The inhibitive effect of Curry leaves extract (CLE) on the corrosion behavior of aluminum in sulfuric acid (pH = 3) was investigated by potentiodynamic polarization and electrochemical impedance spectroscopy (EIS) techniques in the temperature range of 30 °C to 50 °C. The study was done by varying the concentrations of inhibitor from 0.05 g L−1 to 0.4 g L−1. The surface morphology was studied using scanning electron microscopy (SEM) and Energy-dispersive X-ray spectroscopy (EDX). Inhibition efficiency was found to increase with increase in inhibitor concentration and decrease with increase in temperature. CLE acted as an anodic type inhibitor at lower concentrations of inhibitor and behaved as a mixed type at higher concentrations of inhibitor and underwent both physisorption and chemisorption on the surface of the metal and followed the Langmuir adsorption isotherm.
1. Introduction
Aluminum is one of the most important metals used in industrial and domestic purposes due to its excellent electrical conductivity, good forming properties, low cost and other relatively noble properties. It is widely used in aerospace, household articles, industries, reaction vessels, pipes, machinery and chemical batteries [1-3]. The applications of aluminum and its alloys are often possible because of the natural tendency of aluminum to form a passivating oxide layer. However in aggressive media, the passivating layer will be destroyed and corrosive attack takes place. The protection of aluminum and its oxide films against the corrosive action of sulfate ions have been extensively studied [4-5].
Sulfuric acid in the pH range of 2-4.5 is mainly used for pickling, chemical and electrochemical etching of aluminum and its alloys [6]. A useful method to protect metals and alloys deployed in service in aggressive environments against corrosion is the addition of inhibitors. A number of organic compounds are known to be applicable as corrosion inhibitors for aluminum and its alloys in acidic environments [7-9]. Such compounds mainly contain nitrogen, oxygen or sulfur in a conjugated system and function via adsorption of the molecules on the metal surface creating a barrier to corrodent attack. The adsorption bond strength is dependent on the composition of the metal, corrodent, inhibitor structure and its concentration. Despite of broad spectrum of organic compounds, the choice of appropriate inhibitor for a particular application is restricted by several factors. These include increased environmental awareness and the need to promote environment friendly processes.
There exists a need to develop a new class of corrosion inhibitors with low toxicity and good efficiency. The exploration of natural products of plant origin as non-expensive eco-friendly inhibitor is an essential field study [10-13]. These plant products are not only eco- friendly, but also readily available, cost effective renewable source of materials and contain organic compounds such as amino acids, tannins and alkaloids which have inhibitive effect. The principle of inhibition is the adsorption of phytomolecules in the plant on the surface of the metal resulting in the replacement of water molecules at the metal surface. Till date lots of works have been reported for using natural product as corrosion inhibitors [14-16] for aluminum in acidic media. Investigation of natural inhibitors is particularly interesting because they are non-expensive, ecologically acceptable and possess no threat to the environment.
As a part of our research work with natural inhibitors for the corrosion control of aluminum and its alloys [17-19], we report herein, the results of utility of Curry leaves extracts (CLE) for the corrosion control of aluminum in sulfuric acid medium of pH=3.
Curry plant (Murraya koenigii) is an important leafy vegetable. Its leaves are widely used in Indian cookery for flavoring foodstuffs. The leaves have a slightly pungent, bitter and feebly acidic taste, and they retain their flavor and other qualities even after drying. Curry leaf is also used in many of the Indian Ayurveda and Unani prescriptions. Its nutritional value benefits both the young and the old alike. CLE is composed of numerous organic heterocyclic compounds. Major constituent are reported to be Terpene-4-ol, β–caryophylene and β–elemene [20].
2. Experimental
2.1. Material
The composition of aluminum is: Si: 0.467%, Fe: 0.163%, Mg: 0.530% and aluminum: Balance. Cylindrical test coupons of 10 mm diameter and approximately 20 mm height were machined from the rods of aluminum and metallographically mounted up to 10mm height using cold setting resin. The exposed flat surface of the mounted part was polished as per standard metallographic practice – belt grinding followed by polishing on emery papers (in the range of 600-2000) and finally disc polished using levigated alumina abrasive.
2.2. Preparation of medium
The stock solution of 2 M sulfuric acid was prepared by using analar grade sulfuric acid and double distilled water. Standardization was done by volumetric method. From the standard solution, sulfuric acid solution of required concentration was prepared as and when required. Experiments were carried out at different temperatures using a calibrated thermostat under unstirred conditions.
2.3. Preparation of the inhibitor (CLE)
Curry leaves extract (CLE) was prepared by literature method [20]. The Curry leaves were cut into pieces. These were completely air-dried at room temperature for 4 weeks. The dried leaves were pulverized into fine powder using a domestic mixer grinder. 40 g of the powder was boiled in 500 mL of distilled water for 5 h. It was filtered using a piece of clean white cotton gauze. The filtrate was evaporated to complete dryness at 40 ℃, producing a fine sweet smelling and chocolate color solid residue. The extraction process was repeated 4 times and the solid residue weighed after extraction was pooled together in an air and water-proof container and kept in a refrigerator at 4 ℃. Aqueous solution of the inhibitor of required concentration was prepared freshly as and when required.
2.4. Characterization of inhibitor
FTIR spectrum of the dried sample was recorded using spectrophotometer (Shimadz model) in the frequency range of 4000 to 400 cm–1 using KBr pellet technique.
2.5. Electrochemical measurements
Electrochemical studies were carried out using a potentiostat (CH600D-series, U.S.model with CH Instruments beta software). Studies were done by using conventional three electrode Pyrex glass cell with platinum as counter electrode and SCE as reference electrode. Working electrode was aluminum.
Finely polished aluminum specimen was exposed to sulfuric acid (pH=3) at different temperatures (30 ℃ to 50 ℃) and allowed to establish a steady state open circuit potential by immersing the electrodes in corrosive medium for 30 minutes.
2.5.1. Potentiodynamic polarization (PDP) measurements
The potentiodynamic polarization measurements were done by polarizing the specimen from −250 mV cathodically to +250 mV anodically with respect to open circuit potential at the scan rate of 1 mV sec−1. Corresponding potential –current plots were developed simultaneously. The experiments were carried out in absence and presence of different concentrations of inhibitor.
2.5.2. Electrochemical impedance spectroscopy (EIS) studies
In electrochemical impedance method a small amplitude ac signal of 10 mV and frequency spectrum from 100 k Hz to 0.01 Hz was impressed at the O.C.P. and resulting impedance data was analyzed using the Nyquist plot.
Electrochemical impedance method was immediately carried out after potentidynamic polarization method without further surface treatment. In both measurements, minimum of 3-4 trails were done and the average of best three was reported.
2.6. Scanning electron microscopy (SEM) and Energy –dispersive X-ray spectroscopy (EDX) analysis
The surface morphology of aluminum surface, in absence and presence of the inhibitor was studied by immersing the material in sulfuric acid medium (pH=3) for 2 h. using JEOL JSM-6380L Analytical scanning electron microscope in the magnification of 500 X. EDX investigation was carried out to identify the species which were responsible for the corrosion of the metal sample.
3. Results and Discussion
3.1. Fourier transform infrared (FTIR) spectroscopy of CLE
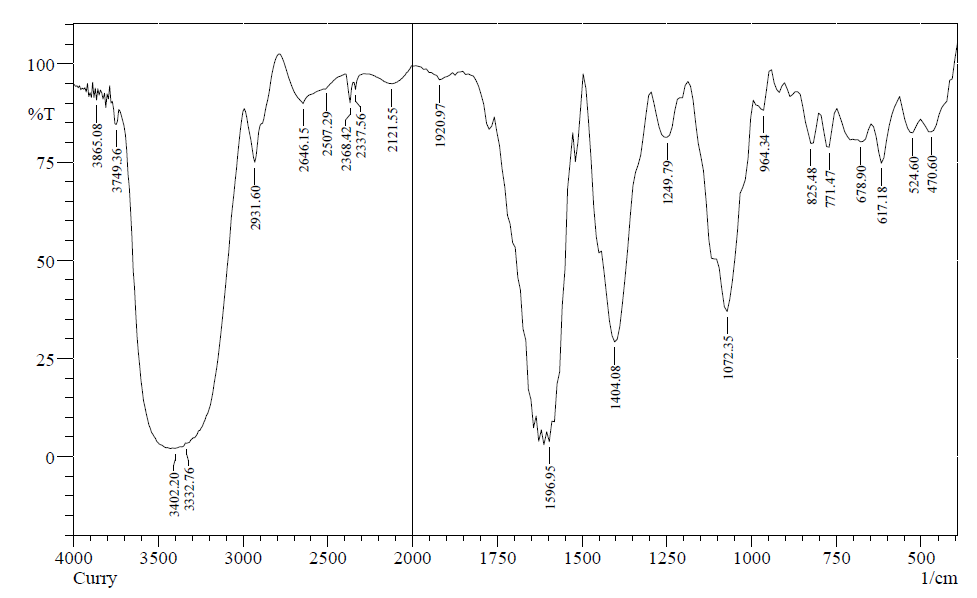
Figure 1 shows the FTIR spectrum of aqueous extracts of CLE. Absorption at 3425.34 cm− 1 can be assigned to stretching frequency of O-H. The peak at 2931 cm− 1 can be assigned to stretching mode of C–H group of alkanes. The 1404.04 cm− 1 band is assigned to the C–H bending. The adsorption band at 1596 cm− 1 is assigned to the C=C stretch of alkenes.
3.2. Potentiodynamic polarization (PDP) measurements
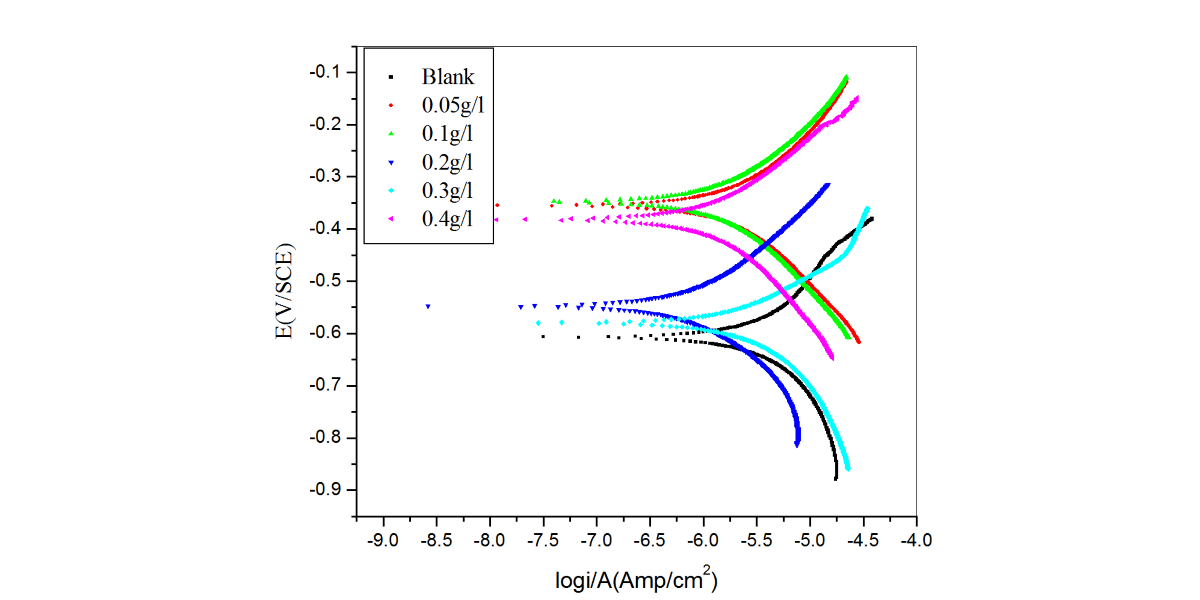
Figure 2 shows the potentiodynamic polarization plot for the corrosion of aluminum in sulfuric acid (pH=3) containing different concentrations of CLE at 30 °C.
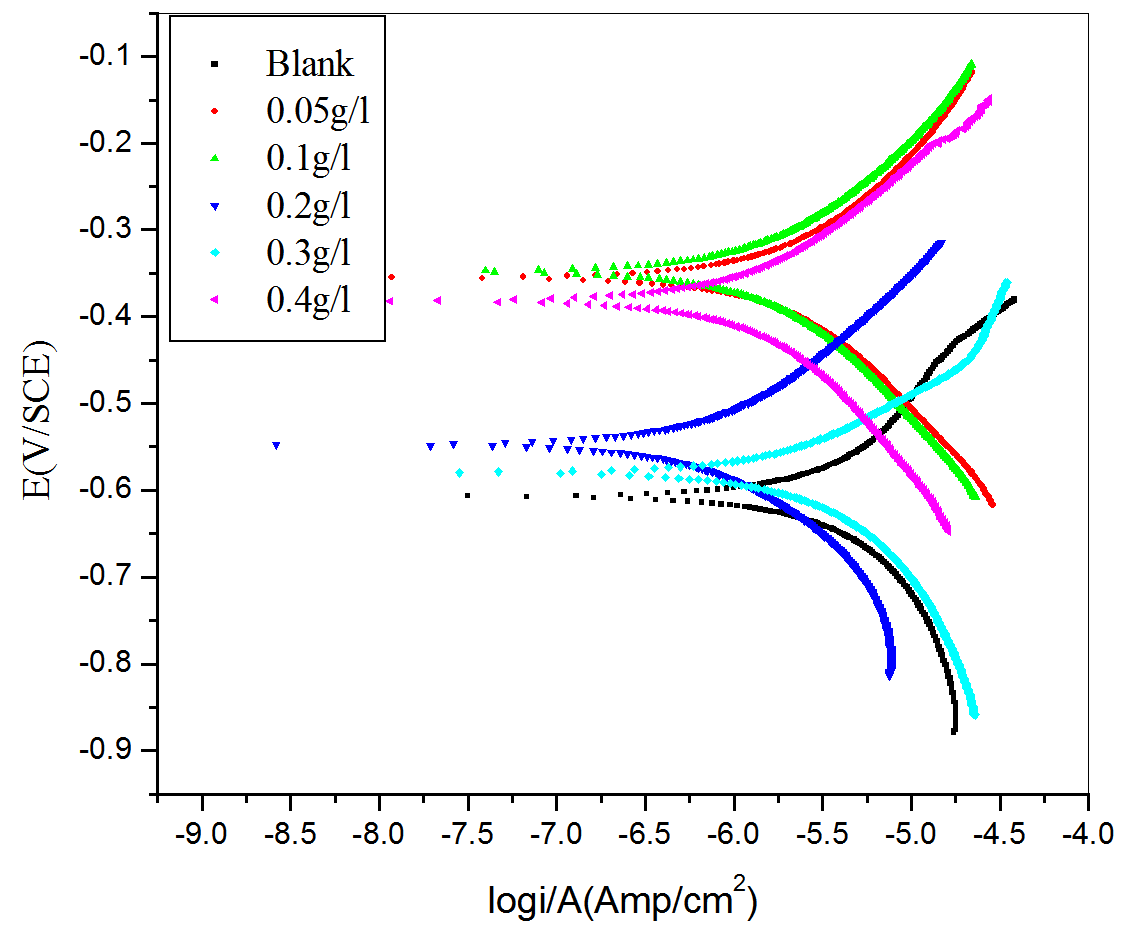
Fig. 2: Potentiodynamic polarization plots for the corrosion of aluminum containing different concentrations of CLE in H2SO4 (pH=3) at 30 oC
Valuable potentiometric polarization parameters like, corrosion current density (icorr.), corrosion potential (Ecorr.), anodic Tafel slope (βa) and cathodic Tafel slope (−βc) were obtained from the potentiodynamic polarization plot. Corrosion rate was calculated using equation (1) [21].
Equation 1

where 3270 is a constant that defines the unit of corrosion rate, icorr = corrosion current density in A cm–2, ρ = density of the corroding material (in g cm–3), M = Atomic mass of the metal, and Z = Number of electrons transferred per metal atom. For aluminum Z= 27 and ρ = 2.7.
Percentage inhibition efficiency was calculated using the equation (2).
Equation 2:
![]()
where
Equation 3:

where icorr. and icorr. (inh) are the corrosion current densities in the absence and in the presence of inhibitor, respectively.
The experiments were repeated at four other temperatures (35 °C to 50 °C) Corrosion potential (Ecorr.), corrosion current density (icorr.), corrosion rate (CR) and percentage inhibition efficiency (% I.E) were determined. The results of the potentiodynamic polarization measurement for corrosion of aluminium aluminum in sulfuric acid (pH=3) containing different concentrations of inhibitors at different temperatures are tabulated in Table 1.
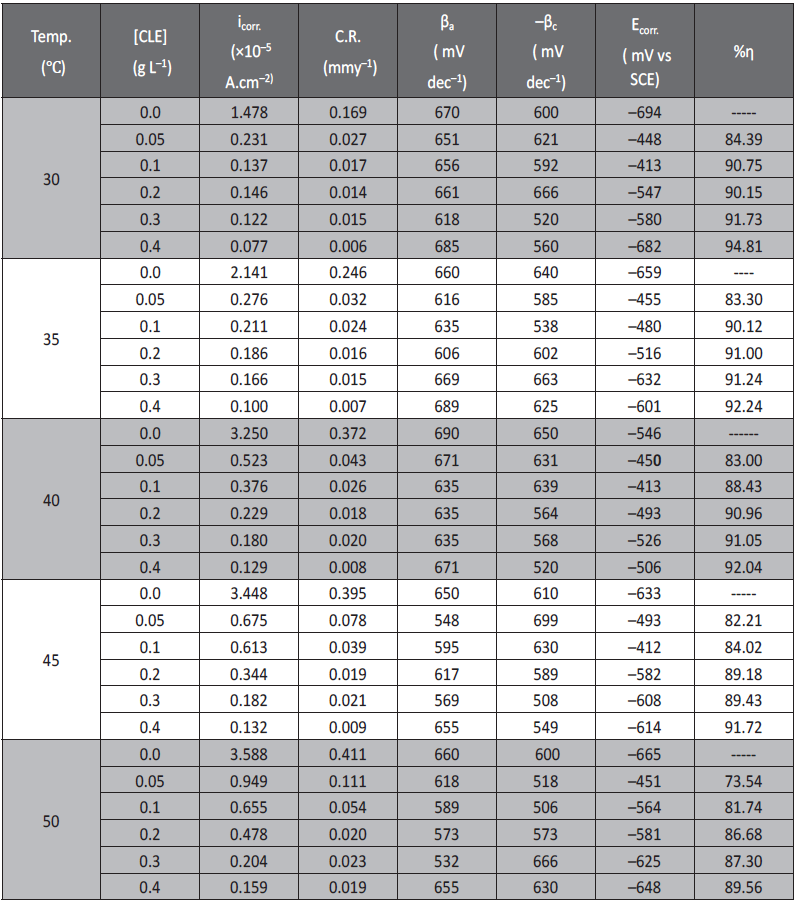
Tab. 1: Results of potentiodynamic polarization measurements for the corrosion of aluminum in sulfuric acid (pH =3) containing different concentrations of CLE different temperatures
It was observed that with increase in the concentrations of the CLE corrosion current density decreased. Inhibition efficiency increased with increasing inhibitor concentrations. The observed inhibition efficiency of CLE may be due to the adsorption of its components on the aluminum surface. The adsorbed molecules may isolate the metal surface from the aggressive medium leading to decreasing the corrosion rate [21].
The anodic and cathodic Tafel slopes remained almost unchanged for uninhibited and inhibited solutions. This indicates that the inhibitive action of CLE may be due to simple blocking of the available surface area on the surface of the metal. In other words, the inhibitor decreases the surface area available for hydrogen evolution without affecting the reaction mechanism and only causes inactivation of part of the surface [21-23].
There was measurable positive shift in corrosion potential (Ecorr) after the addition of inhibitor at lower concentrations (0.05-0.2g L−1). According to Li et al [24-25], if the displacement in corrosion potential is more than ±85 mV with respect to corrosion potential of the blank, then the inhibitor can be considered distinctively as a cathodic or anodic type. However, the maximum displacement in the present investigations for aluminum in sulfuric acid at lower concentrations of inhibitor was more than that of 85 mV, towards the positive direction. This observation suggests that constituents of inhibitor molecule may act as an anodic type and brings the anodic reaction under control [21]. But at higher concentrations of inhibitor the shift in corrosion potential was less than ±85 mV . This observation suggested that constituents of inhibitor molecule acts as mixed type and brings both anodic and cathodic reactions under control.
3.3. Electrochemical impedance spectroscopy (EIS)
In order to gain more information about the corrosion inhibition phenomenon, electrochemical impedance spectroscopy measurements were carried out for the aluminum in sulfuric acid (pH = 3) in the presence and absence of aqueous extracts of CLE at different temperatures. Figure 3 represent the Nyquist plot of aluminum in the sulfuric acid (pH=3) containing various concentrations of aqueous extracts of CLE at 30 °C. Similar plots are obtained at other temperatures.
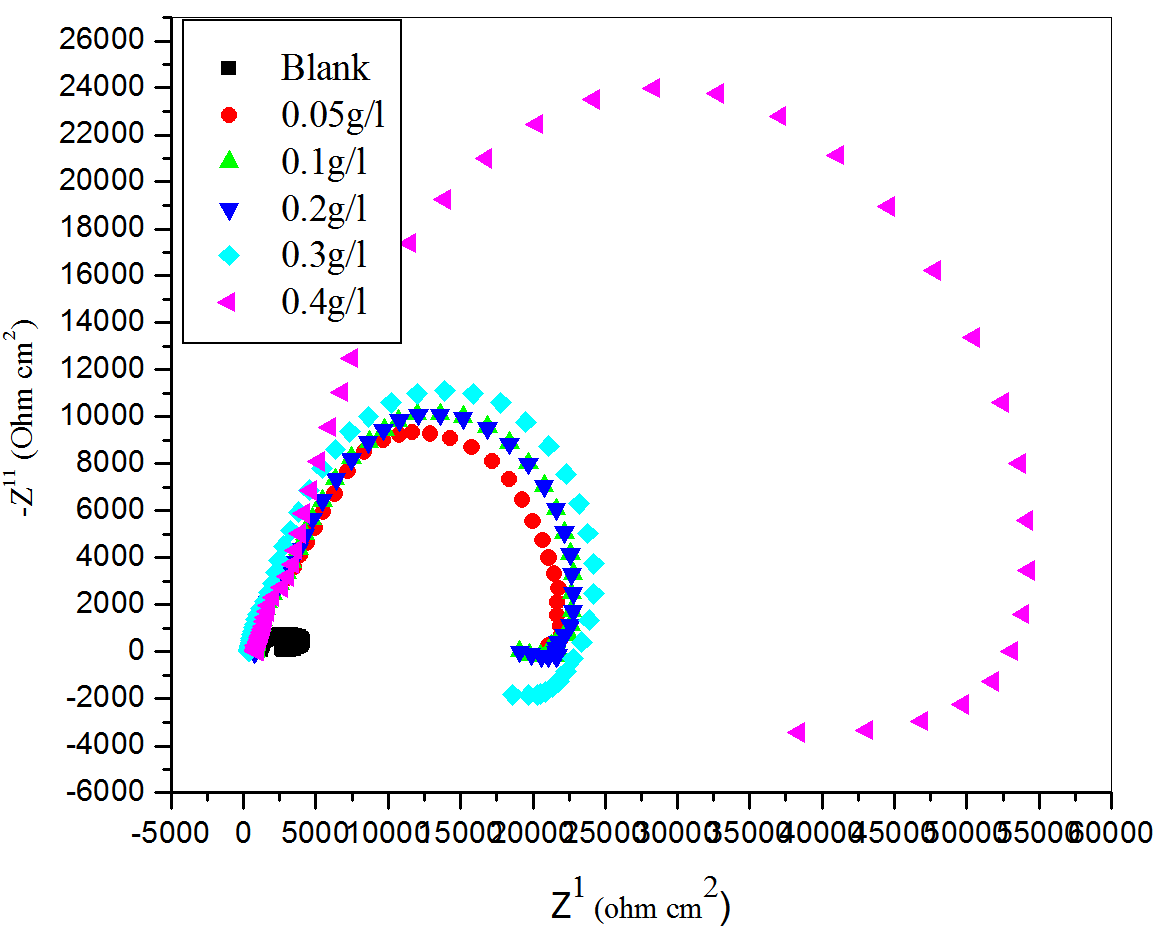
Fig. 3: Nyquist plots for the corrosion of aluminum in H2SO4 (pH =3) containing different concentrations of CLE at 30 ℃.
As can be seen from Figure 3 the impedance diagram show semicircles, indicating that the corrosion process is mainly charge transfer controlled. The presence of inhibitor increased the impedance but did not change other aspects of the behavior. These observations suggested that inhibitor did not alter the mechanism of electrochemical reactions responsible for corrosion. It inhibits corrosion primarily through its adsorption on the metal surface [26]. The impedance plot was with a depressed capacitive loop at high-frequency range (HF) whose diameter increased with increase in inhibitor concentration, followed by an inductive loop at low-frequency (LF) region. Similar impedance plots have been reported in the literature for the corrosion of pure aluminum and aluminum alloys in various electrolytes [27–28].
The high frequency capacitive loop could be assigned to the charge transfer of the corrosion process and to the formation of oxide layer [29]. The oxide film is considered to be a parallel circuit of a resistor due to the ionic conduction in the oxide film and a capacitor due to its dielectric properties. According to Brett [30], the capacitive loop is corresponding to the interfacial reactions, particularly, the reaction of aluminum oxidation at the metal/oxide/electrolyte interface. The process includes the formation of Al+ ions at the metal/oxide interface, and their migration through the oxide/solution interface where they are oxidized to Al3+. At the oxide/solution interface, OH− or O2− ions are also formed. The fact that all the three processes are represented by only one loop could be attributed either to the overlapping of the loops of processes, or to the assumption that one process dominates and, therefore, excludes the other processes [31]. The other explanation offered to the high frequency capacitive loop is the oxide film itself. This was supported by a linear relationship between the inverse of the capacitance and the potential found by Bessone et al.s [32] and Wit and Lenderink [31]. The origin of the inductive loop has often been attributed to surface or bulk relaxation of species in the oxide layer [33]. The LF inductive loop may be related to the relaxation process obtained by adsorption and incorporation of sulphate ions, oxide ion and charged intermediates on and into the oxide film. The observed deviations from perfect semicircles are often referred to the frequency dispersion of interfacial impedance. This anomalous phenomenon is interpreted as reflecting inhomogeneity of electrode surfaces arising from surface roughness or interfacial origin such as those found in adsorption processes.
As shown in Figure 3 the obtained semicircles in absence or in presence of inhibitor were depressed. Deviation of this kind is referred to as frequency dispersion. It has been attributed to inhomogeneities of solid surfaces. Mansfeld et al. [21] have suggested an exponent n in the impedance function as a deviation parameter from the ideal behavior. By this suggestion, the capacitor in the equivalent circuit can be replaced by a so-called constant phase element (CPE) that is a frequency-dependent element and related to surface roughness [31].
The impedance function of a CPE has the following equation
Equation 4:
![]()
where
Equation 5:
![]()
where the amplitude Yo and n are frequency independent, and ω is the angular frequency for which –Z11 reaches its maximum values, n is dependent on the surface morphology, with values,−1 ≤ n ≤1. Yo and n can be calculated by the equations proved by Mansfeld et al [21].
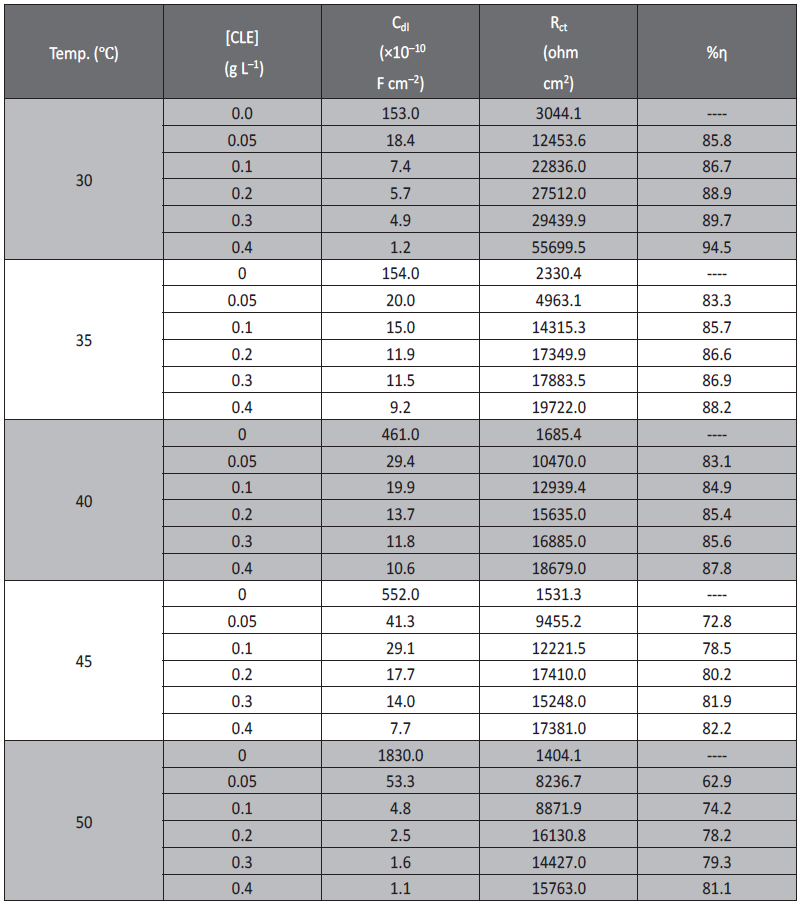
The measured value of polarization resistance was found to increase with the increasing concentrations of CLE. It may be due to the decrease in the corrosion rate for the aluminum with increase in inhibitor concentration. This is in accordance with the observations obtained from potentiodynamic measurement. An equivalent circuit of nine elements depicted in Figure 4b and Figure 4a was used to interpret the measured impedance data of the aluminum in sulfuric acid (pH=3).

Fig. 4: Equivalent circuit fit for the corrosion of aluminum in H2SO4 (pH =3) containing CLE (0.3 g L–1) at 45 ℃.
In this equivalent circuit Rs is the solution resistance and Rt is the charge transfer resistance. RL and L represents the inductive elements. CPE (Q) is constant phase element in parallel to the series capacitors C1, C2 and series resistors R1, R2, RL and Rt. RL is parallel with the inductor L.
The double layer capacitance Cdl was calculated from equation (6).
Equation 6:
![]()
And the polarization resistance Rp was calculated using the equation (7).
Equation 7:
![]()
Since Rp is inversely proportional to the corrosion current and it can be used to calculate the percentage inhibition efficiency using the relation (4).
As seen from Figure 3 solution resistance (Rs) remained almost constant, with and without addition of aqueous extracts CLE on aluminum. It was also observed that the value of constant phase element Q, decreased, while the values of Rt increased with increasing concentration of inhibitor, indicating that the inhibition efficiency increased with the increase in concentration of aqueous extracts of CLE.
The double layer between the charged metal surface and the solution is considered as an electrical capacitor. The adsorption of inhibitor molecules on the aluminum surface decreases its electrical capacity because they displace the water molecules and other ions originally adsorbed on the surface. The decrease in this capacity with increase in inhibitor concentrations may be attributed to the formation of a protective layer on the electrode surface. The obtained CPE (Q) values decreased noticeably with increase in the concentrations of inhibitor. This may be due to the adsorption of constituents of CLE on the metal surface as Q is inversely propositional to the thickness of the protective layer [26]. The impedance parameters derived from the Nyquist plot and inhibition efficiency of CLE in sulfuric acid (pH=3) at different temperatures are given in Table 2.
Tab. 2: Impendence parameters for corrosion of aluminum in H2SO4 (pH = 3) containing different concentrations of CLE at different temperatures
3.4. Effect of temperature
Inhibition efficiency increased with increase in the concentrations of the CLE but decreased with increase in temperature. This can be attributed to the decrease in the protective nature of the inhibitive film formed on the metal surface (or desorption of the inhibitor molecules from the metal surface) at higher temperatures [34]. This suggests physical adsorption mechanism. Physical (electrostatic) adsorption may take place when inhibition efficiency decreases with increase in temperature (where as chemical adsorption takes place when inhibition efficiency increases with increase in temperature) [32-33]. However, at higher concentrations of inhibitor this decrease was small. The study of effect of temperature was used to calculate energy of activation (Ea) for the corrosion process in the presence and absence of inhibitor using Arrhenius law Equation (8) [21].
Equation 8:

where B is a constant which depends on the metal type and R is the universal gas constant (R= 8.314 J mol−1 K−1), T is the absolute temperature. Plot of ln (CR) versus 1/T gave straight line with a slope (slope = -Ea/R). From the slope energy of activation was calculated.
The Arrhenius plot for the aluminum in the presence of inhibitor in sulfuric acid (pH=3) is shown in Figure 5.

Fig. 5: Arrhenius plots for the corrosion of aluminum in H2SO4 (pH =3) containing different concentrations of CLE.
Enthalpy of activation (∆Ha) and entropy of activation (∆Sa) for the dissolution of specimen were calculated from transition state Equation (9) [25].
Equation 9:

where h is Plank’s constant (h = 6.626×10–34 J s) and N is Avogadro’s number (N =-6.023×1023).
Plot of ln (CR/T) versus 1/T for the aluminum in various concentrations of inhibitor in sulfuric acid (pH=3) is shown in Figure 6. Plot of ln (CR/T) versus 1/T gave straight lines with slope (slope = ∆Ha/R) and intercept (Intercept = lnR/Nh +∆Sa/R). The calculated values of Ea, ∆Ha and ∆Sa obtained for aluminum in sulfuric acid (pH=3) is given in Table 3.
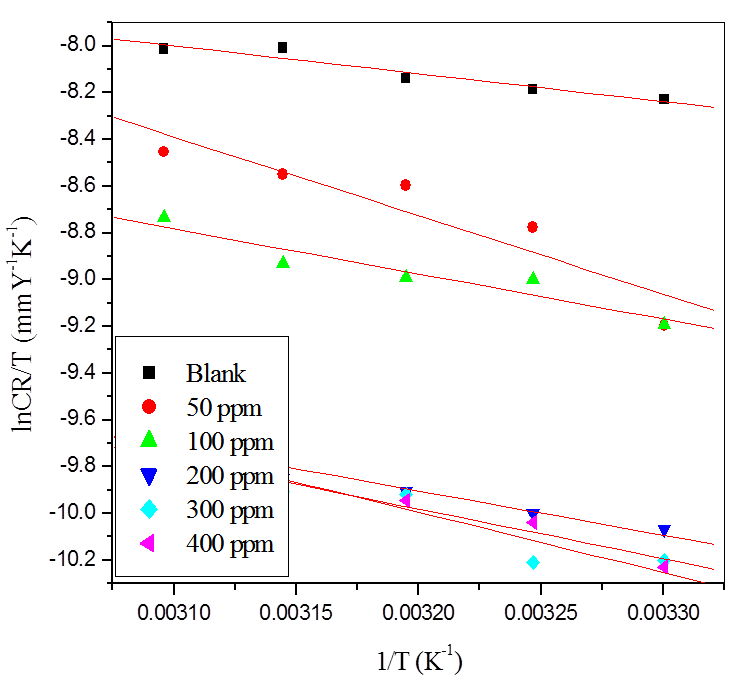
Fig. 6: Plots of ln (CR/T) vs 1/T for the corrosion of aluminum in (a) 0.001 M HCl (b) 0.001 M H2SO4 containing different concentrations of CLE.
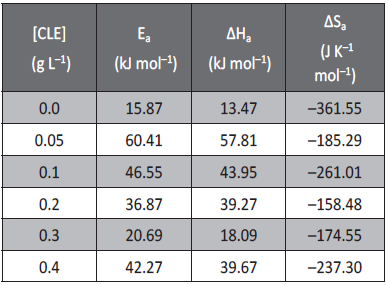
Tab. 3: Activation parameters for the corrosion of aluminum
in H2SO4 (pH =3) containing different concentrations of CLE.
As reported in Table 3, Ea values increased significantly after the addition of the inhibitor. The increase in activation energy (Ea) of inhibited solutions compared to the blank suggests that inhibitor gets adsorbed on the corroding metal surface. The increase in the Ea values, with increasing inhibitor concentration indicates the increase in energy barrier for the corrosion reaction [21]. The adsorption of the inhibitor molecules on the surface of the aluminum blocks the charge transfer during corrosion reaction, thereby increasing the activation energy. In other words, the adsorption of the inhibitor on the electrode surface leads to the formation of a physical barrier that reduces the metal reactivity in the electrochemical reactions of corrosion [34].
The inhibition efficiency decreased with increase in temperature which may be due to the desorption of inhibitor molecule with increase in temperature [35].The values of ΔSa were higher for inhibited solutions than those for the uninhibited solutions. This suggested that an increase in randomness occurred on going from reactants to the activated complex. This might be the results of the adsorption of organic compound present in the CLE from the acidic solution which could be regarded as a quasi-substitution process between the organic compound in the aqueous phase and water molecules at electrode surface. In this situation, the adsorption of inhibitor is accompanied by desorption of water molecules from the surface. Thus the increase in entropy of activation was attributed to the increase in solvent entropy [36].
3.5. Adsorption considerations
Inhibitors are known to decrease metal dissolution via adsorption on the metal/corrodent interface to form a protective film which separates the metal surface from the corrosive medium. The adsorption route is usually regarded as a substitution process between the inhibitor in the aqueous solution [Inh (sol) ] and water molecules adsorbed at the metal surface [H2O (ads) ] as given in reaction (10) [37].
Equation 10:
![]()
where n represents the number of water molecules replaced by one molecule of adsorbed inhibitor. The adsorption bond strength is dependent on the composition of the metal, corrodent, inhibitor structure, concentration and orientation as well as temperature. Adsorption isotherms are usually employed to explain the mechanism of interaction between an inhibitor (adsorbate) and an adsorbent surface. This is usually achieved by fitting the degree of surface coverage data into various adsorption isotherms or models and the correlation coefficients (highest) used to determine the best fit isotherm which can then be used to describe the inhibitor adsorption mechanism. In this work, the best fitted isotherm was the Langmuir adsorption model which relates the degree of surface coverage (θ) to the concentration of the extracts (Cinh.) according to equation (11).
Equation 11:
![]()
Where Kads is the equilibrium constant of the inhibitor adsorption process and C is the inhibitor concentration, and θ is the degree of the surface coverage, which is calculated using Equation (3).
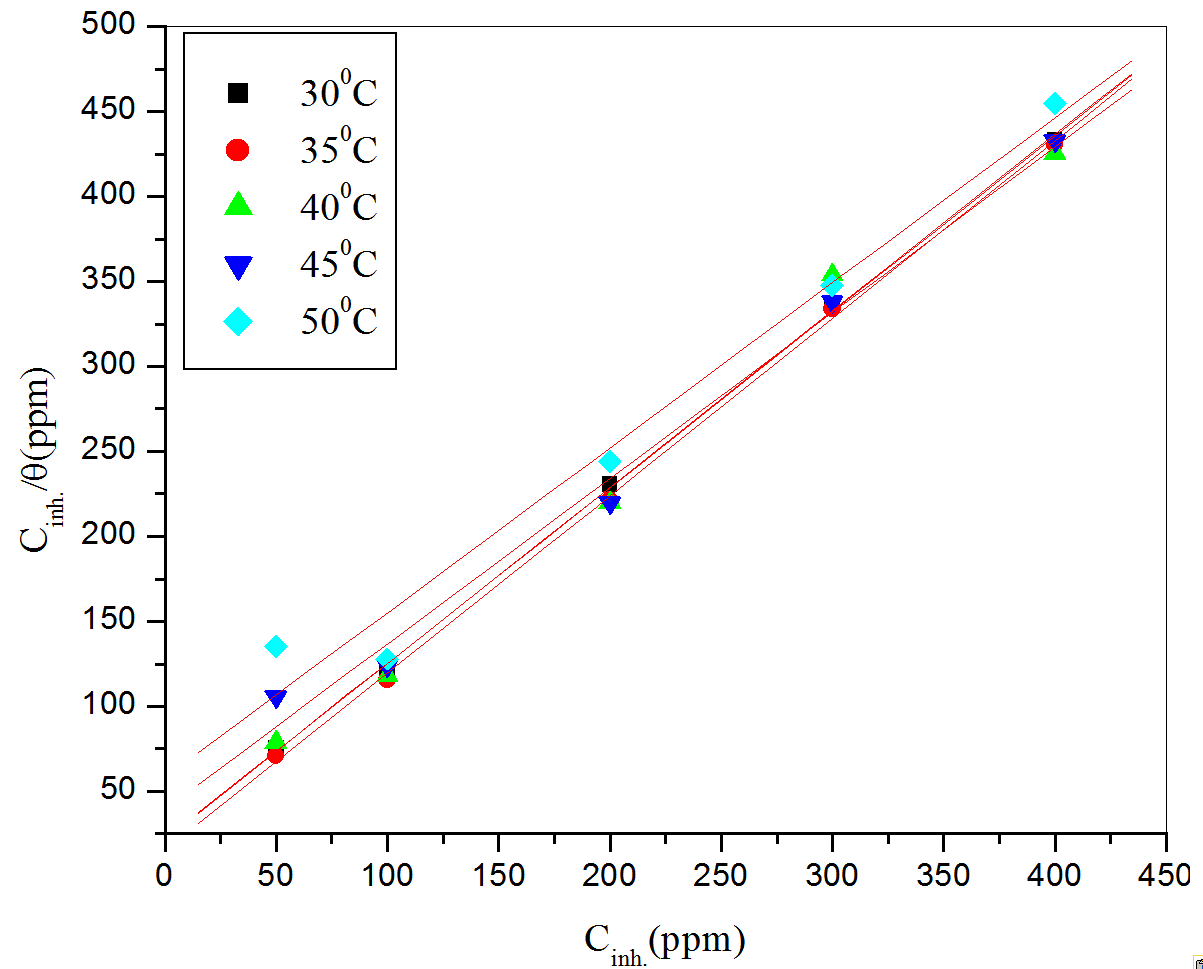
This model has also been used for other inhibitor systems [38-39]. The plot of Cinh/θ versus Cinh gave a straight line with an intercept (intercept = 1/K). It is shown in Figure 7.
The correlation coefficient (R2) was used to choose the isotherm that best fit the experimental data. The linear regression coefficients were close to unity, and the slopes of straight lines were nearly unity, suggesting that the adsorption of organic compounds present in CLE obeyed Langmuir’s adsorption isotherm, and there is negligible interaction between the adsorbed molecules [37].
The high values of K for the studied inhibitor indicate strong adsorption of inhibitor on the metal surface. The value of K increased with increase in inhibitor concentration also indicates the physical adsorption of the inhibitor on the metal surface.
The values of standard free energy of adsorption are related to K by the relation (12).
Equation 12:

where Cwater is the concentration of water in solution expressed in g L–1, R is the universal gas constant (8.314 J mol–1 K–1) and T is the absolute temperature. It must be noted that the concentration unit of water molecules has to be same as that of the inhibitor (the unit of Cwater is g L–1 with the value of approximate 1 ×103) [40].
The plot of ΔG°ads versus T as shown in Figure 8 was used to calculate enthalpy of adsorption (ΔH°ads) and standard adsorption entropy (ΔS°ads) using thermodynamic Equation (13). The negative values of ∆G°ads suggested the spontaneous adsorption of inhibitor on the surface of metal and the stability of the adsorbed layer on the aluminum.
Equation 13:
![]()
The thermodynamic parameters obtained for aluminum in sulfuric acid (pH=3) is tabulated in Table 4.
In general, the standard free energy values of −20 kJ mol−1 or less negative are associated with an electrostatic interaction between charged molecules and charged metal surface, resulting in physisorption, and standard free energy values of − 40 kJ mol−1 or more negative involve charge sharing or transfer from the inhibitor molecules to the metal surface to form a coordinate covalent bond, resulting in chemisorption [21]. The ∆G°ads values obtained for the studied inhibitor on the aluminum surface in sulfuric acid (pH=3) is more than –20 kJ mol–1 and less than –40kJ mol–1 which indicates the adsorption of constituents of CLE on the surface of aluminum is a mixture of both the process [35].
The values of thermodynamic parameters for the adsorption of inhibitors can provide valuable information about the mechanism of inhibition. While an endothermic adsorption process (∆H°ads > 0) is attributed unequivocally to chemisorption, an exothermic adsorption process (∆H° <ads 0) may involve either physisorption or chemisorptions or a mixture of both the processes. In an exothermic process, physisorption is distinguished from chemisorption by considering the absolute value of adsorption enthalpy. In this process, the enthalpy of a physisorption process is lower than 41.86 kJ mol–1, while that of a chemisorption process approaches 100kJ mol–1 [35]. In the present case, the calculated value of ∆H°ads is −57.28 kJ mol−1, which indicates the adsorption of CLE on the surface of aluminum is a mixture of both process. The ∆S°ads value is negative, indicating that an ordering takes place when the inhibitor gets adsorbed on the metal surface [41].
3.6. Mechanism of Inhibition
Surface of aluminum is covered with thin layer of γ alumina which initially thickens on exposure to neutral aqueous solution with the formation of a layer of crystalline hydrated alumina. The aluminum surface has positive charge in acidic environment in contact with sulfuric acid [41-42]. The mechanism of adsorption can be predicted on the basis of the mechanism proposed for the corrosion of aluminum in sulfuric acid [40].
Anodic reactions
Equation 14:
![]()
Equation 15:
![]()
Equation 16:
![]()
Equation 17:
![]()
Cathodic reaction
Equation 18:
![]()
Equation 19:
![]()
CLE is composed of numerous naturally organic heterocyclic compounds. Major constituents of the aqueous extract of CLE are reported to be Terpene-4-ol, β–caryophylene and β–elemene [20]. The structures of same are given in Figures 9a, 9b and 9c.
 Two modes of adsorption process could be suggested to explain the inhibitory action of CLE on aluminum corrosion in sulfuric acid. Thus, in aqueous acidic solutions, Terpene-4-ol molecules may exist either as neutral molecule or in the form of protonated species [43] while β–caryophylene and β–elemene molecules exist as neutral species.
Two modes of adsorption process could be suggested to explain the inhibitory action of CLE on aluminum corrosion in sulfuric acid. Thus, in aqueous acidic solutions, Terpene-4-ol molecules may exist either as neutral molecule or in the form of protonated species [43] while β–caryophylene and β–elemene molecules exist as neutral species.
CLE acts as an anodic type at lower concentrations of inhibitor, and brings down anodic reaction under control. In acid medium, aluminum is positively charged since the pH of the potential of zero charge for aluminum at the oxide/solution interface is 9.0-9.1. This will attract negative sulfate ions from
the solution, and these ions will get adsorbed on the surface of the metal [44]. In acidic solution the hydroxyl groups of Terpene-4-ol present in CLE can be easily protonated because of their high electron density. The protonated molecules are attracted to the anionic layer which is formed on the metal surface, and get physically adsorbed on the same. This will result in the formation of protective barrier on the surface of the metal. This barrier will prevent the further dissolution of the metal and prevent the metal from undergoing corrosion. Enhanced corrosion inhibition may be also due to the π electrons of the double bond of constituents of the extract which also acts as high electron rich centers.
But as the inhibitor concentrations increases CLE acts as a mixed type of inhibitor by bringing both anodic and cathodic reaction under control. The neutral molecules Terpene 4–ol, β–caryophylene and β–elemene present in CLE molecules may be adsorbed on the metal surface on the basis of donor–acceptor interactions between π-electrons of double bonds in alkene and vacant p-orbitals of Al[45]. Hence the neutral molecules Terpene 4–ol, α-pinene and Seberine present in CLE may be adsorbed on the metal surface through the chemisorption. As these molecules have large size they cover the entire metal surface and form a protective film, thereby bring down both anodic and cathodic reaction under control and hence acts as mixed inhibitor.
3.7. Scanning Electron Microscopy and Energy-dispersive X-ray spectroscopy
The SEM image of aluminum in sulfuric acid (pH=3) are shown in Figure 10a.
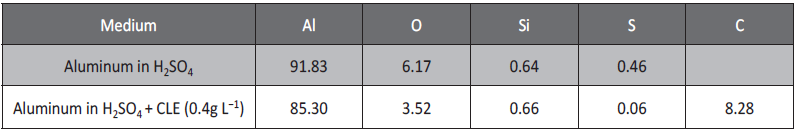
The SEM image showed the heterogeneous surface with small pits, which indicates that the metal undergoes pitting corrosion in sulfuric acid [42]. The EDX spectrum showed the presence of highest percentage of aluminum and small percentage of sulfur. The presence of peak for sulfur in EDX spectrum indicates the interaction of aluminum with sulfuric acid.
The SEM image of aluminum after the addition of CLE (0.4 g L–1) to sulfuric acid (pH=3) are shown in Figure 10b.
The SEM image showed the decrease in the number of pits and increase in the smoothness of the surface. This is due to the adsorption of constituents of CLE on the surface of aluminum. This is confirmed by EDX spectrum as it showed the decrease in the percentage of aluminum and sulfur and increase in the percentage of carbon. Results of EDX analysis are tabulated in Table 5.
Conclusions
- CLE is a green inhibitor, which is readily available and cost effective inhibitor for the corrosion of aluminum.
- The inhibition efficiency increased with increase in inhibitor concentration and decreased with increase in temperature.
- Aqueous extracts of CLE acted as anodic inhibitor at lower concentrations of inhibitor and mixed type at higher concentrations of inhibitor.
- CLE got adsorbed on the aluminum surface via both physisorption and chemisorption process and followed Langmuir’s adsorption model.
- The inhibition efficiency obtained from potentiodynamic polarization, and EIS techniques were in good agreement with each other.
Acknowledgement
Pushpanjali M gratefully acknowledges the laboratory facilities provided by Department of Chemistry, MIT, Manipal University Manipal.
References
- F.-H. Cao, Z. Zhang, J.-F. Li, Y.-L. Cheng, J.-Q. Zhang and C.-N. Cao, ” Exfoliation corrosion of aluminum alloy AA7075 examined by electrochemical impedance spectroscopy”, J. Materials and Corrosion, 55 (2004) 18-23.
- M. A. Amin, S. Abd EI-Rehim, E. F. El-Sherbini, O. A. Hazzazi, M. N. bbas, “Polyacrylic acid as a corrosion inhibitor for commercial sample of aluminum in weakly alkaline solutions. Part I: Weight loss, polarization, impedance EFM and EDX studies”, J. Corros. Sci., 51 (2009) 658–667.
- A. Y. El-Etre, “Inhibition of aluminum corrosion using Opuntia extract”, J. Corros. Sci., 45 (2003) 2485–2495.
- D. Mercier, M.-G. Barthés-Labrousse, “The role of chelating agents on the corrosion mechanisms of aluminum in alkaline aqueous solutions”, J. Corros. Sci., 51 (2009) 339–348.
- J. B. Bajat, V. B. Miškovic-S tankovic, Z. Kacˇarevic. Popovic”, Corrosion stability of epoxycoatings on aluminum pretreated by vinyltriethoxysilane”, J. Corros. Sci., 50 (2008) 2078–2084.
- I. B. Obot, N. O. Obi-Egbedi, “Ginseng Root: A new Efficient and Effective Eco-Friendly Corrosion Inhibitor for Commercial sample of aluminum Alloy of type AA 1060 in Sulfuric acidSolution”, Int. J. Electrochem. Sci., 4 (2009) 1277 – 1288.
- I. B. Obot, N. O. Obi-Egbedi, “Ipomoea Involcrataas an Ecofriendly Inhibitor for Commercial sample of aluminum in Alkaline Medium”, PortugaliaeElectrochi. Acta, (2009) 27 (4), 517-524.
- Ali and N. Foaud, “Inhibition of aluminum corrosion in sulfuric acidsolution using black mulberry extract”, J. Mater. Environ. Sci., 3 (5) (2012) 917-924.
- M. Saleh, A. A. Ismail, A. G. El Hosary, “Corrosion inhibition by naturally occuring substances-IX. The effect of the aqueous extracts of some stems, leaves, fruits and fruits peels on the corrosion of Al in NaOH”, J. Corros Science, 23 (1983) 1239.
- G. O. Avwiri, F. O. Igbo, “Inhibition action of Vernoniaamygdalina on the corrosion of aluminum alloys in acidic media”, Materials Letters, 57 (2003) 3705.
- Umoren, S. A., Obot, I. B., Ebenso, E. E., Okafor, P. C. “Eco-friendly inhibitors from naturally occurring exudates gums for aluminum corrosion inhibition in acidic medium”, PortugaliaeElectrochimica. Acta, 26 (2008) 267.
- Umoren, S. A., Obot, I. B., Ebenso, E. E., Obi-Egbedi, N. O., “Inhibition of commercial sample of aluminum corrosion in sulfuric acidsolution by exudates gum from Raphiahookeri”, Desalination, 247 (2009) 561.
- Halambek J., Žutinić A., Berković K, “Ocimum basilicum L. Oil as Corrosion Inhibitor for Aluminum in Sulfuric acid Solution”, Int. J. of Electrochemic. Sci., 8 (2013) 11201-11214.
- O. K. Abiola, J. O. E. Otaigbe, O. J. Kio, “Gossipiumhirsutum L. Extracts as green corrosion inhibitor for aluminum in NaOH solution”, J. of Corros. Sci., 51 (2009) 1879.
- I. B. Obot, N. O. Obi-Egbedi, “Ginseng Root: A new Efficient and Effective Eco-Friendly corrosion Inhibitor for Aluminum Alloy of type AA 1060 in Sulfuric acidSolution”, Int. J. of Electrochemic. Sci., 4 (2009) 1277-1288.
- I. B. Obot, 1, N. O. Obi-Egbedi, “Ipomoea Involcrata as an Ecofriendly Inhibitor for Aluminum in Alkaline Medium”, Portugaliae Electrochimica Acta, (2009) 27 (4), 517-524.
- Deepa Prabhu., Rao Padmalatha., Coriandrum sativum L. —A novel green inhibitor for the corrosion inhibition of aluminium in 1. 0 M phosphoric acid solution J. Environ. Chem. Eng., 1 (2013) 676-683.
- Deepa Prabhu., Padmalatha Rao, “Garcinia indica as an Environmentally Safe Corrosion Inhibitor for Aluminium in 0. 5M Phosphoric Acid”’ Int. J. Chem. Env. Eng., 1 (2013) 675-683.
- Pushpanjali M, Suma A Rao, Padmalatha, Inhibitive effect of Carica papaya seed extract on aluminium in H2SO4 medium J. of Mater. Environ. Sci., 5 (2) (2014) 591-598.
- Sinha Parul*, Akhtar Javed, Batra Neha, Jain Honey, Bhardwaj Anuj, “Curry Leaves – A Medicinal Herb review article”, Asian J. Pharm. Res., 2 (2012) 51-53.
- MG. Fontana, “Corrosion engineering”, McGraw-Hill, Singapore, (1987) 106, 3rd edn.
- F. B. Mansfeld, “Corrosion mechanisms”, Marcel Dekkar, New York, (1987) 165-209.
- Kadher, A.; El Warraky, J. M.; Abd el Aziz, A. M. Corrosion inhibition of mild steel by sodium tungstate in neutral solution. Br. Corr. J. 1998, 33 (2) 139.
- Khaled, K. F.; Al-Qahtani, M. M. The inhibitive effect of some tetrazole derivatives towards Al corrosion in acid solution: Chemical, electrochemical and theoretical studies. Mater. Chem. Phys. 2009, 113 (1) 150–158.
- W. H. Li, Q. He, C. L. Pei, B. R. Hou, “Some new triazole deravatives as inhibitors for mild steel corrosion in acid medium”, J. of Appl. Electrochem., 38 (2008) 289.
- W. H. Li, Q. He, C. L. Pei, B. R. Hou, “Experimental and theoretical investigation of the adsorption behaviour of new triazole derivatives as inhibitors for mild steel corrosion in acid media”, J. of Electrochim. Acta, 52 (2007) 6386.
- J. Ross Macdonald, “Impedance Spectroscopy”, John Wiley and Sons, 2nd edition (1987).
- C. M. A. Brett, “Electrochemical impendence spectroscopy applied to study of aluminum corrosion”, J. of Appl. Electrochem., 20 (1990) 1000.
- F. Mansfeld, S. Lin, . Kim, H. Shih, “Electrochemical Impedance Spectroscopy as a Monitoring Tool for Passivation and Localized Corrosion of Al Alloys”, J. of Mater. Corros., 39 (1988) 487.
- F. Mansfeld, S. Lin, K. Kim, H. Shih, “Pitting and Surface Modification of SiC/Al”, J. of Corros. Sci., 27 (1987) 997.
- C. M. A. Brett, “Electrochemical behavior of aluminum in basic chloride solution”, J. Corros. sci., 33 (2) (1992) 203-210.
- H. Wit, H. J. W. lenderink, “Electrochemical impendence spectroscopy as a tool to obtain mechanistic information on the passive behavior of commercial sample of aluminum” J. Electrochem. Acta., 41 (7-8) (1996) 1111-1119.
- J. B. Bessone, D. R. Salinas, C. Mayer, M. Ebert, W. J. Lorenz, “An EIS study of aluminum barrier-type oxide films formed in different media”, J. of Electrochim. Acta, (1992) 37 2283.
- M. Schorr, Yahalom, “The significance of the energy of activation for the dissolution reaction of metal in acids”, J. of Corros. Sci., 12 (1972) 867.
- M. Bouklah, B. Hammouti, A. Aounti, T. Benhadda, “Thiophene derivatives as effective inhibitors for the corrosion of steel in 0. 5M H2SO4”, J. of Prog. Org. Coat., 49 (2004) 227.
- M. I. Awad, “Eco friendly corrosion inhibitors: Inhibitive action of quinine for corrosion of low carbon steel in 1m HCl”, J. of Appl. Electrochem., 36 (10) (2006) 1163-1168, DOI: 10.1007/s10800-006-9204-1
- M. Sahin, S. Bilgic, H. Yilmaz, “The inhibition effects of some cyclic nitrogen compounds on the corrosion of the steel in NaCl mediums”, J. of Appl. Surf. Sci., 195 (1) (2002) 1-314.
- B. Ateya, B. E. El-Anadouli, F. M. El-Nizamy, “The adsorption of thiourea on mild steel” J. of Corros. Sci., 24 (6) (1984) 509-515.
- F. Mansfeld, Corrosion, (1973) 29, 1st edition.
- E. E. Oguzie, V. O. Njoku, C. K. Enenebeaku, C. O. Akalezi, C. Obi, “Effect of hexamethylpararosaniline chloride (crystal violet) on mild steel corrosion in acidic media”, J. of Corros. Sci., 50 (2008) 3481.
- T. Sanaa Arab, “Inhibition action of thiosemicabazone and some of it is ρ-substituted compounds on the corrosion of iron-base metallic glass alloy in 0. 5 M H2SO4 at 30 °C”, J. of Mater. Res. Bull, 43 (2008) 516-521.
- O. Olivares, N. V. Likhanova, B. Gomez, J. Navarrete, M. E. Llanos-Serrano, E. Arce, J. M. Hallen, J. of Appl. Surf. Sci., (2006) 252, 2894. (42).
- S. E. Frers; M. M. Stefenel, C. Mayer, T. Chierchie, “AC-Impedance measurements on aluminum in chloride containing solns and below the pitting potential”, J. of Appl. Electrochem., 20 (6) (1990) 996–999.
- E. E. Oguzie, Y. Li, F. H. Wang, “Corrosion effect of Allylthiourea on bulk Nanocrystalline Ingot iron in diluted acidic sulphate solution”, J. Electrochem. acta, 52 (2007) 3950-3957.
- E. I. Atinga, S. A. Umoren, I. I. Udousoro, E. E. Ebenso, A. P. ‘Udoh, Inhibitive Action of Anethum graveolens L. oil on Aluminium Corrosion in Acidic Media” J. Green Chem. Lett. Rev., 3 (2010) 61.
PDF Version of the article |
Flash Version of the article |
|
| [qr-code size=”2″] | ||