Tin-cobalt alloys were electrodeposited onto nickel-plated brass substrates using a pyrophosphate electrolyte. Deposition conditions were as follows: current density, 0.2-0.3 A/dm2, bath temperature, 45±2°C, pH 8-8.2, ratio of Sn:Co 1.2:1 and current efficiency, approximately 90%. The bath exhibited good throwing power. Deposit morphology was uniform, with cauliflower-like microstructure, mean grain size 125nm. Visual appearance was very similar to electrodeposited chromium with a bluish-white colour. Electrochemical corrosion measurements using Tafel extrapolation and impedance data (as Nyquist plots) showed that Sn-Co alloy and decorative chromium had comparable corrosion resistance with values of 7.77*10-7 A/cm2 and 200 000 Ωcm2 respectively. The results demonstrate that electrodeposited tin-cobalt alloy can be a viable replacement for decorative electroplated chromium.
1. Introduction
Electrodeposition of chromium, whether for decorative or functional purposes, is one of the most widely used surface finishing operations. The brightness, high hardness and corrosion resistance of such coatings underlies their widespread use. The classical technology for the electrodeposition of these, is based on the use of hexavalent chromium (as chromic acid) [1]. However problems such as low current efficiency, low deposition current density and low throwing power remain largely unsolved. Far more serious is the recent recognition that hexavalent chromium is toxic and carcinogenic. As a result, operation of hexavalent chromium plating baths are now subject to extremely stringent conditions in terms of effluent and airborne emissions. thus prompting an ongoing search for cost-effective alternatives[2].
Tin alloy electrodeposition is widely used on the industrial scale. Tin alloys have high corrosion resistance, good brazeability and solderability, high hardness and the other valuable properties not unlike those of electroplated chromium. That such alloys might be used to replace electroplated chromium was suggested as long ago as 1951 [3]. More recently, developments in brightener technology for SnCo electrodeposits has strengthened the case for their use as alternatives to decorative chromium, especially where high corrosion resistance is not essential [4]. Under some conditions, their corrosion resistance can actually be superior to that of electroplated chromium. Tin-cobalt alloys can also be used, in certain applications, as substitutes for precious metal coatings, thereby avoiding use of cyanide-based electrolytes. It follows that tin-cobalt electrodeposits can be attractive coatings in a wide variety of applications where high hardness is not essential and the service environment is not unduly aggressive.[5, 6].
The present work reports a parametric study of tin-cobalt electrodeposition in terms of deposit morphology and alloy composition, using an alkaline pyrophosphate bath. Deposit properties were then compared with those of decorative chromium coating in terms of their physical parameters and corrosion resistance.
2. Experiment
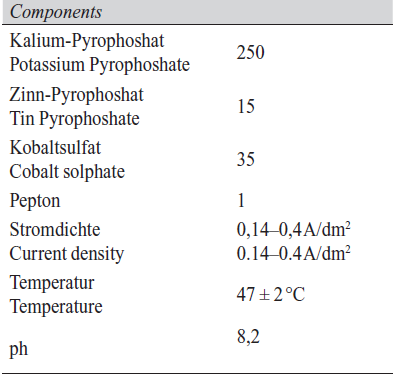
In this work, a pyrophosphate electrodeposition electrolyte was selected for environmental reasons, such as those related to effluent treatment and discharge [7]. The alkaline pyrophosphate system is also one allowing considerable latitude in terms of operating conditions and parameters. Characteristics of the bath used in this work are shown in Table 1.
All chemicals used were 99 % purity (Merck) Sulphuric acid (20%) and potassium hydroxide (30 %) were used for adjusting pH. Magnetic stirring was used during electrodeposition. Cathode substrates were brass plate or mild steel, measuring 0.5 · 50 · 130 mm, stainless steel anodes were used. The exposed area was 100 · 50 mm on each side, giving a total exposed area of 1 dm2. A proprietary chromium plating bath (DC 150) was used for preparing the bright chromium coatings. Following the recommendation of Dillenberg and et al.[8], a bright nickel coating was used as an underlayer prior to Sn-Co alloy deposition. A standard 267 ml Hull-cell was used to determine throwing power of Sn-Co and Cr electrolytes. Deposit morphologies and compositions and the effect of deposition conditions on these, were studied using an SEM (Cambridge, Model S4160) with EDS elemental analysis facility. Optical properties of electrodeposits, including brightness were determined using a Hitachi spectrophotometer (u-3410). ASTM Standard B571-84 procedure was used to determine adhesion of Sn-Co alloy deposit to substrate. Corrosion resistance of Sn-Co alloy and decorative chromium coatings was determined using Tafel extrapola- tion in 3.5 % NaCl solution at ambient temperature, as well as electrochemical impedance using an Iviumatat system (Ivium Co, NL).
3. Results and Discussion
3.1. Effect of current density on deposit composition and morphology
Figure 1 shows SEM images of deposits at current density 0.1–0.4 A/dm2, temperature 47 ± 2 °C and pH approximately 8.2. with concentration ratio in solution Sn] : [Co] = 1 : 2. Details of alloy composition and cathode efficiency are shown in Table 2.
![Tab. 2: Effect of current density on alloy composition (T = 47 ± 2 °C, pH = 8.2, Sn] / [Co] = 1 : 2)](https://www.jept.de/wp-content/uploads/2015/05/tab-2.png)
Tab. 2: Effect of current density on alloy composition (T = 47 ± 2 °C, pH = 8.2, Sn] / [Co] = 1 : 2)
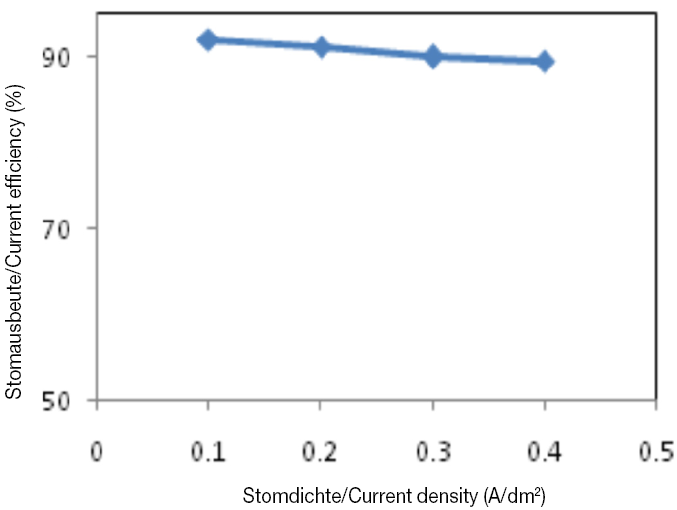
Figure 1 shows the grain structure of deposits formed at current densities 0.1 and 0.2 A/dm2. Further increase in current density to 0.3 A/dm2 results in a more compact morphology with cauliflower-like formations and columnar structures formed by agglomeration of fine grains. At the lower current densities, (as indicated by the white areas) EDS analysis revealed a tin-rich second phase. At current densities > 0.3 A/dm2, coatings were matt due to enhancement of over potential came from increasing current as well as vertical growth of coating particle prevention [9]. The cobalt content present in the deposit decreases when the current density is increased up to 0.4 A/dm2. Increased current density causes to increased tin content within deposition. With increasing the current density to 0.3 A/dm2 and 20%wt cobalt in the coating, a completely bright and uniform coating is found, in agreement with work able with the results reported in [10, 11]. The effect of current density variations on the cathode efficiency of Sn-Co plating bath was shown in figure 2. As seen in this figure, the cathode efficiency is high with a minimum value of approximately 88%. This should be contrasted with the cathode efficiency of a decorative chromium bath which rarely exceeds 45 % [5].
3.2. Effect of [Sn]:[Co] concentration ratio in solution on deposit morphology and composition
The effect of solution composition (expressed as ratios of [Sn] :[ Co] 0.9, 1.2, and 1.6) on deposit alloy composition and the associated SEM images are shown in Table 3 and Figure 3 respectively. Current density was 0.3 A/dm2, temperature 47 ± 2 °C, pH approximately 8.2. Variation of metal ion concentrations in solution was achived by adjusting cobalt concentrations.
Given that the cobalt deposition rate is slower than that of the tin, the increase in cobalt concentration in the bath results in macro particle coating morphology due to delay in deposition nucleation [12]. The typical coating has a more compact and uniform morphology with a ratio of 1.2. This result is compatible with the desired cobalt concentration range in the deposit of (20–22 wt%).
3.3. Effect of temperature in bath on the composition and morphology of coating
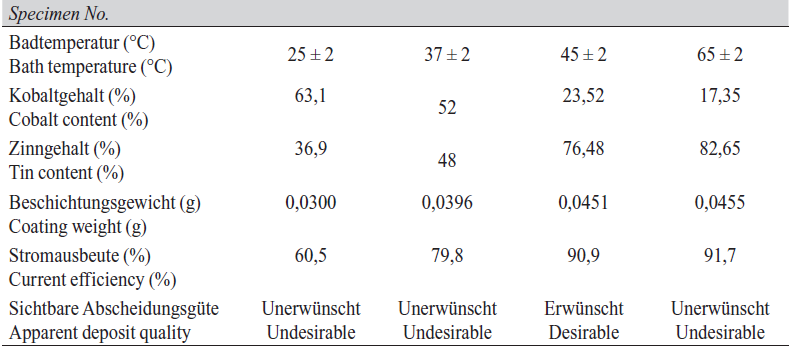
The SEM images obtained from coated surfaces after deposition at ambient (25 ºC), 37 ºC, 45 ºC and 65 ºC with current density 0.3 A/dm2, ratio of Sn : Co in solution from 1 : 1 to 1 : 2 and pH ca. 8.2 are shown in Figure 4. Values for deposited alloy composition and cathode efficiency are given in Table 4.
At low bath temperatures (25–37 ºC), the deposition rate was very low and deposit structure consisted of very tiny nuclei with minimal agglomeration or grain growth. Under these conditions, the initial coating has poor adhesion to the substrate and is easily separated from the undercoat even with modest bending of the test specimen. By increasing the temperature to 45 ºC, the deposit becomes more uniform without any porosity. This is also achieved by use of higher concentration of tin ions in solution and thus in the cathode double layer, resulting in enhanced Sn content and reduced cobalt content in the deposited alloy.
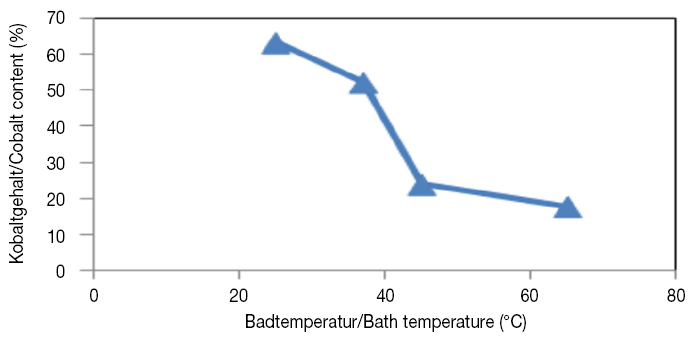
Also the morphology changed from granular to cauliflower-like structure [13]. By increasing the temperature to 65 ºC, as determined from was found out from analysis results of coating compositions, and with the reduction of cobalt content as a coating brightness factor, the coating was matt. in terms of Apparent deposit quality (brightness) that wasn’t suitable. The Figure 5 depicts the variations of coating composition in different temperatures of plating bath. By increasing the bath temperature, the amount of cobalt presented in coating has reduced.
In general, increase in temperature results in increased conductivity of the electrolyte, and enhance diffusion rates and redissolution in cathode. Increasing the temperature often reduces the polarization of more noble metal But when the temperature is more effective on the degree of polarization of active metal [13]. Thus the temperature has double effect as follows: in the one hand, increasing the temperature causes to decrease the polarization of more noble metal and on the other hand, with the increase diffusion of more noble metal to the double layer cathode, causes to decrease the pouring of layer adjacent from more active metal. Changing the alloy composition is dependent on overcoming each factor on another one. Due to the observed behavior in this system, it can be said that the effect of polarization reduction of more noble metal has overcome the increase diffusion of more active metal.
3.4. Throwing power of Sn-Co electrolytes
In order to study the throwing power of the Sn-Co alloy electrolyte, a Hull cell test panel was coated under optimum plating bath conditions. The results are seen in Figure 6 which shows complete coverage by the Sn-Co deposit across the entire cathode surface. Under optimum deposition conditions, the entire test panel is perfectly coated, representing a range of high to low current densities, demonstrating excellent throwing power. By contrast, for decorative chromium plating under optimum conditions, maximum coverage was only 75 % and this was further reduced with a modest deviations from optimum plating conditions.
3.5. Investigation of the Sn-Co coating brightness
Achieving the optimum conditions to obtain a bright Sn-Co alloy deposit was a major aim of this project allowing such coatings to compete with bright chromium in terms of physical characteristics such as brightness and color. For this, while using optimum deposition conditions for Sn-Co alloy coatings, specimens were deposited onto perfectly polished brass plate with a nickel underlayer under the following conditions: current density: 0.3 A/dm2, bath temperature 47 ± 2 ºC, ratio of Sn : Co: 1.2 and pH 8.2.
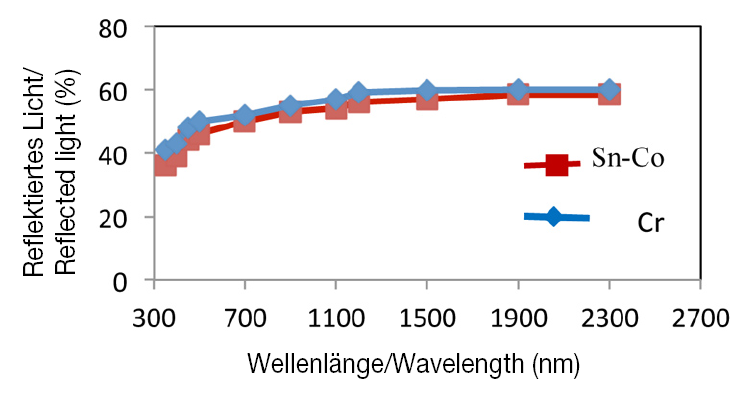
These conditions yielded extremely bright Sn-Co alloy coatings, which were compared with bright chromium coatings. The intensity of reflected light in the wavelength range of 350–2 500 nm was used to compare the coating brightness of the two sample types. The results are shown in figure 7.
The reflectance spectra show that reflectivity of the Sn-Co alloy coating was almost identical to that of bright chromium, approximately 60 % over a wide range of wavelengths, thus quantitatively confirming the similar visual appearance of the two coatings. As seen in Figure 6, both coatings exhibit a bluish white colour. These findings confirm that, in terms of visual appearance, Sn-Co alloys are viable alternatives to bright chromium.
3.6. Investigation of Sn-Co alloy coating adhesion
A thermal shock test was applied in order to study the adhesion of Sn-Co alloy coatings to substrate. Under optimum bath conditions of Sn-Co alloy plating, no blistering was observed after applying a thermal shock at 260 ºC for 1 hr, nor after subsequent cooling. This can be explained in terms of absence of shear stress resulting from different thermal expansion coefficients of coating and underlayer. Figure 8 shows the surface of the alloy coating after a thermal shock test. After heating to 260 ºC, the deposit morphology changed from cauliflower-like structure to a granular one, owing to enhanced diffusion. The results of bend- and thermal shock tests indicated that the Sn-Co alloy deposit formed under optimum conditions had sufficient adhesion.

Fig. 8: SEM images of the surface coating after termal shoking test in magnification: a) 15 000, b) 30 000
3.7. Corrosion resistance of Sn-Co compared to decorative chromium coating
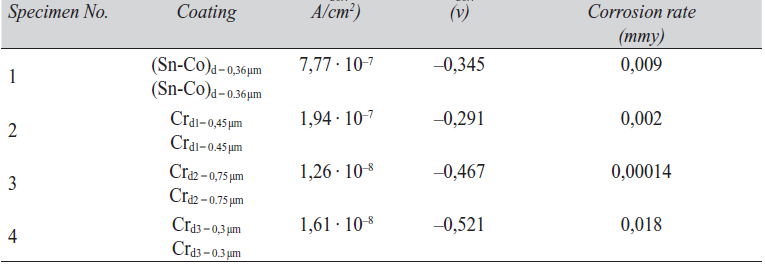
Corrosion measurements were made using the Tafel extrapolation and electrochemical impedance methods. Deposit thickness for Sn-Co and Cr samples were identical, and thickness values were determined using SEM images of metallographic cross-sections, as shown in Figure 9. Using these values, optimum alloy deposition rates of ca. 0.12 µ/min. can be estimated.
Figure 10 compares polarization curves for Sn-Co alloy deposition under optimum bath conditions with those for decorative chromium. In order to compare the corrosion characteristics of these two coatings, decorative chromium coatings were prepared at different thickness with an approximate deposition rate of 0.15 µ/min in the current density range 10–15 A/dm2. The essential parameters of these curves are shown in Table 5. As seen in Figure 10, there was little difference in corrosion current densities of specimen 1 plated under optimum conditions and chromium, especially for the specimen with thickness of 0.45 µm. This indicates that the corrosion resistance of a Sn-Co alloy deposit is comparable to that of a decorative chromium deposit of approximately the same thickness. It is also seen that the behavior of the Tafel extrapolation curve for Sn-Co deposits is similar to that for the decorative chromium coating. In terms of the corrosion current density and corrosion rates of Sn-Co deposits obtained in these tests, it can be concluded that Sn-Co alloy deposits with composition (20–22) wt% cobalt, had a good resistance to tarnishing as well as atmospheric corrosion, comparable to that of decorative chromium. It follows that where high corrosion resistance is not needed, Sn-Co alloy electroplating offers a good replacement for decorative chromium plate.
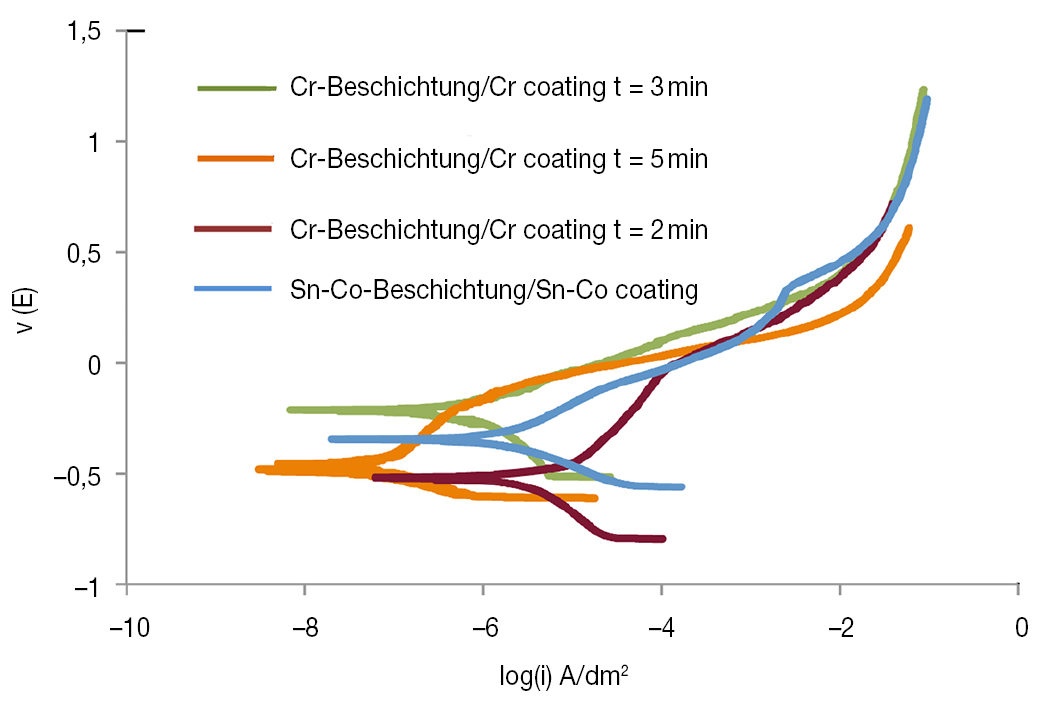
Fig. 10: Comparison of polarization curves between Sn-Co alloy deposits formed under optimum bath condition and decorative chromium deposits for three different deposition times
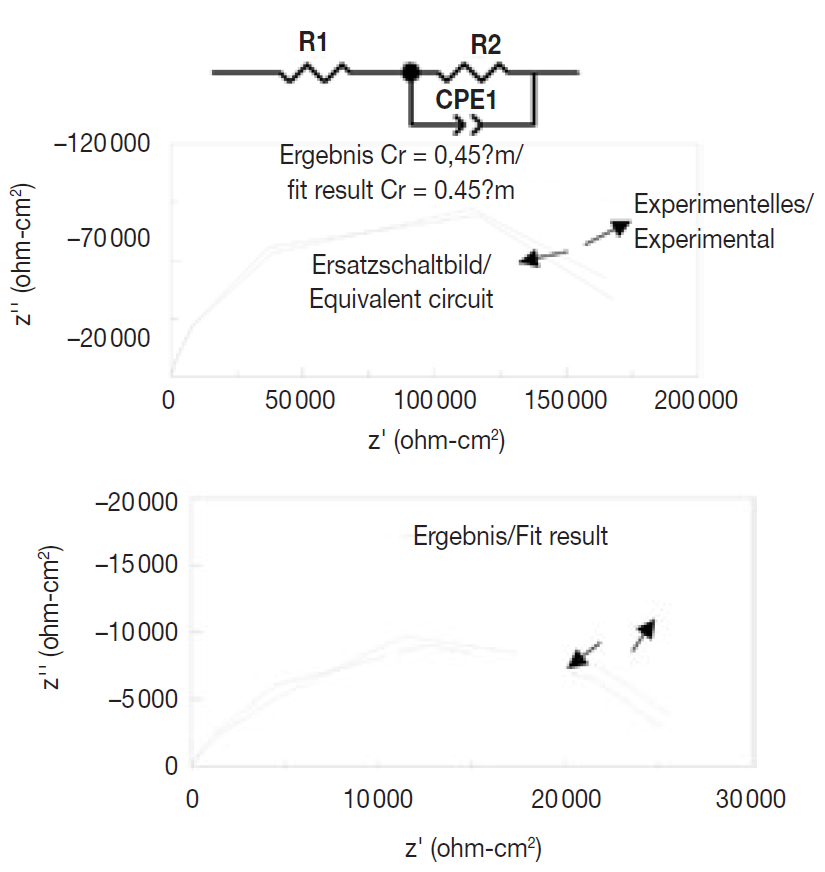
Further experiments to investigate the corrosion behavior of Sn-Co alloy deposits and those of decorative chromium as well as to confirm the results of Tafel extrapolation, were carried out using electrochemical impedance measurements from which Nyquist plots for these deposits were derived. The equivalent circuit and Nyquist plot for Sn-Co alloy deposits is shown in Figure 11. As seen, the experimentally obtained spectrum is in agreement with the impedance spectrum calculated from equivalent circuit. From this, the values of the equivalent circuit elements (R1, R2, and CPE1) were calculated. The values for these equivalent circuit elements calculated from the equivalent circuit for the Sn-Co alloy deposit are shown in Table 6 for an approximate thickness of 0.36 µm. From the data in Table 6, an impedance value of approx. 200 000 Ωcm2 was calculated for the Sn-Co alloy coating.
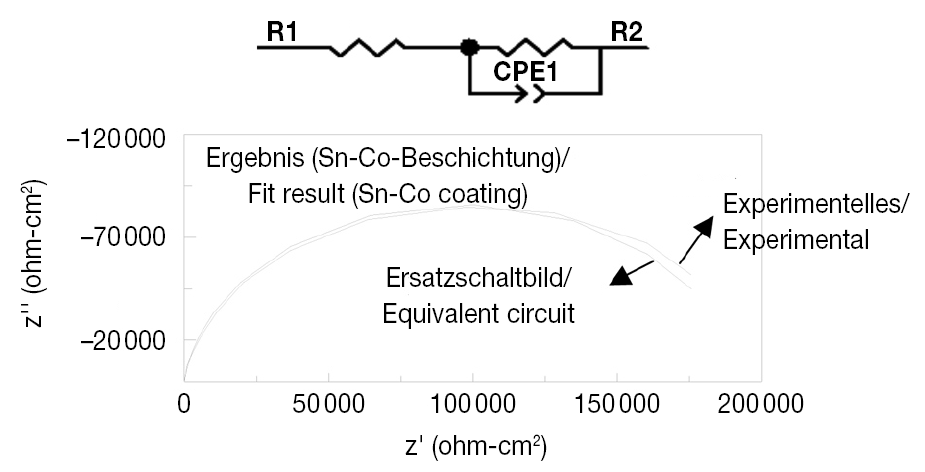
Fig. 11: Equivalent circuit and Nayquist curve of the Sn-Co coatings at open-circuit potential in 3.5% salt solution
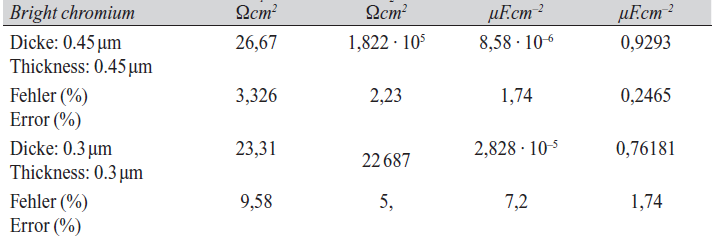
In order to compare the impedance spectra of Sn-Co and bright chromium coatings, the impedance Nyquist spectrum of a bright chromium coating was obtained for deposits of approximately of 0.45 µm and 0.3 µm thickness. The equivalent circuit and Nyquist impedance curve of bright chromium coating is shown in Figure 12 for these different thickness values. The parameters of impedance spectrum related to bright chromium coating are shown in Table 7.
In order to compare data shown in Tables 6 and 7 as well as comparing the impedance component (R2) of Sn-Co with bright chromium coatings, it noted that for approximately equal thickness values, the resistive component values of Sn-Co and bright chromium deposits are in the same range. This observation confirms the assertion that Sn-Co deposits have a corrosion resistance comparable to bright chromium coating at approximately equal values of thickness.
Figure 13 shows the Nyquist impedance spectra of Sn-Co alloy deposits compared with bright chromium coating.
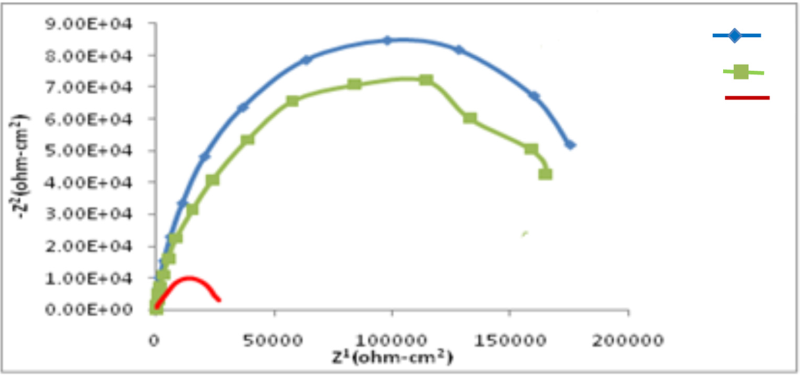
Fig. 13: Nyquist plots for Sn-Co and bright chromium deposits at open circuit potential in 3.5% NaCl solution
As seen in Figure 13, there are clear similarities between the impedance spectra of Sn-Co deposits and decorative chromium coating, with their semi-circular shape, as well as the similar coating polarization resistance values which, in turn, are in agreement with the results of Tafel curve procedures. These results confirm that Sn-Co alloy coating with 20–22% cobalt of 20–22% content has a resistance to tarnish and atmospheric corrosion comparable to decorative chromium and can thus be considered as a replacement for it.
4. Conclusions
- An Sn-Co alloy coating was successfully electroplated from an alkaline pyrophosphate bath using direct current. Optimum deposition conditions were: current density: 0.3 A/dm2, bath temperature: 47 ± 2 ºC, ratio of Sn to Co in solution: 1 : 2 and pH: 8–8.2, resulting in an alloy deposit of composition Sn-22 wt% Co.
- The morphology of Sn-(20-22) wt% Co alloy deposits under optimum condition was compact and cauliflower-like, with columnar structures of grown, uniform, crack-free fine particle agglomeration.
- The Sn-Co alloy in terms of physical properties (colour, brightness, smoothness) possessed a bluish white color similar to that of chromium coatings.
- Current efficiency decreases with increasing current density in the Sn-Co coating bath, but in the range of optimum current density, it exhibits a high current efficiency (90 %) as compared with decorative chromium electrolytes.
- The throwing power of Sn-Co alloy plating solutions is higher than those for chromium plating baths for 100 % coating.
- Sn-Co alloy deposits had good resistance to thermal shock without blistering.
- From the results of Tafel extrapolation, the corrosion current density and corrosion rate of Sn-(20-22 wt%) Co alloy coating was calculated to be 7,77 · 10–7 A/cm2 and 0.009 mm/y respectively. By comparison, the corrosion current density of decorative chromium coatings of approximately equal thickness, was found to be 16,14 · 10–7 A/cm2 with a corrosion rate of 0.018 mm/y. Thus the corrosion resistance of these two deposits are quite similar.
- Values derived from Nyquist plots of electrochemical impedance spectra were in agreement with data from the Tafel curves, with polarization resistance values for Sn-Co alloy coating was close to that of decorative chromium in the region of 200 000 Ωcm2 .
- In terms of operator health and safety, ease of process operation and effluent treatment and discharge, the Sn-Co plating baths, devoid of hexavalent chromium ions, chloride and fluorides as well as ammonia compounds, are preferable to conventional hexavalent chromium plating electrolytes.
- From the present work, it appears that the Sn-Co electrodeposition process reported here can be easily used for both rack and barrel plating, in contrast to chromium deposition.
The Sn-Co electrolyte reported here has a significantly lower metal content in the bath than conventional chromium plating solutions.
- Ghorbani.M, “Coating of metal “, Sharif University Press, Volume I, 2006.
- S K.CHO,B.H.RO,J.L PARK,” Tin- Cobalt Alloy Plating In A Pyrophosphate Bath”, The 4thAsia- Pacific Interfinish Congerss, October 27-30, 1998.
- Y. Song, D. Chin, “Current Efficinency and polarization behavior of trivalent Chromium Electrodeposition process”, Electrochimica Acta, 48(2002) 349–356.
- Arvydas Srvia,Zenius Mockus,” Kinetics of Sn and Co Codeposition in citrate solutions”,Electrochimica Acta 46(2000) 571–577.
- US Patent No 3,914,160 1973.
- E. Gomez, E. Guaus, J. Torrent,X . Alcobe and E. Valles,” Tin- Cobalt electrodeposition from sulfate- gluconate baths”,Journal of applied electrochemistry 31 (2001)349–354
- Dr. Manfred Jordan, “The Electrodeposition of Tin and its Alloys”, Eugen G. Leuze Publishers, Germany, 1995,ch 10 pp137–139.
- US Patent No 3,772,168, 1972
- Mordechay Schlesinger, Milan Paunovic,“ Modern electroplating”,Fourth Edition, 2000, part b physical Consideration, 29-44 Ch 18 . Peinciples of Alloy plating, pp 487–504.
- Brenner.A,”Electrodeposition of alloys”,Academic press, New York & London, vol 2, 1963
- US Patent No 4,035,249, 1976
- S.K. Cho, H.S. Han, C.K. Lee, C.I. Ahn, J.I, Park, “tin- cobalt alloy plating from the pyrophosphate bath” Materials Science forum vol. 439, pp 57–61,2003
- ZHANG J J, XIA Y Y,” Co-Sn alloys as negative electrode materials for eechargeable lithium batteries”, J Electrochem Soc53( 2006)1466–1471
PDF Version of the article |
> | Flash Version of the article |
| [qr-code size=”2″] | ||

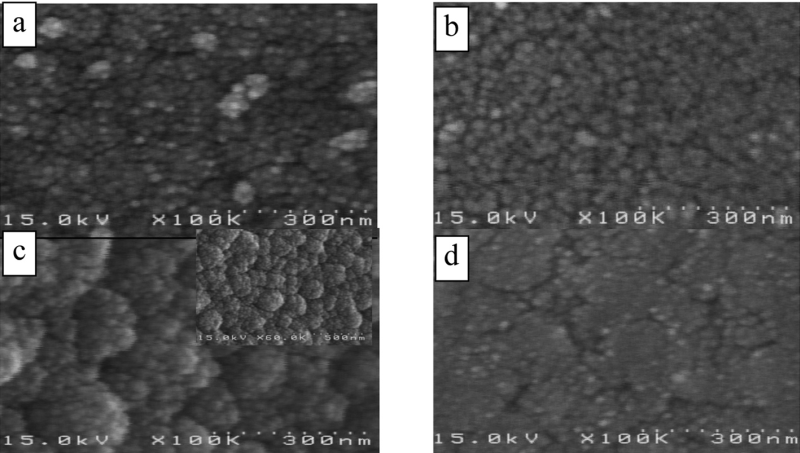
![Tab. 3: Effect change in [Sn]: [Co] in solution on deposit composition](https://www.jept.de/wp-content/uploads/2015/05/tab-3.png)
![Fig. 3: SEM images of deposit surfaces for [Sn] : [Co] ratios in solution: a) 1.6, b) 1.2, c) 0.9](https://www.jept.de/wp-content/uploads/2015/05/DamarcheliFig3.png)