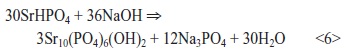For the first time, strontium hydrogen phosphate (SrHPO4) was electrocrystallized on titanium substrate by means of electrochemical deposition technique, and converted to strontium hydroxyapatite (Sr10(PO4)6(OH)2) to improve implant adhesion and bone mineralization. Brushite (calcium phosphate dihydrate CaHPO4·2H2O) and strontium hydrogen phosphate were co-electrocrystallized on titanium substrate. With increasing SrCl2 and decreasing CaCl2 in the solution, Sr concentration in the coating was increased. Calcium substitution by strontium ranged from 0 to 100 atomic percent, thus having significant effect on layer thickness, morphology, and composition. Layers containing brushite and strontium hydrogen phosphate were converted to calcium hydroxyapatite and strontium hydroxyapatite. Strontium hydroxyapatite was formed in the case of 100 percent SrCl2 substituting CaCl2. Surface morphology, chemical composition, and phase identification of the coatings were studied by scanning electron microscopy combined with energy dispersive spectrometry (SEM-EDXS) and by X-ray diffractometry (XRD). Effects of the varying Sr substitution on the microstructure and properties are discussed.
1 Introduction
Hydroxyapatite has received much attention and has been clinically applied in orthopaedics and dentistry due to its excellent biocompatibility. Hydroxyapatite (Ca10(PO4)6(OH)2), HAP, constitutes about 70 percent of natural human bone. HAP is osteoconductive and accelerates bone growth as well as attachment to the surface of implants. In addition, HAP improves fixation and lifetime of implants. Although mechanical strength is too poor for being used in load-bearing applications, HAP coatings on bioinert metallic prostheses were investigated to improve mechanical properties while maintaining biocompatibility [1–5]. Several methods for depositing HAP onto implant surfaces have been reported, for example, plasma spraying [5–9], sputtering [10–12], pulsed-laser deposition [13], sol-gel [14], electrophoresis [15], and electrochemical deposition [16–18].
Strontium, with a charge-to-size ratio similar to that of calcium, can readily substitute calcium in the lattice of HAP at any proportion. In some cases, substitution of Ca by Sr is reasonable as it plays an important role in treating osteoporosis and enhancing bone remineralisation since Sr is associated with a reduction of bone resorption, an increase in the formation of new bone, and a decrease in the risk of bone fracture [19]. Consequently, a number of Sr-containing materials have been tested as bone fillers, such as partially strontium-substituted hydroxyapatite Srx–HAP [20–23], tri-calcium phosphate [24] and CaSiO3 [25], as well as strontium-containing cement [26, 27]. In addition, strontium-containing toothpaste has been developed for enhancing remineralization of dental enamel [28], and recently, drugs such as strontium ranelate (SrR) [29] were suggested for use as a daily supplement to increase bone mineral density.
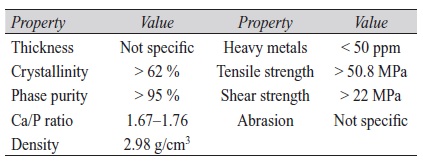
By soaking in sodium hydroxide solution at 80 °C, brushite (CaHPO4·2H2O) coatings are easily converted into HAP coatings. Brushite can be deposited electrochemically by applying current densities between 0.375 mA/cm2 and 6 mA/cm2 [18, 30–33]. Specifications of HAP coatings (Tab. 1) are found in the Food and Drug Administration guidelines as well as in ISO standards [6, 34, 35].
Tab. 1: Specifications of HAP coatings [6, 34, 35]
The coatings discussed here were electrocrystallized to form brushite with a subsequent treatment in sodium hydroxide solution to form HAP. Maintaining a constant (Ca+Sr)/P ratio of 1.67, selected volume ratios of SrCl2/CaCl2+SrCl2 were used to substitute calcium with strontium. The main objective of this research was to study the effects of this substitution on the formed calcium phosphate phase(s) as well as the coating properties. To our knowledge, no data are available on partially strontium-substituted hydroxyapatite coatings on titanium alloy substrates.
2 Experimental Procedure
2.1 Formation of Brushite
A single-compartment electrochemical cell was used for electrocrystallization. Titanium sheets served as cathodes and were cut into disks of 50 mm × 20 mm and a thickness of 0.8 mm. After cutting, the specimens were homogeneously grinded with 600 grit SiC abrasive paper and etched at room temperature for five minutes. The etching solution consisted of 180 ml/l nitric acid (65 %) and 23 ml/l hydrofluoric acid (42 %) in bi-distilled water. After rinsing with distilled water and ultrasonic cleaning with acetone for five minutes, the specimens were carefully airdried and weighed. A platinum sheet of 4 cm2 served as anode. It was ultrasonically cleaned in acetone prior to use. A saturated calomel electrode (SCE) was used as reference electrode along with a potentiostat/galvanostat instrument for electrochemical measurements.
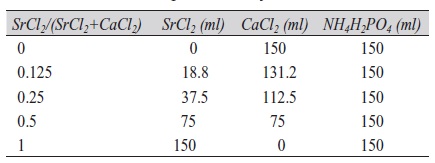
The electrolyte solution used for the electrocrystallization was prepared from three stock solutions: 0.167 M CaCl2, 0.167 M SrCl2, and 0.1 M NH4H2PO4. Different volumes of CaCl2 and SrCl2 were mixed with a constant volume of NH4H2PO4 by magnetic stirring for a total volume of 300 ml with constant (Ca+Sr)/P ratio of 1.67 in the mixed solution (Tab. 2). The pH value ranged from 4.2 to 4.6. Temperature was kept constant at (25±1) °C. Deposition was carried out galvanostatically for one hour at a current density of 1.5 mA/cm2. After coating, specimens were rinsed in bi-distilled water in order to remove residual electrolyte, then sprayed with ethanol, air-dried, and weighed to evaluate weight gain.
Tab. 2: Composition of electrolyte solution
2.2 Formation of hydroxyapatite
Samples were characterized as-deposited and after conversion to HAP. For conversion, the layers were soaked in 1 M NaOH solution for 60 min at 80 to 90 °C. Subsequently, the specimens were rinsed in water and ethanol, air-dried, and weighed to evaluate weight loss.
2.3 Characterization of coatings
Crystalline phases of the specimens were examined by X-ray diffraction (XRD, D5000, Siemens) with monochromatic Cu Kα radiation (λ = 0.154052 nm). The current and voltage of the X-ray tube were 15 mA and 40 kV, respectively. Scanning angles of specimens varied from 10° to 70°. Crystallite size was computed applying Scherrer’s equation. Coating morphology and microstructure were investigated by scanning electron microscopy (NEON 40EsB, Zeiss). In addition, elemental composition was determined by energy-dispersive X-ray spectrometry (EDXS, EDAX). Cross-sections were embedded in epoxy prior to cutting, grinding, and polishing to protect the porous brittle layer during preparation.
3 Results and discussion
The process of electrochemical deposition of calcium phosphate and strontium phosphate differs completely from conventional metal electrodeposition. It includes electrolytic and electrophoretic reactions. A layer is deposited when the pH-value-depending solubility product of the phosphates is exceeded. Due to super-saturation, precipitates are formed and crystallize under the influence of the applied electrical field. The resulting layer is a mixture of calcium phosphate and strontium phosphate depending on electrolyte composition [36–38].
Crystallization of dicalcium phosphate dihydrate, for example, can be a combination of the following reactions:
- Ionization of CaCl2, SrCl2, and NH4H2PO4, forming ions such as Ca2+, Sr2+, Cl–, NH4+, and H2PO4–.
- Reduction of water on the cathodic surface (titanium) under the influence of the electric field, producing hydroxide ions:
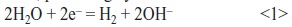
- The reaction in Equation <1> results in high pH in the vicinity of the cathode. Local pH, measured using an in situ pH-microsensor, ranged from 6.7 to 7.4, depending on applied current density [38]. High pH promotes the following reaction:
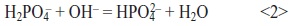
- Two-dimensional nucleation due to local supersaturation in the vicinity of the cathode is followed by crystal growth of brushite and/or strontium monohydrogen phosphate on the titanium surface according to the following equilibrium reaction:
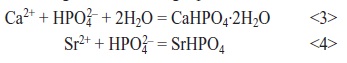
The effect of Sr substitution was studied on as-deposited layers and layers after the conversion to HAP. For this, different amounts (0 to 100 volume percent) of CaCl2 were substituted by SrCl2 at constant (Ca+Sr)/P ratio and constant deposition conditions (Tab. 2). Obtained weight gains are given in Table 3, compositions of the coatings in Figure 1. A slight decrease in weight gain was observed with increasing substitution of Ca by Sr. Considering the significant difference in atomic weight between Ca (40.08) and Sr (87.62), strontium remarkably hinders electrocrystallization of Ca phosphate and/or Sr phosphate phases. This is confirmed by the striking decrease of layer thickness with increasing Sr content.
Tab. 3: Effect of SrCl2/(SrCl2+CaCl2) volume ratio in electrolyte on weight gain

Fig. 1: Effect of SrCl2/(SrCl2+CaCl2) volume ratio on coating composition
As confirmed by XRD, the formed layer comprises dicalcium phosphate dihydrate CaHPO4∙2H2O (brushite) and strontium hydrogen phosphate SrHPO4. Conversion to HAP and strontium hydroxyapatite was carried out in hot sodium hydroxide solution according to the following reactions:
and
Experiments revealed a weight loss of 35 percent for calcium hydroxyapatite (41.6 % in theory) and of 23 percent for strontium hydroxyapatite (19.4 % in theory), which is too low for HAP and too high for strontium hydroxyapatite.
Similar to weight loss, Sr, Ca, and P concentrations in the coating before and after sodium hydroxide treatment can be predicted. In as-deposited films, they should amount to 8 atomic percent for Ca and P in brushite and 16.6 atomic percent for Sr and P in strontium hydrogen phosphate. After conversion to HAP, 29.4 atomic percent of Ca should be present in calcium hydroxyapatite and 29.4 atomic percent of Sr in pure strontium hydroxyapatite as well as 15.8 atomic percent of P. Under appropriate conditions, the absolute error of the quantitative analysis of the layers by EDXS is about 1 atomic percent. However, the accuracy of the results reported is limited due to the surface roughness of the samples. A more reasonable explanation is the formation of amorphous oxides and different hydration stages of the electrocrystallized phosphates. This is consistent with the low weight loss after calcium hydroxyapatite formation and the high weight loss after strontium hydroxyapatite formation.
Despite these deviations, Sr content in the coating rises as expected with increasing SrCl2 in the electrolyte, Ca content decreases and P content remains nearly constant. A notable incorporation of strontium into the HAP coatings was confirmed by EDXS and XRD for a SrCl2/(SrCl2+CaCl2) ratio of 0.5.
As-deposited brushite and strontium hydrogen phosphate layers as well as the converted layers were characterized using XRD, SEM, and EDXS. Crystallite sizes for as-deposited and converted layers derived from XRD results are given in Figure 2. The crystallite size of the as-deposited layers decreases with increasing Sr concentration. Due to the conversion treatment, the crystallite size of the hydroxyapatite layers drops remarkably but remains nearly constant for a SrCl2/(SrCl2+CaCl2) ratio up to 0.5. The change after conversion of layers with complete substitution of Ca by Sr is relatively small. The crystalline phase is confirmed as HAP except for complete substitution, in this case the formed phase is strontium hydroxyapatite.
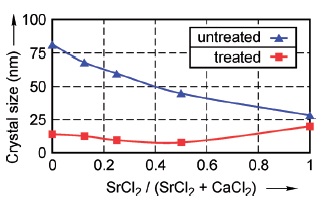
Fig. 2: Effect of SrCl2/(SrCl2+CaCl2) volume ratio on crystal size for as-deposited (blue triangles) and converted (red squares) layers
Figure 3 shows SEM images of cross-sections of Sr-containing electrocrystallized calcium phosphate dihydrate layers on titanium substrates. The nanometer-sized crystals form flat crystal aggregates growing on the substrate surface in randomly inclined stacks. With increasing Sr content, packing density of the crystal aggregates seems increased. This is confirmed by SEM images of the coating surfaces (Fig. 4). The plate-like crystal aggregates are stacked together and show good bonding to the surface of the titanium substrate. At high Sr substitution, the stacks of crystal aggregates appear to be reduced in size and to be arranged more densely. This is confirmed by the surface appearance of the converted HAP coatings which seems to be unchanged by the soaking treatment (Fig. 5).

Fig. 3: Cross-sections of electrocrystallized strontium-substituted calcium phosphate dihydrate on titanium; SrCl2/(SrCl2+CaCl2) ratios: 0 (a), 0.125 (b), 0.25 (c), and 0.5 (d)

Fig. 4: Surfaces of strontium-substituted calcium phosphate dihydrate; SrCl2/(SrCl2+CaCl2) ratios: 0 (a), 0.125 (b), 0.25 (c), and 0.5 (d)

Fig. 5: Surfaces of strontium-substituted hydroxyapatite; SrCl2/(SrCl2+CaCl2) ratios: 0 (a), 0.125 (b), 0.25 (c), and 0.5
At complete Sr substitution, strontium hydrogen phosphate crystals form needle-like aggregates which are arranged in clusters and flower-like formations (Fig. 6a). Transformation to strontium hydroxyapatite decreases the size of the crystal aggregates (Fig. 6b).
Mechanical properties of the deposited layers can hardly be evaluated due to the brittleness of the material. However, influence on the friction coefficient can be studied (Fig. 7). Obtained results show that the friction coefficient (μ) is increased at low SrCl2/(SrCl2+CaCl2) ratios (up to 0.25) and decreased at high SrCl2/(SrCl2+CaCl2) ratios (above 0.5). This is in good correlation with the denser and finer-grained microstructure of the layers.

Fig. 6: Surfaces of strontium hydrogen phosphate (a) and strontium hydroxyapatite after conversion (b)

Fig. 7: Friction coefficient of hydroxyapatite at low (a) and high (b) Sr substitution compared to unsubstituted calcium hydroxyapatite (baseline) and uncoated titanium
4 Summary
Strontium hydrogen phosphate was electrocrystallized by electrochemical deposition and converted to strontium hydroxyapatite. Brushite and strontium hydrogen phosphate were co-electrocrystallized on titanium substrates. The morphology of as-deposited layers is characterized by plate-like crystals of brushite. Calcium-free layers are composed of acicular aggregates. The effect of calcium substitution by strontium on the composition of the as-deposited and converted layers was confirmed by EDX. The crystallite size of brushite is decreased with increasing substitution by Sr and by conversion to hydroxyapatite. High Sr-substituted hydroxyapatite shows a reduced friction coefficient, compared to calcium hydroxyapatite.
References
- L. L. Hench: Bioceramics: From Concept to Clinic; J. Am. Ceram. Soc., 74 (1991) 7, 1487–1510
- M. C. Kuo, S. K. Yen: The process of electrochemical deposited Hydroxyapatite coatings on biomedical titanium at room temperature; Materials science and Engineering C 20 (2002), 153–160
- M. Jarcho: Calcium phosphate ceramics as hard tissue prosthetics; Clin Orthop Relat R. 157 (1981), 259
- S. D. Cook, K. A. Thomas, J. F. Kay, M. Jarcho: Hydroxyapatite coated titanium for orthopedic implant applications. Clin. coated titanium for orthopedic implant applications; Clin. Orthop. Relat. Res. 230 (1988), 225–243
- K. de Groot, R. Geesink, C. P. Klein, P. Serekian: Plasma sprayed coatings of hydroxylapatite; Journal of Biomedical Materials Research, 21 (1987), 1375–1381
- L. Sun, C. C. Berndt, K. A. Gross, A. Kucuk: Material fundamentals and clinical performance of plasma-sprayed Hydroxyapatite coatings: a review; J Biomed Mater Res (Appl Biomater) 58 (2001), 570–92
- V. Sergo, O. Sbaizero, D. R. Clarke: Mechanical and Chemical Consequences of the Residual Stresses in Plasma Sprayed Hydroxyapatite Coatings; Biomaterials, 18 (1997), 477–482
- C. F. Feng, K. A. Khor, E. J. Liu, P. Cheang: Phase transformation in plasma-sprayed Hydroxyapatite coatings; Scripta Mater. 42 (1999) 1, 103–109
- S. R. Radin, P. Ducheyne: The effect of plasma sprayed induced changes in characteristics on the in vitro stability of calcium phosphate ceramic. J Mater Sci: Mater Med 3 (1992), 33–42
- Y. Yang, K. H. Kim, J. L. Ong: A review on calcium phosphate coatings produced using a sputtering process – an alternative to plasma spraying; Biomaterials 26 (2005), 327–337
- K. van Dijk, H. G. Schaeken, J. G. G. Wolke, J. A. Jansen: Influence of annealing temperature on RF-magnetron sputtered calcium phosphate coating; Biomaterials 17 (1998), 159–163
- K. Yamashita, T. Arashi, K. Kitagaki, S. Yamada, T. Umegaki: Preparation of Apatite Thin Films through Sputtering from Calcium Phosphate Glasses; J. Am. Ceram. Soc. 77 (1994) 9, 2401–2407
- C. K. Wang, J. H. Chern Lin: Structure characterization of pulsed laser-deposition hydroxyapatite film on titanium substrate; Biomaterials 18 (1997), 1331–1338
- C. S. Chai, B. Ben-Nissan: Bioactive nanocrystalline sol-gel hydroxyapatite coatings; J Mat Scie: Mat Med 10 (1999) 8, 465–469
- R. Damodaran, B. M. Moudgil: Electrophoretic deposition of calcium phosphates from non-aqueous media; Colloid. Surf. A 80 (1993), 191–195
- M. Shirkhanzadeh: Calcium phosphate coatings prepared by electrocrystallization from aqueous electrolytes; J. Mater. Sci. Med. 6 (1995), 90–93
- M. Manso, C. Jimenez, C. Morant, P. Herrero, J. M. Martinez-Duart: Electrodeposition of hydroxyapatite coatings in basic conditions; Biomaterials 21 (2000), 1755–1761
- X. Hou, X. Liu, J. Xu, J. Shen, X. Liu: A self-optimizing electrodeposition process for fabrication of calcium phosphate coatings; Mater Lett 50 (2001), 103–107
- J. Buehler, P. Chappuis, J. L. Saffar, Y. Tsouderos, A. Vignery: Strontium ranelate inhibits bone resorption while maintaining bone formation in alveolar bone in monkeys (Macaca fascicularis); Bone 29 (2001) 2, 176–179
- W. C. Xue, H. L. Hosick, A. Bandyopadhyay, S. Bose, C. X. Ding, K. D. K. Luk: Preparation and cell-materials interactions of plasma sprayed strontium-containing hydroxyapatite coating. Surf Coat Tech 201 (2007) 8, 4685–4693
- A. L. Oliveira, R. L. Reis, P. Li: Strontium-substituted apatite coating grown on Ti6Al4V substrate through biomimetic synthesis; J Biomed Mater Res B 83B (2007) 1, 258–265
- A. Bigi, E. Boanini, C. Capuccini, M. Gazzano: Strontium-substituted hydroxyapatite nanocrystals; Inorg Chim Acta 360 (2007) 3, 1009–1016
- B. Donazzon, G. Dechambre, J. L. Lacout: Calcium-strontium hydroxyapatite: hydrothermal preparation; Ann Chim Sci Mater 23 (1998), 53–56
- S. J. Saint-Jean, C. L. Camire, P. Nevsten, S. Hansen, M. P. Ginebra: Study of the reactivity and in vitro bioactivity of Sr-substituted a-TCP cements; J Mater Sci Mater Med 2005, 16(11), 993–1001
- C. T. Wu, Y. Ramaswamy, D. Kwik, H. Zreiqat: The effect of strontium incorporation into CaSiO3 ceramics on their physical and biological properties; Biomaterials 28 (2007) 21, 3171–3181
- G. X. Ni, K. Y. Chiu, W. W. Lu, Y. Wang, Y. G. Zhang, L. B. Hao, Z. Y. Li, W. M. Lam, S. B. Lu, K. D. K. Luk; Strontium-containing hydroxyapatite bioactive bone cement in revision hip arthroplasty. Biomaterials 27 (2006) 24, 4348–4355
- D. G. Guo, K. W. Xu, X. Y. Zhao, Y. Han: Development of a strontiumcontaining hydroxyapatite bone cement; Biomaterials 26 (2005) 19, 4073–4083
- A. Surdacka, J. Stopa, L. Torlinski: In situ effect of strontium toothpaste on artificially decalcified human enamel; Biol Trace Elem Res 116 (2007), 147–153
- J. Y. Reginster: Strontium ranelate in osteoporosis; Curr Pharm Design 8 (2002) 21, 1907–1916
- S. K. Yen, C. M. Lin: Cathodic reactions of electrolytic Hydroxyapatite coatite on pure titanium; Mat Chem Phys 77 (2002), 70–76
- Y. Y. Zhang, J. Tao, Y. C. Pang, W. Wang, T. Wang: Electrochemical deposition of hydroxyapatite coatings on titanium; Trans. Nonferrous Met. Soc. China, 16 (2006), 633–637
- S. H. Wang, W. J. Shih, W.-L. Li, M. H. Hon, M. C. Wang: Morphology of calcium phosphate coatings deposited on Ti-6Al-4V substrate by an electrolytic method under 80 Torr; J Europ Ceram Soc 25 (2005), 3287–3292
- E. A. Abdel-Aal, D. Dietrich, S. Steinhäuser: Electrocrystallization of Calcium Phosphate Coatings on Titanium at Different Current Densities; Surf Coat Techn, 202 (2008), 5895–5900
- FDA: Calcium phosphate (Ca-P) coating draft guidance for preparation of FDA submissions for orthopedic and dental endosseous implants; Washington, DC: Food and Drug Administration (1992), 1–14
- ISO. Implants for surgery: coating for hydroxyapatite ceramics; (1996), 1–8
- C. Chu, X. Xue, J. Zhu, Z. Yin: In vivo study on biocompatibility and bonding strength of Ti/Ti-20 vol.% HA / Ti-40 vol. % HA functionally graded biomaterial with bone tissues in the rabbit; Mat Sci Eng A 429 (2006), 18–24
- N. Dumelie, H. Benhayoune, C. Rousse-Bertrand, S. Bouthors, A. Perchet, L. Wortham, J. Douglade, D. Laurent-Maquin, G. Balossier: Characterization of electrodeposited calcium phosphate coatings by complementary scanning electron microscopy and scanning-transmission electron microscopy associated to X-ray microanalysis; Thin Solid Films 492 (2005), 131–139
- J. M. Zhang, C. J. Lin, Z. D. Feng, Z. W. Tian: Mechanistic studies of electrodeposition for bioceramic coatings of calcium phosphates by an in situ pH-microsensor teachnique; J Electroanal Chem 452 (1998), 235–240
PDF Version of the article |
Flash Version of the article |
|
| [qr-code size=”2″] | ||