCl solution in the absence and presence of various concentrations of compound III at 30oC as an example of the studied non ionic surfactants compounds. Similar curves were obtained for other two compounds (not shown). As it can be seen from this Figure 8, the Nyquist plots contain depressed semi-circle with the centre under the real axis, whose size increases with the inhibitor concentration, indicating a charge transfer process mainly controlling the corrosion of Al. Such behaviour, is characteristic for solid electrodes and often refers to a frequency dispersion, has been attributed to roughness and other in homogeneities of the solid surface [26, 27]. It is apparent, from these plots that the impedance response of Al in uninhibited acid solution has significantly changed after the addition of inhibitor compound in the corrosive solutions.

Tab. 4: Electrochemical parameters obtained from by electrochemical impedance spectroscopy technique for aluminum electrode in 1.0 M HCl solution in absence and presence of various concentrations of three non ionic surfactants derived from phenol compounds
This indicated that the impedance of the inhibited substrate has increased with increasing concentration of inhibitor. The characteristic para associated to the impedance diagrams (Rct and Cdl) and % IE are given in Table 4. The percentage inhibition efficiency % IE was calculated from the following equation
where, R°ct and Rct are the uninhibited and inhibited charge transfer resistance, respectively. The results obtained from the decrease of the local dielectric constant and/or from the increase of thickness of the electrical double layer suggested that the EIS data (Tab. 4) show that the Rct values increases and the Cdl values decreases with increasing the inhibitor concentrations. This is due to the gradual replacement of water molecules by the adsorption of the inhibitor molecules on the metal surface, decreasing the extent of dissolution reaction. The higher (Rct) values, are generally associated with slower corroding system [28].
The decrease in the Cdl can inhibitor molecules function by adsorption at the metal/solution interface. The IE% obtained from EIS measurements are close to those deduced from polarization measurements. The order of inhibition efficiency obtained from EIS measurements decreases in the following sequence is III > II > I.
3.7. Mechanism of inhibition
The corrosion inhibition of Al in 0.1 M HCl solution by three non ionic surfactant compounds as indicated from hydrogen evolution reaction, weight loss and galvanostatic polarization techniques were found to depend on the concentration and nature of the inhibitor. The observed corrosion parameters in the presence of non ionic surfactant compounds, namely, the volume of hydrogen evolution reaction, weight loss and the corrosion current density decreases and hence, the percentage inhibition efficiency increase.
The transition of metal/solution interface from a state of active dissolution to the passive state can be attributed to the adsorption of the inhibitor molecules at the metal/solution interface, forming a protective film. It is well recognized that the non ionic surfactant compounds set up their inhibition action via the adsorption of the inhibitor molecules onto the metal/solution interface. The adsorption process occurred through ethoxy groups on both the anodic and cathodic areas while the hydrocarbon parts protrude brush-like into the solution. One cannot exclude, however, the possible adsorption of these compounds via π-electrons of the double bonds present in the benzene ring. The presence of more than one double bond in the inhibitor molecule is beneficial to inhibition efficiency [29].
The percentage inhibition efficiency obtained from different techniques increased with an increase in the number of ethylene oxide unit in the following order:
Compound III > II > I
Number of ethylene oxide unit 7 > 5 > 3
The increase in concentration of the inhibitor would result in a lowering of the interfacial tension at the metal surface. This lowering in the interfacial tension is thought to cause a decrease of the bulk concentration of the inhibitor and an increase in its concentration at the metal surface.
4. Conclusions
- Non-ionic surfactants derived from phenol inhibit the corrosion of Al in 1 M HCl solution.
- The percentage inhibition efficiency increases with concentration of inhibitor, number of ethylene oxide and decreasing temperature.
- The inhibitors acted as mixed –type inhibitors and the inhibition is caused by active sites blocking effect.
- The inhibition was explained due to the adsorption of these compounds on the Al surface. The adsorption of inhibitors obeyed Langmuir isotherm.
- Electrochemical impedance spectroscopy indicates that the corrosion reaction is controlled by charge transfer process.
References
- K. F. Khaled, M. M. Qahtani, The inhibitive effect of some tetrazole derivatives towards Al corrosion in acid solution: Chemical, electrochemical and theoretical studies, Mater. Chem. and Physics, 113 (2009), 150-158.
- A. Y. Musa, A. A. Kadhum, A. B. Mohamed, M. S. Takriff, E. P. Chee, Inhibition of aluminum corrosion by phthalazinoneand synergisitic effect of halide ion in 1. 0 M HCl, Current Appl, Physics, 12 (2012), 325-330.
- A. S. Fouda, M. Abdallah, T. Y. Mohamed, Phenazone and aminophenazone as corrosion inhibitors for aluminum in HCl solution, Protection of Metals and Physical Chemistry of surfaces, 47 (6) (2011), 803-812.
- S. Safak, B. Duran, A. Yurt, G. Turkoglu, Schiff bases as corrosion inhibitors for aluminum in HCl solution, Corros. Sci, 54 (2012), 251-259.
- N. O. Eddy, H. M. Yahaya, E. E. Oguzie, Theoritcal and experimental studies on the corrosion inhibition potentials of some purines for aluminum in 0.1 M HCl, Journal of Advanced Research (2014).
- O. A. Hazazi, M. Abdallah, Prazole compounds as inhibitors for corrosion of Aluminum in Hydrochloric acid, Int. J. Electrochem Soc, 8 (6) (2013), 8138 – 8152.
- M. Abdallah, Tetradecyl-1, 2-Diol propen oxylates as inhibitors for corrosion of aluminum in hydrochloric acid. Bull. of Electrochem.: 16 (6) (2000), 258.
- M. Abdallah., H. E. Megahed, A. Y. El-Etre, M. A. Obied and E. M. Mabrouk, Polyamide compounds as corrosion inhibitors for corrosion of aluminum in oxalic acid solutions. Bull of Electrochemistry: 20 (6) (2004), 277.
- A. S. Fouda, M. Abdallah. I. S. Ahmed and M. Eissa, Corrosion inhibition of Al in H3PO4 solutions by ethanolamines, Arab. J. of Chem., 5 (3) (2012), 297-307.
- R. Yıldız, A. Döner, T. Doğan, İ. Dehri, Experimental studies of 2-pyridinecarbonitrile as corrosion inhibitors for mild steel in hydrochloric acid solution, Corros. Sci., 82 (2014), 125-132.
- M. Abdallah, I. Zaafarany, A. Fawzy, M. A. Radwan and E. Abdfattah, Inhibition of aluminum corrosion in hydrochroric acid by cellouse and chitosan, J. of Amer. Sci., 9 (4) (2013), 580-586.
- X. Li, S. Deng, H. Fu, Inhibition by tetradecylpyridinium bromide of the corrosion of aluminum in hydrochloric acid, solution, Corros. Sci., 53 (2011), 1529-1536.
- X. Li, S. Deng, X. Xie, Experimental and theoritcal study on corrosion inhibition of oxime compounds for aluminum in HCl solution, Corros. Sci., 81 (2014), 162-175.
- M. Abdallah, H. E. Megahed, M. A. Radwan and E. Abdfattah Polyethylene glycol compounds as corrosion inhibitors for aluminum in 0. 5 M hydrochloric acid solutions, J. of Amer. Sci., 8 (11) (2012), 49-55.
- M. Abdallah, I. Zaafarany, S. O. Al-Karanee and A. A. Abd Fattah, Antihypertensive drugs as an inhibitors for corrosion of aluminum and aluminum silicon alloys in aqueous solutions, Arab. J. of Chem., 5 (2) (2012), 225-234.
- S. M. Abd El Haleem, S. Abd El Wanees, E. E. Abd El Aal, A. Farouk, Factors affecting the corrosion behaviour of aluminum in acid solutions. I. Nitrogen and/or sulphur-containing organic compounds as corrosion inhibitors for Al in HCl solutions, Corros. Sci., 68 (2013), 1-13.
- A. S. Fouda, M. Abdallah and M. Eissa, Corrosion inhibition of aluminum in 1 M phosphoric acid solutions using some Chalcones derivatives and synergistic action with halide ions, African J. Pure Appl. Chem., 7 (12) (2013), 394-404.
- A. Y. El-Etre, Inhibition of aluminum corrosion using opuntia extract, Corros. Sci., 45 (2003), 2485-2495.
- M. A. Hegazy, M. Abdallah, M. K. Awad, M. Rezk, Three novel di- quaternary ammonium salts as novel corrosion inhibitors for API X65 steel pipeline in acidic solution. Part I: Experimental Results, Corros. Sci., 81( 2014), 54-64.
- M. Abdallah, M. E. Moustafa, Inhibition of acidic corrosion of carbon steel by some mono and bis azo dyes based on 1, 5- dihydroxy naphthalene, Annali Di Chimica, 94 (7-8) (2004), 601-611.
- M. Abdallah, Antibacterial Drugs as Corrosion inhibitors for corrosion of aluminum in hydrochloric acid solution, Corros. Sci., 46 (8) (2004), 1981-1996.
- M. H. M. Ahmed, M. M. H. Arief, w. M. Kairy, Preparation and characterization of some new surfactant drived from phenol, J of Oil, Soap and cosmetic, 60 (1-2) (2011), 17-22.
- O. A. Hazzazi, M. Abdallah E. A. M. Gad, Inhibition Effect of some cationic surfactants on the corrosion of carbon steel in sulphuric acid solutions: Surface and structural properties, Int. J. Electrochem Soc, 8 (2014), 2237 – 2253.
- I. Putilova, S. Balezin, I. N. Barannik, V. P. Bishop, Metall. Corros. Inhibt., Pergamon, Oxford, 1960, p. 196.
- M. Abdallah, A. Y. El-Etre, M. F. Moustafa, Amidopoly ethylamines as corrosion inhibitors for zinc dissolution in different acidic electrolyte, Port. Electrochimica Acta., 27 (5) (2009), 615-630.
- M. A. Hegazy, M. Abdallah, M. K. Awad, M. Rezk, Three novel di-quaternary ammonium salts as novel corrosion inhibitors for API X65 steel pipeline in acidic solution. Part I: Experimental Results, Corros. Sci., 81 (2014), 54-64.
- M. Sobhi, M. Abdallah., K. S. Khairou, Sildenafil citrate (Viagra) as a corrosion inhibitor for carbon steel in hydrochloric acid solutions, Monatshefte fur Chemie, 143 (2012), 1379-1387.
- A. S. Fouda, M. M. Farahat, M. Abdallah, Cephalosporin antibiotics as new corrosion inhibitor for nickel in HCl solution, Research on Chemical Intermediates, 40 (2014), 1249-1266.
- S. M. Abd El Haleem, M. Abdallah and A. I. Mead. Fatty acid ethoxylates from soya bean oils as corrosion inhibitors for copper in HNO3 solution. Proc. 8th European Symp. on Corrosion Inhibitors (8 SEIC) Ann Univ. Ferrara, N. S., V, Suppl, N. 10, 693 (1995).
PDF Version of the article |
Epub Version of the article |
Flash Version of the article |
| [qr-code size=”2″] | ||