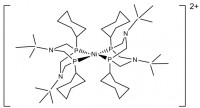
Gold and glassy carbon working electrode materials are compared as suitable substrates for the hydrogen oxidation reaction with Ni(PCy2Nt-Bu2)2(BF4)2 used as a catalyst. Voltammetric responses showing electrocatalytic hydrogen oxidation mediated by the homogeneous electrocatalyst Ni(PCy2Nt-Bu2)2(BF4)2 are identical at glassy carbon and gold electrodes, which shows that gold electrode can be used for hydrogen oxidation reaction.

